Abstract
Initial studies indicate an increased prevalence of patent foramen ovale (PFO) in migraineurs with aura, and an increased prevalence of migraine and migraine with aura in persons with PFO. Retrospective analyses of PFO closure suggest clinically significant improvements in migraine patterns. The aim of this study was to examine the prevalence of migraine in patients with PFO, the prevalence of PFO in migraineurs, and the effect of PFO closure on migraine. We conducted a quantitative systematic review of articles on migraine and PFO that met inclusion criteria, then reviewed, appraised, and subjected them to data extraction. Of 134 articles identified, 18 met a priori selection criteria. The estimated strength of association between PFO and migraine, reflected by summary odds ratios (ORs), was 5.13 [95% confidence interval (CI) 4.67, 5.59], and between PFO and migraine with aura the OR was 3.21 (95% CI 2.38, 4.17). The grade of evidence was low. The association between migraine and PFO was OR 2.54 (95% CI 2.01, 3.08). The grade of evidence was low to moderate. Six studies of PFO closure suggested improvement in migraine, but had a very low grade of evidence. The low-to-moderate grade of evidence from observational studies supports an apparent association between PFO and migraine. Although PFO closure seemed to affect migraine patterns favourably, the very low grade of available evidence to support this association precludes definitive conclusions.
Keywords
Introduction
An association between patent foramen ovale (PFO) and migraine has been identified in multiple studies. PFO is the result of incomplete fusion of the septum primum and septum secundum that normally occurs shortly after birth, when left atrial pressures exceed those in the right atrium. PFO occurs in about one-quarter of the general population (1). Some patients with PFO have right-to-left shunting that occurs either at rest or with Valsalva manoeuvre. Circulatory bypass of normal pulmonary filtering via PFO may allow for paradoxical embolism and passage of higher concentrations of vasoactive substances into the arterial system.
Migraine is a common and disabling disorder with a prevalence of 8–13% in the population of the Western hemisphere (2, 3). About one-third of migraineurs have at least an occasional migraine with aura (MA) (4). Although migraine is most commonly considered a primary headache disorder, it may also occur as a symptomatic manifestation of an underlying disorder such as cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL), mitochondrial encephalomyopathy, lactic acidosis, and stroke-like symptoms (MELAS), arteriovenous malformations, or cerebral ischaemia (5–7). In a subgroup of migraineurs, PFO may act as a causal or triggering factor.
Available data suggest that PFO is more common in migraineurs with aura and that MA is more common in patients with PFO (8–11). In addition, retrospective studies examining PFO closure and the effect on migraine suggest a benefit to PFO closure (9, 12). Our objective was to systematically review and grade the available data about the prevalence of PFO in migraineurs with and without aura, the prevalence of migraine with and without aura in patients with PFO, and the effect of PFO closure on migraine.
Materials and methods
Search strategy
Two investigators reached consensus on the search strategy to be used and independently searched the MEDLINE database for all potentially relevant articles published between 1966 and April 2007 using the following search terms: (‘heart septal defects, atrial’ [MeSH] [exploded] OR ‘patent foramen ovale’ [search term]) AND (‘migraine disorders’ [MeSH] [exploded] OR ‘migraine’ [search term]). Only articles published in English were selected for review. We also searched reference lists of identified articles, review articles, and the authors' own personal files, and consulted other experts for additional articles. The title and abstract of each identified article were independently screened by the two investigators for possible inclusion. Disagreements were resolved by discussion to reach consensus. A third party was not required.
Study selection criteria
The study selection criteria were established a priori by investigator consensus. We selected primary research articles or meta-analyses describing observational studies that examined the association between PFO and migraine. PFO must have been detected either by angiography, transthoracic echocardiography with peripheral injection, transoesophageal echocardiography with peripheral injection, or transcranial Doppler ultrasonography with injection. Migraine diagnoses must have been based on the diagnostic criteria of the International Headache Society (IHS). For the outcome studies, data about headache frequency or severity must have been collected for at least 3 months before and after PFO closure. The established selection criteria were independently applied to each screened article by two investigators.
Study quality assessment and data extraction
The quality of each included study was judged by the two independent investigators using a system created by members of the Grades of Recommendation Assessment, Development and Evaluation (GRADE) working group (Appendix I) (13).
Data extraction was conducted by the two independent reviewers, and disagreements were resolved by discussion to reach consensus.
Statistical methods
For the meta-analyses of the case–control observational studies, the odds ratio (OR) was the parameter selected to summarize each study. A fixed-effects meta-analytical technique was used to combine the results of the individual studies, and summary ORs and 95% confidence intervals (CIs) were calculated. Visual evaluation of heterogeneity was conducted. A funnel plot was constructed to explore the possibility of publication bias.
Results
Search results
The search strategy yielded a total of 134 articles. Screening of titles and abstracts of all 134 articles yielded 29 that were further investigated for possible inclusion. Of these, 18 different articles met inclusion criteria for one or more objectives (Fig. 1). Eleven articles met inclusion criteria for the study of the prevalence of migraine in patients with PFO (with six of 11 providing data on both patients and controls); seven met criteria for the prevalence of PFO in migraineurs (with five of seven providing data on both cases and controls); and six met criteria for the effect of PFO closure on migraine. Six studies met criteria for more than one objective. Eleven of the total 18 studies could be used for meta-analyses since they included cases and controls.

Flow diagram demonstrating search results.
Migraine in patients with PFO
Eleven studies addressing the prevalence of migraine in patients with PFO met our selection criteria (Table 1) (8, 9, 12, 14–21). These studies evaluated a total of 1517 patients with PFO for the presence of migraine. The prevalence of migraine in patients with PFO ranged from 22.3% to 64.3%; the prevalence of MA ranged from 12.9% to 50%; and the prevalence of migraine without aura (MoA) ranged from 2.8% to 25%. Six studies included control groups, which allowed for the calculation of ORs to estimate the strength of the association between migraine and PFO. The ORs for migraine and PFO ranged from 1.82 to 5.88, with a summary OR of 5.13 (95% CI 4.67, 5.59) (Fig. 2). This parameter reflects the ratio of odds of migraine in a group of patients with PFO (cases) to the odds of migraine in a group of patients without PFO (controls). The ORs for PFO and MA ranged from 1.74 to 9.22 with a summary OR of 3.21 (95% CI 2.38, 4.17) (Fig. 3). This parameter reflects the ratio of odds of aura in a group of migraine patients with PFO (cases) to the odds of aura in a group of migraine patients without PFO (controls). The grade of evidence for these studies was determined to be low, which suggests that further research is likely to change the estimate and precision of the strength of these associations.
n = 1517.
Values are number (percentage) unless indicated otherwise.
MA, migraine with aura; MoA, migraine without aura; PFO, patent foramen ovale.

Strength of association between patent foramen ovale (PFO) and migraine. The odds ratios (ORs) for migraine and PFO ranged from 1.82 to 5.88, with a summary OR of 5.13 [95% confidence interval (CI) 4.67, 5.59].

Strength of association between patent foramen ovale (PFO) and migraine with aura. The odds ratios (ORs) for PFO and migraine with aura ranged from 1.74 to 9.22, with a summary OR of 3.21 [95% confidence interval (CI) 2.38, 4.17].
PFO in migraineurs
Seven studies about the prevalence of PFO in migraineurs met the selection criteria (Table 2) (10, 11, 22–26). Four studies included migraineurs both with and without aura, whereas the other three included only migraineurs with aura. A total of 906 migraineurs was evaluated for the presence of PFO, which included 665 with MA and 241 with MoA. The prevalence of PFO in migraineurs ranged from 39.8% to 72.0%. The prevalence of PFO in migraineurs with aura ranged from 40.9% to 72.0% and in migraineurs without aura from 16.2% to 33.7%. Five of the studies included control groups, which allowed for calculation of ORs to estimate the strength of the association between PFO and migraine. The ORs ranged from 1.87 to 5.88 with a summary OR of 2.54 (95% CI 2.01, 3.08) (Fig. 4). This parameter reflects the ratio of the odds of PFO in a group of patients with migraine (cases) to the odds of PFO in a group of patients without migraine (controls). Studies were given a low to moderate grade, which suggests that further research is likely or very likely to have an impact on the magnitude and precision of the estimated association.
Patent foramen ovale in migraineurs∗

n = 906.
Values are number (percentage) unless indicated otherwise.
MA, migraine with aura; MoA, migraine without aura; NR, not reported; PFO, patent foramen ovale.

Strength of association between migraine and patent foramen ovale (PFO). The odds ratios (ORs) ranged from 1.87 to 5.88, with a summary OR of 2.54 [95% confidence interval (CI) 2.01, 3.08].
PFO closure and effect on migraine
Six studies, all retrospective, met our selection criteria for evaluation of the effect of PFO closure on migraine (Table 3) (8, 9, 12, 17, 20, 23). One of these six studies used a control group of patients with PFO and migraine who were managed medically. Patients in five of the six studies received antiplatelet therapy for 6 months after PFO closure; the report for one study did not indicate the duration of antiplatelet use. Follow-up varied, ranging from 3 months to 1 year. Study grade was determined to be very low in five of these studies and low in one study. Therefore, any estimate of effect is uncertain. Studies were excluded if the patient population included patients with atrial septal defects, and the results were reported in conjunction with those for PFO closure. Four of the studies reported data qualitatively, assigning patients to an ‘improved’ group without specifically defining such improvement. Thus, the magnitude of change in this group and among studies is impossible to determine. In addition, one study reported only pre-PFO and post-PFO closure prevalence of migraine without further quantitative data and without individual patient data; this study was excluded from analysis.
Patent foramen ovale closure and its effect on migraine
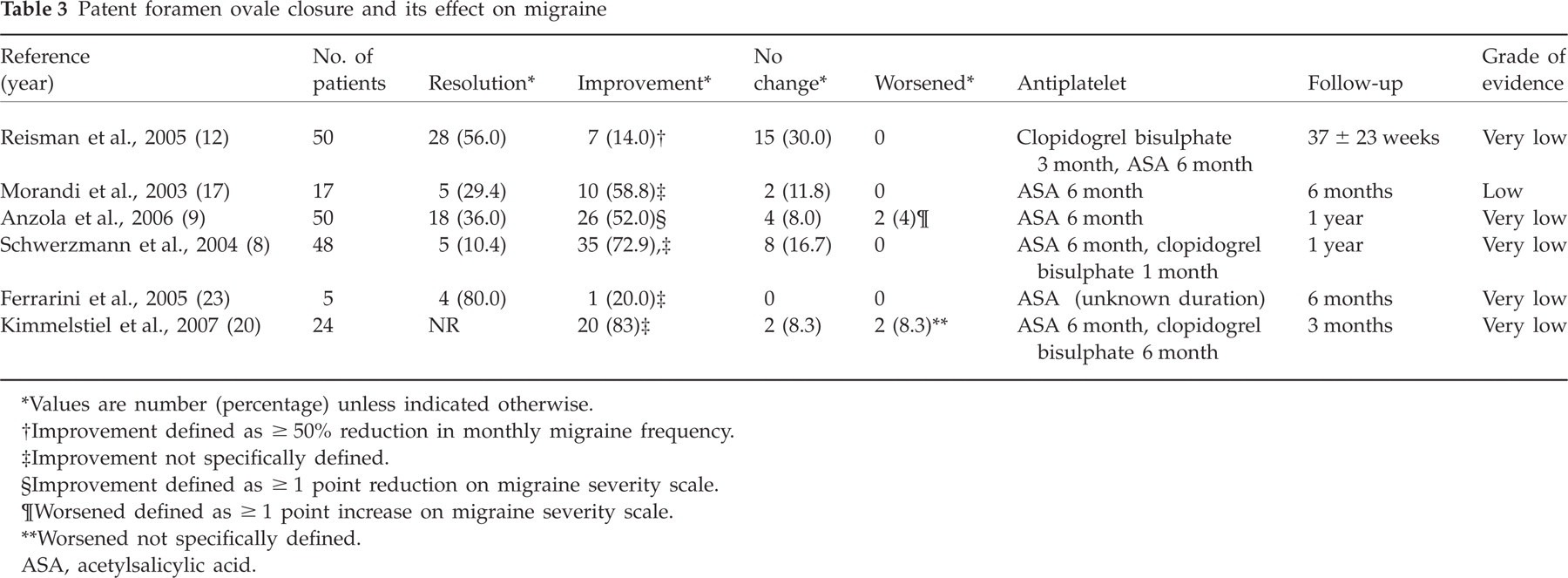
Values are number (percentage) unless indicated otherwise.
Improvement defined as ≥ 50% reduction in monthly migraine frequency.
Improvement not specifically defined.
Improvement defined as ≥ 1 point reduction on migraine severity scale.
Worsened defined as ≥ 1 point increase on migraine severity scale.
Worsened not specifically defined.
ASA, acetylsalicylic acid.
The effect of PFO closure on migraine was studied in a total of 194 patients. In migraineurs with and without aura, resolution of headaches occurred in 10.4–80.0%. Improvement was reported in an additional 14.0–83%. In one study that defined improvement as a ≥ 50% reduction in monthly headache frequency, seven of 22 migraineurs without resolution of headaches had improvement. In one study that defined improvement as a one point or greater decline on a ‘severity scale’, 26 of 32 migraineurs without headache resolution had improvement. In four studies that did not specifically define ‘improvement’, 66 of 94 patients improved.
Assessment for heterogeneity and publication bias
For heterogeneity and publication bias assessment, observational data from the 11 studies examining the prevalence of migraine in patients with PFO (Table 1) and the prevalence of PFO in migraineurs (Table 2), which included both case and control groups, were pooled to calculate the overall estimated strength of the association between these two entities [summary OR of 4.52 (95% CI 3.97, 5.08)]. Significant heterogeneity was found among the point estimates of the ORs derived from these studies (Figs 2–4). We explored this visible heterogeneity along the lines of patient (case and control) characteristics, diagnostic parameters (migraine and PFO), follow-up, outcome measures, study methods and quality. The funnel plot (Fig. 5) demonstrates asymmetry. There is a paucity of small observational studies that reported high ORs of 6–8 at the bottom right-hand corner to match the ones reporting small ORs of 2–4 at the bottom left-hand corner of the plot. This funnel plot asymmetry suggests that publication bias may be a factor.

Funnel plot of studies reporting the association between migraine and patent foramen ovale (PFO). Each circle represents the point estimate odds ratio for an individual study. Although a pattern resembling an inverted funnel with smaller studies symmetrically distributed around the pooled estimate (arrow line) should be expected, the expected small studies in the bottom right quadrant are missing. This finding suggests publication bias.
Discussion
The association between PFO and migraine has recently received considerable attention from the medical community. Much of the data addressing this relationship has been observational and extracted from patients with a presumed paradoxical embolism. We undertook this quantitative systematic review to evaluate the strength of existing data that suggest an increased prevalence of MA in patients with PFO, an increased prevalence of PFO in migraineurs with aura, and improvement in migraine patterns after PFO closure.
We established our selection criteria with the intent of including a substantial proportion of available data. It was determined necessary for all the patient population under study to have had PFO, for PFO to be diagnosed by an acceptable and validated diagnostic method, and for migraine diagnoses to be made according to the criteria of the IHS. In addition, the closure studies must have collected headache data for at least 3 months before and after PFO closure. Application of these criteria yielded only 11 studies of migraine prevalence in patients with PFO, seven studies of the prevalence of PFO in migraineurs, and six studies of migraine patterns after PFO closure.
Our evaluation of the studies examining the prevalence of migraine in patients with PFO led us to conclude that there is a low grade of evidence that MA is more common in this patient group. This same trend was not found for MoA. Five studies included in this systematic review examined the relationship between shunt size and migraine (15, 18, 19, 24, 25). Each study concluded that MA was more prevalent in patients with larger shunts.
These studies had several shortcomings. All the patients studied were viewed as having symptomatic PFO and were being studied because of presumed paradoxical embolism or decompression illness. However, it is not clear whether this study population and these results are representative of the general population with PFO, many of whom are asymptomatic. Since larger shunts may be more likely to be associated with both paradoxical embolism and migraine, it is possible that use of these populations could result in an overestimate of migraine prevalence. Studies with control groups that examined the prevalence of migraine in patients who were suspected to have a PFO, but in whom none was detected, were used in only four studies. Thus, much of the data were compared with historical controls, which led to the conclusion that there is an increased prevalence of MA.
Available data suggest that there is a low-to-moderate grade of evidence that PFO is more prevalent in migraineurs with aura than in the general population. It does not seem that PFO is more prevalent in migraineurs without aura, but the total sample size is small. Five of the studies included a control group.
Although 10 of the 11 studies included in this meta-analysis support an association between migraine and PFO, heterogeneity of ORs between studies was apparent. Heterogeneity is most likely to be due to differences in the characteristics of the case and control groups. Patients (cases) were identified by different means, including out-patient neurology consultations for MA, emergency room visits for MA, complaints of decompression illness, and treatment for ischaemic stroke. Controls were variably identified, some as ‘healthy’ volunteers from the community or medical centre, whereas other subjects presented with decompression illness, acute stroke, a family history of PFO, and atrial septal mobility identified by echocardiogram. In addition, differences in the ages and sex of patients among studies may have contributed to heterogeneity.
The underlying mechanism of the possible relationship between PFO and migraine remains speculative. It has been postulated that PFO may allow venous-circulating, migraine-triggering, vasoactive chemicals to bypass the pulmonary filter and reach the cerebral circulation to induce a migraine attack (27). Paradoxical emboli themselves appear to have a propensity for the posterior circulation, the area in which hypoperfusion occurs during a migraine aura (28). Perhaps the most compelling suggestion is that a particular genetic composition might govern the co-development of atrial septal abnormalities and migraine (29). We have presented the best and most precise estimate to date of the strength of the association between these two conditions on the basis of our meta-analysis of observational studies, which included a total of 2636 subjects. Neither the heterogeneity nor the presence of possible publication bias poses a significant threat of overturning the conclusions. Ten of 11 individual studies included in the meta-analysis reported a statistically significant OR in favour of an association. If a publication bias in this medical literature exists, we estimate that, under its influence, we may have erroneously underestimated the strength of the association rather than the converse.
Wammes-van der Heijden et al. systematically reviewed the available medical literature to quantify the strength of the relationship between right-to-left shunt and migraine in patients with and without ischaemic stroke (30). They elected to conduct their analysis unidirectionally, assuming that right-to-left shunt is the exposure (or independent variable) and that migraine is the outcome (or dependent variable). They reported pooled ORs of 3.5 (95% CI 2.1, 5.8) for patients without ischaemic stroke and 2.1 (95% CI 1.6, 2.9) for patients with ischaemic stroke. Their systematic review contained multiple methodological weaknesses. They neglected to specify whether two or more reviewers independently searched, screened, applied criteria, and extracted data from the articles. They did not establish a set of rigorous inclusion and exclusion criteria, did not evaluate the quality and grade of evidence, did not determine if significant heterogeneity was present before pooling, and did not interrogate for the possible existence of publication bias.
In a meta-analysis, results from several primary studies are combined statistically. The pooling of primary study results can be done using either a fixed-effects model or a random-effects model. The fixed-effects model, selected for this analysis, restricts inferences to the set of studies included in the meta-analysis and assumes that there is a single true value underlying all the study results. By contrast, the random-effects model assumes that the studies included are a random sample of a population of studies addressing the question posed in the meta-analysis. Compared with the fixed-effects model, the random-effects model gives smaller studies proportionally greater weight in the pooled estimate. Consequently, the direction and magnitude of the pooled estimate are influenced more by smaller studies. If the smaller studies are farther from the null result, the random-effects model will tend to produce larger estimates of association than will the fixed-effects model.
Our main reservation about using a random-effects model was its increased susceptibility to publication bias as a result of its increased weighting of small studies. A pooled estimate derived from a random-effects model would be more susceptible to publication bias, a phenomenon that primarily affects smaller studies (31). Nonetheless, we also conducted the meta-analyses using a random-effects model and confirmed that there were no substantial differences in the magnitude or direction of the pooled estimates of association.
Although initial data suggest improvement in migraine patterns after PFO closure, no conclusions can be drawn from existing data. The six studies meeting our selection criteria included a total of 194 patients with migraine who underwent PFO closure. A control group was used in only one study. However, the control group was a group of migraineurs with PFO who continued to be managed medically. A more appropriate control group would consist of migraineurs with PFO who undergo sham closure given the possibility of a large placebo response to an interventional cardiac procedure. In addition, in each of the studies, data about headache patterns were collected partially or completely in retrospect. Retrospective data collection opens the door for significant recall bias. All patients in these studies were also treated with antiplatelet medication after PFO closure. Although antiplatelet medication is probably necessary to reduce the risk of possible thrombus formation on the closure device before endothelialization, the magnitude of the effect of daily antiplatelet use on migraine patterns in each study is unmeasured. If antiplatelet medications were administered during the same period in control patients who underwent sham closure, both cases and controls would be treated more similarly. Also, it may be faulty to extrapolate data to the general migraine population that were obtained from patients who underwent PFO closure most commonly for suspected paradoxical embolism or decompression illness.
Results from prospective, sham-controlled trials of PFO closure for the treatment of migraine are expected in the future. To date, one such study, MIST (Migraine Intervention with STARflex® Technology) has been completed. Although only the results from the diagnostic phase of the study have been published in abstract form, response to closure has been presented in oral format at scientific meetings (American College of Cardiology 55th Annual Scientific Session; American Academy of Neurology 58th Annual Meeting) (32). Subjects were aged 18–60 years, met IHS diagnostic criteria for MA, were refractory to two or more classes of prophylactic medications and had at least five migraine headache days and seven headache-free days per month. Those with moderate to large PFO detected by transthoracic echocardiography were randomized to sham procedure (n = 73) or PFO closure (n = 74). All subjects were treated with aspirin and clopidogrel for 3 months following intervention. Outcomes were determined during months 4–6 post intervention. The study failed to meet its primary end-point of complete migraine resolution in 40% of subjects in the treatment arm. Three patients in both the treatment and sham arm had complete migraine resolution. Results on secondary end-points cannot be fairly interpreted until all results are made public. Five subjects in the implant group had complications, which included cardiac tamponade, pericardial effusion, bleeding at the puncture site, atrial fibrillation, and atypical chest pain. Four subjects in the sham group had reported complications including bleeding at the puncture site, bleeding secondary to antiplatelets, and stroke (4 months after randomization).
Conclusion
There is evidence of a low grade for an increased prevalence of migraine in patients with PFO, perhaps limited to patients with a large PFO. In addition, there is low to moderate evidence of an increased prevalence of PFO in patients with migraine. Although an overall association was evident between PFO and migraine, no association was found between PFO and MoA. Furthermore, the number of patients with MoA was quite small and did not contribute significantly to the overall relationship between PFO and migraine. Given very low to low evidence, no conclusions can be drawn about the efficacy of PFO closure for treatment of migraine. In addition, the data concerning the safety of PFO closure in migraineurs are insufficient to draw conclusions. Studies are needed that focus on the prevalence of PFO in migraineurs without aura. Although initial data from the MIST trial are disappointing, additional prospective, randomized, sham-controlled trials of PFO closure for the treatment of migraine are currently enrolling and will help to determine the efficacy and safety of such an intervention.
Footnotes
Acknowledgements
We would like to thank Nichole L. Boruff and Thomas E. Bibby for their graphic design of the figures. Editing, proofreading and reference verification were provided by the Section of Scientific Publications, Mayo Clinic.

