Abstract
Aim: To compare the effectiveness of early or late triptan intake in the treatment of acute migraine attacks.
Methods: The TEMPO study was a French prospective, multicentre, two-phase study conducted in neurological practice. Two-hundred-and-ten migraine patients who were regular triptan users were enrolled. In the first phase, patients treated three attacks as they usually did. In the second phase, those who initially practiced late dosing ( ≥ 1 hour after headache onset) were instructed to change to early dosing ( < 1 hour).
Results: A total of 144 patients completed the first phase. Seventy-nine patients constituted the ‘early dosers’ group and 65 patients the ‘late dosers’ group. In this phase, early dosing produced higher rates (n = 38; 52.8%) of freedom from pain at 2 hours in at least two of three attacks compared with late dosing (n = 19; 30.2%; p < 0.01). In the second phase, switching from late to early dosing following the physician’s instruction (n = 42 patients) also improved the rates of freedom from pain at 2 hours (from 38.1% (n = 16) to 53.7% (n = 22); p < 0.05).
Conclusion: This suggests that advising patients on the importance of early triptan intake after headache onset may help improve the efficacy of acute migraine treatments.
Introduction
Triptans, which are selective agonists of serotonin 5-HT1B/1D receptors, are widely used for the acute treatment of migraine attacks. Although their high efficacy has been amply demonstrated, how to use triptans optimally remains an undecided question, and less than half of patients achieve complete headache resolution (pain-free state) within 2 hours after dosing in at least two of three triptan-treated migraine attacks (1). Clinical data have suggested that an important factor accounting for the variable efficacy of triptans within patients and between attacks might be the time of treatment intake relative to headache onset. In this respect, it has been suggested that early triptan administration can prevent the sensitization of central pain neurons that leads to cutaneous allodynia, whereas late triptan administration cannot counteract established sensitization and therefore may be less effective in inducing a pain-free state (2–5). In addition, part of the reason that patients may fare better when treating early symptoms is that headache pain is aborted when the headache is still mild and before severe pain and handicap develops. Several studies have indicated that early triptan intake is more efficacious than late treatment on various clinical endpoints such as pain-free state at 1 or 2 hours, sustained pain-free state between 2 and 24 hours, recurrence rate and rate of redosing (6–13). A recent controlled study (the ‘Act when mild’ study) that compared the response to almotriptan taken early (within 1 hour of headache onset) and when pain intensity was mild with that when it was taken later in the course of migraine attack (when pain had become moderate or severe) found that early treatment provides significant and clinically relevant improvement in triptan efficacy (14).
The TEMPO study, a prospective study carried out in daily neurological practice, was specifically aimed at evaluating the within-patient change in treatment efficacy when the time of triptan intake was reduced from more than 1 hour (late dosing) to less than 1 hour (early dosing) after the onset of migraine headache. The assessment of the influence of the time of triptan intake on clinical outcomes was primarily based on the observed pain-free rate 2 hours after dosing in at least two of three treated consecutive attacks. Another objective of the study was to assess therapeutic patient education in daily practice by identifying the influence of physician advice to migraine patients on their behaviour as regards the length of time before triptan intake.
Methods
This was an open-label multicentre study carried out in France over 6 months in daily neurological practice. The study was conducted in accordance with the requirements of good clinical practice and local laws. Because the study required changes in acute migraine management by the patients, the protocol was submitted to the institutional review board of the University Hospital of Nice, France, for approval, and the selected patients provided written informed consent before inclusion. Given that individual data were computerized, the study was also submitted to the French National Commission on Data Processing and Liberties for approval. Individual patient data were anonymized on the case report form.
Patient selection
Patients were recruited from people with headaches aged 18–65 years consulting a neurologist, who confirmed the diagnosis of migraine without aura according to the second edition of the International Classification of Headache Disorders (15). Other patient selection criteria were: 1) currently using an oral triptan to treat their migraine attacks; 2) having experienced the first migraine attack before the age of 50; 3) having experienced at least three migraine attacks during the past 3 months but not more than six attacks during the last month, or had not more than 15 days with headaches per month during the last 3 months; 4) being able to differentiate a migraine attack from tension-type headache; 5) not using analgesics more than 15 days per month, or triptans or opioids more than 10 days per month; 6) having had no change in any migraine prophylactic treatment within the last 3 months and not requiring a prophylactic treatment at the inclusion visit.
Study design
The study was divided into two successive phases. During the first phase, patients were requested to keep a detailed record of the next three consecutive migraine attacks, including the following items: 1) time of headache onset; 2) triptan used; 3) time of triptan intake; 4) pain intensity at the time of treatment intake and 2 and 24 hours later; 5) absence or presence of signs of facial allodynia at the time of treatment intake and 2 hours later. Allodynia was detected using a questionnaire similar to those used in studies dealing specifically with this clinical aspect of migraine attacks (16,17). This questionnaire captured information about the existence of pain or unpleasant sensations caused by wearing earrings, a necklace, eyeglasses, or a hat or cap, by running a hand through one’s hair, brushing or washing hair, shaving or taking a shower when water hits the face, and about the occurrence of a burning sensation on the scalp. Patients were allowed to take a second dose of the triptan if the headache recurred between 2 and 24 hours after the first dose. Analgesic rescue medications were authorized after this second dose, as needed.
The reference study visit took place between phase I and phase II, after the patient had treated three consecutive migraine attacks. A single treatment strategy regarding the time of triptan intake relative to headache onset was defined for the next three consecutive attacks (phase II) through straightforward instruction by the neurologist, depending on the data collected from phase I. If the triptan had been taken less than 1 hour after headache onset in at least two of three attacks (early treatment), the patient was asked to treat the next three attacks similarly. Alternatively, if the triptan had been taken more than 1 hour after headache onset in at least two of three attacks (late treatment), the patient was instructed to switch to systematic early dosing (i.e. to take the triptan less than 1 hour after headache onset). All other treatment conditions remained unchanged between phases I and II, notably the individual triptan used.
Evaluation criteria
Efficacy outcomes were assessed at the end of each series of three migraine attacks. The primary efficacy endpoint was the pain-free rate at 2 hours post-dose in at least two of three migraine attacks. Secondary efficacy endpoints included pain-free rate at 2 hours post-dose in three migraine attacks, the rate of headache relief (passage from severe or moderate headache to mild or none) at 2 hours post-dose in at least two of three attacks or in three attacks, the pain-free rate at 2 hours post-dose in moderate or severe attacks, the sustained pain-free rate (from 2 to 24 hours) in at least two of three attacks or in three attacks, relief of migraine-associated symptoms (nausea, vomiting, photo- and phonophobia), the rate and time of redosing with the triptan, and the rate and time of use of an alternative rescue medication. Patient satisfaction with treatment was evaluated on a 10 cm vertical visual analogue scale (VAS) scored from 0 (not satisfied at all) to 10 (totally satisfied). Finally, when applicable, reasons for delayed treatment of migraine attacks were collected.
Statistical analysis
The main analysis was carried out on the population of patients who treated three migraine attacks in each phase according to the study protocol and who switched from late to early dosing as advised by the physician. Treatment efficacy was also compared between dosing times (early or late dosing in at least two of three migraine attacks) on the whole population of patients who participated in phase I of the study. Differences in therapeutic behaviours during phase II (willingness to change to early triptan intake upon physician advice vs refusal to switch from late to early triptan intake) were analysed in terms of efficacy outcomes during phase I. Logistic regressions and generalized estimation equation (GEE) models (for binomial response) were performed to identify variables significantly influencing efficacy outcomes.
Within-patient comparison of treatment efficacy between the two study phases was performed using McNemar’s test on the group of patients with available data in the two phases of the study. Between-dosing time analyses of efficacy outcomes and comparisons between patient groups were performed using the χ2 test or Fisher’s exact test as appropriate. Logistic regression analyses were carried out at the patient level using the Statistical Analysis System (SAS Institute Inc., Cary, NC) Stepwise procedure. For the analysis at the attack level, GEE modelling was performed taking into account the potential correlation of headache variables within the same patient using the Genmod SAS procedure.
Numerical data were summarized by the usual descriptive statistics (number of values, mean, standard deviation (SD)). Categorical data were summarized by the number of values and relative frequency in each modality. A probability value of < 0.05 was taken as statistically significant.
Results
Study progress
Seventy-five neurologists recruited 210 migraine patients, of whom 200 were included in the study. Seven patients were subsequently excluded for violations of the inclusion criteria (age < 18, no oral triptan used, migraine beginning after age 50, no information about prophylactic treatment). During phase I of the study, 148 patients (76.7%) treated three migraine attacks according to study protocol, while five patients (2.6%) treated two attacks, four (2.1%) treated one attack, one (0.5%) treated no attack, and data were missing for 35 patients (18.1%). One patient withdrew from the study owing to lack of treatment efficacy. Thus, 147 patients were recruited for phase II of the study but the time of dosing relative to headache onset was unknown for three of these, who were excluded. Finally, of the 144 patients who entered phase II of the study, 79 (54.9%) had treated at least two of their phase I headaches within 1 hour (‘early interventionists’) and were advised to continue to treat the attacks with no change in time of dosing. The remaining 65 (45.1%), who had treated at least two of their phase I headaches at least 1 hour after headache onset (‘late interventionists’) were advised by the neurologist to switch from late to early dosing. In fact, 42 ‘late interventionists’ (64.6%) switched to early treatment following the physician’s advice whereas 23 (35.4%) did not modify their usual practice of late triptan intake. The flow of patients through the study is depicted in Figure 1.
Patient flow diagram.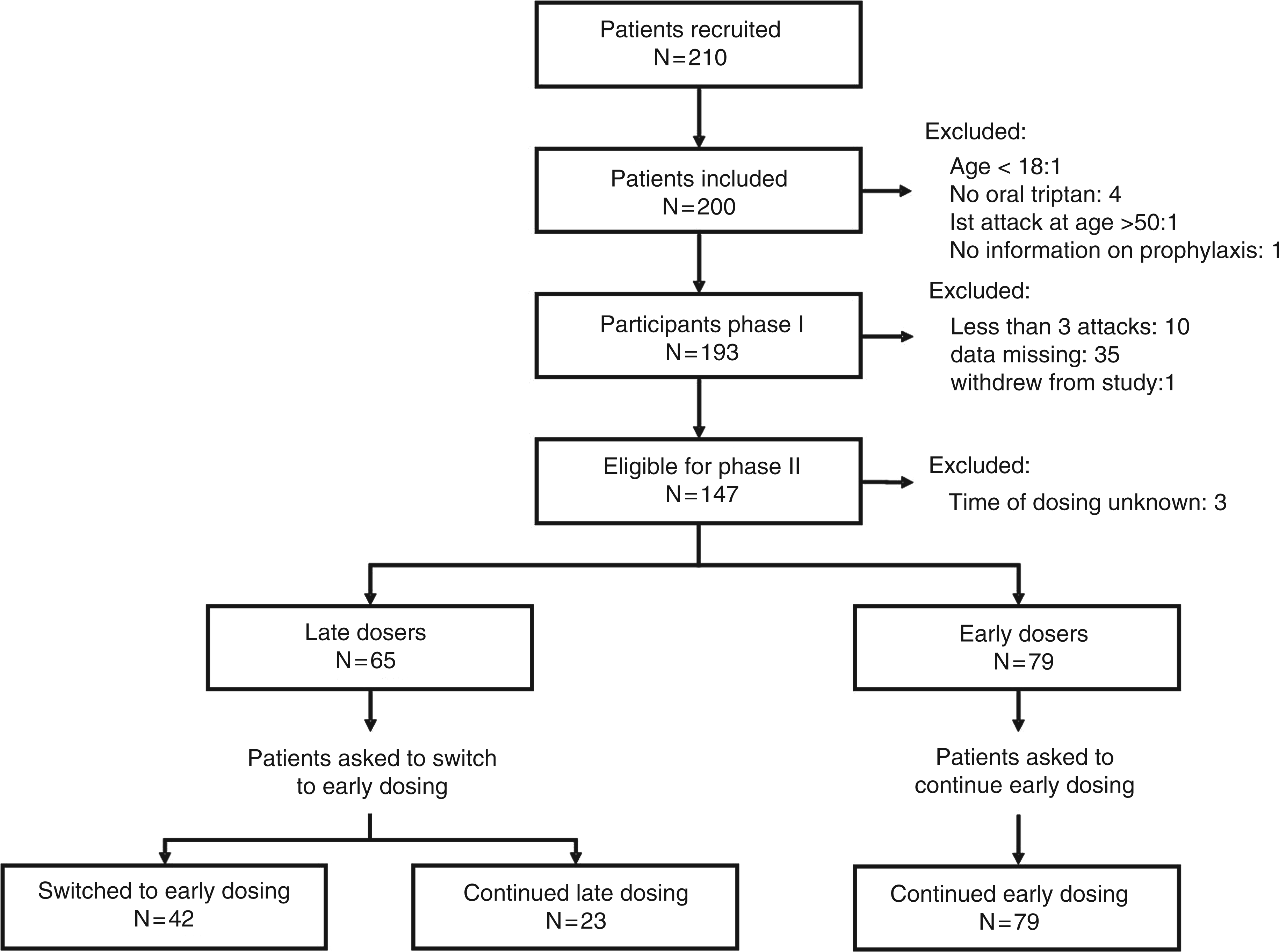
Patient population
Patient characteristics

Triptan efficacy according to dosing time during study phase I
Treatment efficacy on migraine headaches according to dosing interval in patients who treated three migraine attacks with a triptan during phase I of the study

Patient satisfaction and reasons for delaying dosing
Among the population of patients who treated three attacks in phase I, there was no significant difference between ‘early interventionists’ and ‘late interventionists’ regarding satisfaction with the treatment as measured with the VAS (mean scores: 6.5 ± 2.5 vs 6.2 ± 2.5 respectively). The main reasons for delaying treatment as stated by the ‘late interventionists’ in phase I were the belief that the attack would resolve spontaneously (67.7%) or that they should reserve the triptan only for severe attacks (35.4%), the fact that the treatment was not available when the attacks occurred (29.2%), concern about the risk of possible adverse effects (7.7%), the use of an analgesic first (4.6%), difficulty in differentiating a migraine attack from a simple headache (3.1%), occurrence of nocturnal attacks (1.5%), and concern about possible loss of efficacy after repeated triptan use (1.5%).
Comparison of efficacy between phase I and II
Of the 65 patients classified as ‘late interventionists’ at the end of phase I and requested to switch from late to early treatment during phase II, 19 (30.2%) were pain-free at 2 hours in at least two of three migraine attacks in phase I (data missing for two patients). At the end of phase I, the physician advised these patients to switch to early dosing. Among the 52 patients in this group with assessable data at the end of phase II, 29 (55.8%) were pain-free at 2 hours in at least two of three attacks. The within-patient difference in pain-free rates between phases I and II was highly significant (p = 0.005, McNemar’s test).
Among these initially ‘late interventionists’, 42 (64.6%) actually switched from late to early treatment after physician’s instruction whereas 23 (35.4%) continued to treat after 1 hour. There were no differences between these two subgroups in terms of age, gender or headache history. Patients who continued to treat their headaches late were asked why they did so. Although most did not reply (12 out of 23 patients), the reasons given were generally related to severity. Six patients said that they thought that their headache would go away by itself, four said that they preferred to keep their medication for a more severe attack, and one did not have their medication with them at the time of the onset of the attack.
Within-patient comparison of pain-free rates in patients who treated two series of three migraine attacks in phases I and II of the study, according to dosing time with respect to headache onset

Data are presented as the number of patients (%); aMcNemar’s test.
No similar comparison was possible in the group of 23 patients classified as ‘late interventionists’ at the end of study phase I and who did not change their dosing regimen after physician’s instruction, owing to a high rate of patients without assessable data at the end of study phase II (12 patients, 52.2%).
Headache relief rates at 2 hours in at least two of three attacks were 66.7% before instruction and 80.5% after instruction for the 42 ‘late interventionists’ who switched from late to early dosing, versus 80.6% and 82.6%, respectively, for the 79 ‘early interventionists’ who were instructed to carry on with early dosing.
Variables influencing study efficacy outcomes
Logistic regression analysis of factors influencing efficacy outcomes across the six attacks recorded during the study

Treatment tolerability
The most frequent adverse events (AEs) reported by the patients after triptan use in phase I of the study included drowsiness (13.0% of patients), nausea and vomiting (7.8%), euphoria, light-headedness or dizziness (6.2%), dry mouth, swollen lips or throat (4.1%), and tight throat (3.6%). As regards AE intensity, 45.3% of AEs were of mild intensity, 43.4% of moderate intensity and 11.3% of severe intensity. No AE was serious. The relationship to treatment was considered probable or very probable in 70.0% of cases. Most AEs (92.0%) resolved spontaneously without specific intervention. The number of AEs reported decreased dramatically from the first to the sixth treated attack (from 39.6% to 3.8% of all AEs reported), and from the first to the second tablet used (from 92.6% to 7.4% of all AEs reported).
Discussion
The aim of the TEMPO study was to assess potential optimization of acute migraine management in daily neurological practice through early triptan intake (less than 1 hour after headache onset) following advice by the physician. To this end, the efficacies of early and late triptan intake were compared in two ways and at two stages: between groups of patients spontaneously treating their headaches early or late (study phase I), and within patients before (study phase I) and after advice by the physician (study phase II). Three groups of patients were distinguished: 1) patients who switched from late to early treatment following their physician’s advice; 2) patients who did not follow their physician’s advice and carried on with late treatment; 3) patients who continued early treatment as they had done in phase I of the study. Our results indicate that spontaneous early intake achieved better control of migraine attacks than late intake, that switch from late to early intake following advice from the physician improved treatment efficacy. Overall analysis of all migraine attacks treated during the study showed that early intake was significantly associated with greater rates of pain-free state, headache relief and sustained pain-free state.
Previous studies have already suggested that triptan efficacy is greater when treatment is administered early in the development of a migraine attack. However, the largest of these, the ‘Act when mild’ study, the early intake criterion was associated in the design of the study with a mild severity criterion, which is itself usually associated with better efficacy outcomes than moderate or severe pain levels (14). Other studies have been either experimentally designed prospective trials evaluating individual triptans or retrospective patient registries; all have compared parallel groups of early and late interventionists (18). The originality of the TEMPO study was to assess prospectively whether headache outcomes in patients who treat late could be improved by the physician actively advising patients to advance the time of dosing. The study was performed in a naturalistic setting of migraineurs treated with triptans and followed by neurologists in a daily practice setting in France. For this reason, the findings should be relevant to the everyday care of patients with migraine.
The change in the rate of pain-free state in at least two of three migraine attacks was the primary efficacy measure retained for the TEMPO study, as currently recommended for therapeutic trials in migraine (19). The switch from early to late triptan intake increased the rate of pain-free state at 2 hours in at least two attacks from 38% with late treatment to 54% with early treatment (p < 0.05). These results are strikingly similar to those obtained in the ‘Act when mild’ study of single migraine attacks, which reported a pain-free rate of 37.5% at 2 hours after late administration of almotriptan compared with 53% after early treatment (within 1 hour of headache onset when pain intensity was mild) (14). The within-patient difference remained significant even when extended to all late interventionists who were advised to switch, including those who did not follow this instruction (30% versus 56%; p = 0.005). However, this benefit may be somewhat overestimated given that outcome data were missing for over 50% of patients who failed to follow the advice. Nevertheless, this result may give an indication of what physicians might expect to achieve in their own daily practice and could also be relevant for any cost analysis that might be done in the future.
The time limit of 1 hour after headache onset chosen for the definition of early triptan intake in the TEMPO study is consistent with recent observations that triptan administration within the first hour may prevent central sensitization (allodynia), whereas later triptan administration cannot block established allodynia and therefore may be less effective in inducing a pain-free state (5). The interval of 1 hour after headache onset may be considered rather long compared with other studies that evaluated triptan use at the first perception of pain or as early as possible (8,9). For example, Burstein et al. (2004) have observed that treating a migraine attack 1 hour after pain onset was frequently ineffective in inducing a pain-free state if allodynia had already developed, and these authors advocated triptan intake no later than 20 minutes after headache onset (5). On the other hand, the time limit of up to 1 hour for early dosing in the TEMPO study minimized the risk of patients treating non-migraine headaches. Furthermore, our definition of early intake allowed the assessment of treatment efficacy in an elevated proportion (80%) of attacks of moderate or severe intensity. This is in contrast to most other studies, which have generally evaluated the efficacy of early dosing on attacks of mild intensity (18).
Logistic regression analysis showed that the presence of any sign of allodynia at the time of triptan intake was strongly associated with a lower probability of achieving a pain-free state at 2 hours (odds ratio 0.53; p < 0.0001), being 1.9 times higher for patients without allodynia. These data are in good agreement with previous suggestions that early triptan administration may prevent the development of migraine-associated allodynia and therefore improve triptan efficacy on headaches (4,5). However, they are in contrast with the results of two other studies (the ‘Act when mild’ and TAME studies) that found no relationship between the efficacy of almotriptan (14) or rizatriptan (20) on pain and the existence of symptoms of allodynia at the time of dosing.
Among the patients in the TEMPO study who continued to practice late treatment despite the physician’s instruction, some experienced symptoms of allodynia and others did not. Logistic regression analysis showed that there was no association between the presence of allodynia at the time of triptan intake, on the one hand, and early or late treatment behaviour, on the other hand. This rules out the possibility that the patients who carried on with late treatment in the second phase of the study were those who never develop allodynia and might thus be rendered pain-free by a triptan taken any time after headache onset.
The increase in pain-free rate at 2 hours after triptan intake across the three treated attacks in patients switching from late to early intake suggests an increase in consistency of treatment efficacy, although the observed difference was not statistically significant because of the relatively small number of patients. Headache relief at 2 hours across the three treated attacks was also consistently more frequent after early triptan intake in both the between-patient comparison (patients with early or late dosing behaviour in the first phase of the study) and the within-patient comparison (patients who switched from late to early treatment).
The beneficial influence of early intake was not as pronounced on migraine-associated symptoms as it was on pain: compared with late treatment, only vomiting and photophobia showed significant improvement 1 hour after early treatment, and between-group differences were no longer significant at 2 hours after treatment. This suggests that migraine headache and migraine-associated non-painful symptoms may be governed by independent mechanisms and that migraine-associated symptoms may develop independently from allodynia.
Importantly, the switch from late to early treatment following the physician’s instruction did not result in an increase in redosing with the triptan owing to more frequent recurrences of migraine headaches or a higher frequency of migraine attacks. Early dosing thus does not appear liable to increase the risk of drug-induced chronification of headaches. Direct comparisons between the two dosing regimens of the rates of recurrence of migraine headache cannot be performed effectively, however, because they did not yield similar primary efficacy rates (19,21).
After advice by a neurologist, nearly two thirds of patients spontaneously practicing late treatment accepted to change and modified their usual dosing behaviour in favour of early intake, resulting in significant improvement in triptan efficacy. This observation stresses the importance of therapeutic patient education for adequate acute treatment of migraine with triptans and optimization of their efficacy. The reasons for which some patients spontaneously delayed triptan intake were chiefly the belief that the attacks would resolve spontaneously and the tendency to reserve triptan use for severe attacks, although more than 80% of the attacks treated in the study were of moderate or severe intensity. Only few patients voiced concerns about AEs to explain late triptan intake. During the whole study, AEs were uncommon, mostly of mild or moderate intensity with spontaneous resolution, and the incidence of AEs decreased drastically with increasing number of consecutive attacks or number of tablets used in a single attack. The advice delivered to migraine patients by the physicians should therefore focus on overcoming obstacles to early triptan use, as the neurologists did in the TEMPO study.
The study was conducted in a clinical practice setting. Because the study protocol was intricate and required the collection of numerous data from six consecutive migraine attacks, the relatively high attrition rate due to missing data or protocol deviations is not surprising. On the other hand, the primary efficacy variable was based on a within-patient comparison, which increases the sensitivity of the statistics and compensates for the relatively limited number of patients. In patients who continued to practice early treatment according to physician instruction, pain-free and headache relief rates at 2 hours in at least two attacks did not differ substantially between the two phases of the study. Similarly, all efficacy outcomes were globally worse throughout the study for the patients who persisted in practicing late treatment despite the physician’s instruction. This implies that the improvement observed in the patients who switched from late to early treatment after physician instruction was not solely due to medical attention.
The study has several limitations, including the relatively high dropout rate mentioned above. This is particularly problematic when comparing the outcome between the two phases of the study. In patients practicing late dosing in phase I and advised to switch, no data were available on headache outcome in 20% of the patients, all but one of whom were in the subgroup of patients not following their physician’s advice. Selective attrition of patients with poor headache outcomes cannot thus be excluded. The number of patients available for analysis at the end of phase II was relatively low (98 patients overall) and for this reason, the findings should be interpreted with caution until replicated in a larger sample. Similarly, the lack of adjustment for multiple comparisons of the different efficacy outcomes should also argue for caution in interpretation. The choice of the 1 hour cut-off for dichotomizing early and late treatment was an arbitrary one. Given the naturalistic nature of the study, it was important to simplify data collection and we considered that the quality of data would probably be better if the patients only had to tick a box (within the hour) in their headache diary than if they had to specify the time elapsed. Analysis of time since onset as a continuous variable may have yielded a more precise idea of the optimal time window for headache treatment.
In conclusion, the TEMPO study conducted prospectively in a daily clinical practice setting establishes that there are decided advantages in taking triptans early (within 1 hour of headache onset) in the course of migraine attacks. It highlights the importance of patient instruction in early treatment, whatever the triptan used, for migraine patients at low risk of medication overuse, that is, those patients with no more than six attacks a month and able to differentiate a migraine attack from tension-type headache. It also points to the need to educate migraine patients so that they give up inadequate practices or unjustified prejudices about triptan use.
Footnotes
Acknowledgements
This study was funded by Almirall SAS, France.
Disclosure
ML-M received consulting honoraria from Almirall, AstraZeneca, BMS, GlaxoSmithKline, Grünenthal, Janssen-Cilag, Lilly, Medtronic, Menarini, Pfizer, Sant-Jude, Sanofi-Aventis and Schwarz-Pharma. GM received consulting honoraria from Grünenthal, Pfizer, Lilly, Boehringer-Ingelheim, Menarini, Sanofi-Pasteur-MSD, Janssen-Cilag, Wyeth and BMS-Upsa. BA was an employee of Almirall SAS at the time of the study.
