Abstract
To date, no studies have investigated the predictive validity of variables from the initial examination to identify patients with tension-type headache pain who are likely to benefit from muscle trigger point (TrP) therapy. The purpose of this study was to develop a preliminary clinical prediction rule (CPR) to identify chronic tension-type headache (CTTH) patients who are likely to experience a successful response from TrP therapy. Consecutive patients with CTTH underwent a standardized examination and then received six sessions of TrP therapy over 3 weeks (two sessions per week). They were classified as having experienced a successful outcome at short-term (1 week after discharge) and 1-month follow-up based on a 50% reduction on at least one headache parameter (intensity, frequency or duration) and self-report perceived recovery. Potential predictor variables were entered into a stepwise logistic regression model to determine the most accurate set of variables for identifying treatment success. Data from 35 patients were included, of which 19 (55%) experienced a successful outcome. A CPR with four variables for short-term (headache duration < 8.5 h/ day, headache frequency < 5.5 days/week, bodily pain < 47 and vitality < 47.5) and a CPR with two variables for 1-month (headache frequency < 5.5 days/week and bodily pain < 47) follow-up were identified. At short-term follow-up, if three of four variables [positive likelihood ratio (LR) 3.4] were present, the chance of experiencing a successful outcome improved from 54% to 80%, and if all the variables (positive LR 5.9) were present, the probability of success was 87.4%. At 1-month follow-up, if one of two variables (positive LR 2.2) was present, the probability of success increased from 54% to 72%, and if both variables (positive LR 4.6) were present, the probability of success was 84.4%. The present CPR provides the potential to identify CTTH patients who are likely to experience short-term and 1-month follow-up success with a muscle TrP therapy approach. Future studies are necessary to validate the CPR.
Introduction
Primary headaches constitute a major health problem, and there has been increasing interest in the pathogenic mechanisms of these disorders within recent decades. Tension-type headache (TTH) is the most common type of headache (1), with the prevalence increasing in recent years (2). Further, a recent study has found that the disability attributable to TTH is larger worldwide than that due to migraine (3).
Several therapeutic approaches have been proposed for the treatment of headaches (cognitive therapy, exercise, biofeedback, manual/physical therapy, relaxation therapy), with manual therapy being one of the most commonly utilized treatments requested by these patients (4). Although there is evidence that pharmacological (5) or behavioural approach strategies (6) can be effective for headaches, the evidence for the effectiveness of physical/manual therapy remains to be elucidated.
Systematic reviews that have analysed the effects of physical/manual therapies on TTH have concluded that there is insufficient evidence either to support or to refute the effectiveness of physical therapy/exercise (7) or spinal manipulative therapy (8, 9) in this headache disorder. For example, previous studies analysing the effects of spinal manipulation in TTH patients have shown conflicting results (10, 11). As there is no substantial scientific evidence that TTH has its pathogenesis in cervical zygapophyseal joints, this could be a reason why manipulation has not shown satisfactory results in this headache. Further, these equivocal results may be attributed to the fact that previous studies investigating manual therapy approaches in TTH did not identify subgroups of patients who are most likely to benefit from these interventions.
Our group has recently proposed a pain model in which TTH can be explained by referred pain elicited from trigger points (TrPs) in head, neck or shoulder muscles (12). However, no previous studies have investigated the application of muscle TrP therapy on this headache. Since previous studies have shown conflicting results, before conducting a randomized controlled trial it would be interesting to identify subgroups of patients with TTH who are most likely to benefit from muscle TrP therapy.
A clinical prediction rule (CPR) consists of a combination of several variables, obtained from patients' history, self-report outcomes, and clinical examination assisting with the subgrouping of patients into treatment-based classifications. Several CPRs have been found to be useful in classifying patients with low back pain (13) or neck pain (14, 15) who are likely to benefit from a particular intervention, e.g. spinal manipulation. A previous study has tried to identify predictors which are most likely to identify patients with cervicogenic headache who achieved a reduction of at least 50% in headache frequency, after a physical therapy programme; however, authors did not find a consistent pattern of predictor variables for this headache disorder (16).
To date, no studies have specifically investigated the predictive validity of several variables from the initial examination to identify patients with TTH who are likely to benefit from muscle TrP therapy. Therefore, the purpose of the current study was to develop a preliminary CPR to identify the predictors for patients presenting with chronic tension-type headache (CTTH) who would respond to muscle TrP therapy.
Materials and methods
Subjects
We conducted a prospective cohort study of consecutive patients presenting with CTTH derived from the Neurology Department of the Fundación Hospital Alcorcón to an academic clinic located at the Department of Physical Therapy, Occupational Therapy, Rehabilitation and Physical Medicine, Universidad Rey Juan Carlos, Alcorcón, Spain. CTTH was diagnosed according to the criteria established by the International Headache Society (IHS) by an experienced neurologist (17). To be included, they had to describe pain characteristics of TTH: bilateral location, pressing and tightening pain, mild or moderate intensity (≤ 6 on a 10-cm visual analogue scale), and no aggravation of pain during physical activity. No patient reported photophobia, phonophobia, vomiting or nausea during attacks. Further, they had to have headaches for at least 15 days/month for at least the last 3 months. Medication-overuse headache as defined by the IHS was ruled out in all cases. Finally, there was no apparent evidence of secondary headaches. Exclusion criteria were: (i) pain feature(s) of migraine or other headache; (ii) identification of any medical ‘red flags’; (iii) history of a whiplash injury; (iv) history of cervical or cranial surgery; and (v) evidence of any central nervous system involvement.
All subjects had previously taken several preventive (i.e. amitriptyline) drugs, but none maintained their prophylactic therapy at the time of the study. The period during which patients were free from prophylactic drugs ranged from 6 to 10 months. Further, all patients were naive with respect to any previous physiotherapeutic approach. All subjects read and signed a consent form approved by the Institutional Review Board at Fundación Hospital Alcorcón.
Self-reported measures
Subjects provided demographic information following a standardized history. Key elements of the headache history, including family history, headache features, temporal profile and intake medication, were ascertained. Self-report measures included a head–neck diagram to assess the location of head–neck symptoms, a headache diary, and the Medical Outcomes Study 36-Item Short Form (SF-36). A headache diary was used to substantiate the diagnosis (18), and the diary was maintained during the entire study (12 weeks). In this diary, subjects had to register the number of days with a headache (days/week), the headache duration (in h/day) and the headache intensity on an 11-cm numerical pain rate scale (NPRS; range 0 = no pain, 10 = maximum pain) (19). The SF-36 is a self-administered, 36-item questionnaire that measures health-related functions in eight domains: physical functioning; role limitations due to physical problems; body pain; vitality; general health perception; social functioning; role limitations due to emotional problems; and mental health (20). After summing the Likert-scaled items, each domain was standardized ranging from 0 (lowest level of functioning) to 100 (highest level) according to established international standard guidelines (21). This questionnaire has exhibited ability to discriminate between symptomatic and healthy subjects (22).
Examination procedures
At the beginning of the study, an independent assessor, who was not involved in the treatment of the patients, underwent a 1-h training session in which she practised the examination procedures to ensure that they were performed in a standardized fashion in all the included subjects. The following physical examinations were performed by this assessor on all patients before their inclusion in the TrP therapy programme: cervical range of motion, forward head posture, pressure pain threshold, total tenderness score, and manual examination of muscle TrPs.
Neck mobility was assessed with a cervical goniometer (Performance Attainment Associates, Lindstrom, MN, USA), which has been shown to have good intratester [intraclass correlation (ICC) 0.7–0.9] and intertester (ICC 0.8–0.87) reliability (23). The extent of motion for each neck movement in a single movement direction was measured in a standard fashion: forward (flexion), backward (extension), right lateral flexion, left lateral flexion, right rotation, and left rotation. Three measurements were performed for each direction of movement and the mean of the three trials was employed in the analysis. A photograph of the lateral view of each participant was taken in both sitting and standing positions to assess objectively the cranio-vertebral angle, i.e. the angle between the horizontal line passing through C7 level and a line extending from the tragus of the ear to C7 vertebra (24). A smaller cranio-vertebral angle indicated a greater forward head posture. The protocol for the assessment of both cranio-vertebral angle and neck mobility has been described in detail elsewhere (25).
Pressure pain thresholds (amount of pressure where the perception of pressure first changes to pain) (26) were assessed bilaterally in the anterior part of both temporal muscles (cephalic point) (27), and in both upper trapezius muscles (neck point) at a point 2 cm lateral to the halfway point between the spinous process of the seventh cervical vertebra (C7) and the lateral edge of the acromion. A pressure algometer consisting of a 1-cm rubber disk (cm2) connected to a sensor recording pressure in kg was used. Pressure measurement unit is then expressed in kg/cm2. The assessor applied continuous pressure with the algometer at approximately a rate of 1 kg/cm2/s. Patients pressed the button when their pain threshold was reached, and the reading display was stopped. Three consecutive pain threshold measurements at intervals of 30 s were obtained and the mean of the three trials was considered in the analysis. The reliability of this procedure ranges from 0.6 to 0.97 (intra-examiner) and from 0.4 to 0.98 (interexaminer) (28, 29).
The total tenderness score (30), which has been found to be a reliable method of manual assessment (31), was used to investigate tissue tenderness. Briefly, eight pairs of muscles and tendon insertions (masseter, temporalis, frontalis, trapezius, suboccipital and sternocleidomastoid muscles, as well as coronoid and mastoid processes) were palpated. Palpation was performed with small rotational movements of the assessors' second and third fingers during 4–5 s. Tenderness was scored on a four-point (0–3) scale at each location (local tenderness score) and values from left and right sides were summed to a total test score (maximum possible score = 48 points).
Finally, upper trapezius (32), sternocleidomastoid, temporalis (33) and superior oblique (34) muscles were evaluated bilaterally for the presence of active TrPs following previous guidelines (35). Only active TrPs, i.e. those which referred pain elicited by its compression reproduced the headache symptoms (36), were registered.
Treatment
All patients received six sessions of TrP therapy over a 3-week period (two sessions/week) by a manual physical therapist experienced in the treatment of headaches. As we do not know which particular programme is most effective for this headache, we applied a TrP therapy programme focused on the management of head, neck, shoulder muscles (temporalis, suboccipital, upper trapezius, superior oblique, splenius capitis, sternocleidomastoid, semispinalis capitis). Different TrP approaches, e.g. pressure release, muscle energy or soft tissue techniques, were applied to these muscles in order to inactivate active muscle TrPs (Fig. 1). Furthermore, patients were instructed to perform an exercise programme based on deep flexor and extensor muscle low-load and progressive contractions (37, 38). Patients performed slow and controlled cranio-cervical flexion or extension actions. When patients showed good control of the exercise, they were trained to increase the number of contractions progressively for either flexion or extension movement. They were instructed to conduct two series of 10 repetitions for each direction at home on a daily basis.
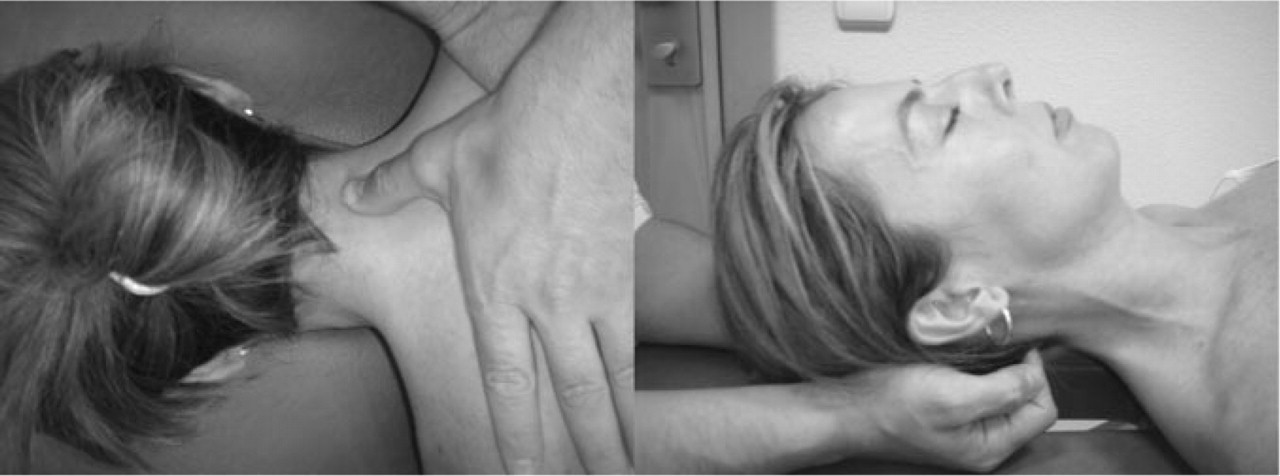
Some manual therapy approaches directed to muscle trigger points in the cervical musculature.
We used a prospective A1-B-A2 design study. Phase A1 (pre-intervention phase) consisted of a 4-week baseline period, when no TrP therapy treatment was applied. Phase B (intervention phase) was the 3-week treatment period through which patients were treated with the above-mentioned programme. Phase A2 (postintervention phase) was a 5-week postintervention period. All patients had three measure appointments: (i) pretreatment (first visit, week 1); (ii) post-treatment (1 week after the end of the intervention, week 8); and (iii) follow-up (1 month after the end of the intervention, week 12). This study design has been previously used (39).
Determination of success
The Global Rating of Change (GROC), satisfaction level and the headache diary were combined as reference criterion for establishing a successful outcome. At the first visit, self-reported and physical outcomes were obtained, whereas at both post-treatment and 1-month follow-up appointments, GROC and satisfaction level were both assessed. The headache diary was maintained during the 12-week period of the study.
At the end of the study the assessor was given the headache diary, and the following variables were calculated: (i) headache intensity, calculated from the mean of the NPRS of the days with headache; (ii) headache frequency, calculated by dividing the number of days with headache by the number of analysed weeks (days/week); and (iii) headache duration, calculated by dividing the sum of the total hours of headache by the number of days with headache (h/day). Pretreatment data of headache intensity, frequency and duration outcomes were obtained from baseline period (first 4 weeks); post-treatment data were derived from the following 4 weeks, and follow-up data were calculated from the last 4 weeks of the evaluation protocol.
Both perceived improvement and satisfaction level were assessed with a 15-point Likert scale. The perceived improvement was assessed with a GROC consisting of a 15-point scale ranging from −7 (a very great deal worse) to +7 (a very great deal better) (40). Descriptors of worsening or improving were assigned with values ranging from −1 to −7 and +1 to +7, respectively (41). A 15-point scale ranging from −7 (a very great unsatisfied) to +7 (a very great satisfied) was also used to evaluate the satisfaction level. It has been reported that scores of +4 and +5 are indicative of moderate changes in patient status and scores of +6 and +7 indicate large changes in patient status (41).
For the headache diary, responsiveness was judged as a 50% reduction in at least one of the headache features (headache intensity, frequency or duration). For the GROC, subjects who rated their perceived recovery as ‘a very great deal better’, ‘a great deal better’, or ‘quite a bit better’ (i.e. a score of ≥ +5) were categorized as having a successful outcome. Finally, for the satisfaction level the same criteria as for GROC was used (i.e. a score of ≥ +5). In the present study we consider as responders those CTTH patients who reported a ≥ +50% reduction in at least one of the headache parameters (intensity, duration or frequency) assessed by the headache diary, in addition to a GROC or satisfaction level successful result (a score of ≥ +5).
Data analysis
Patients were dichotomized as success or non-success based on the treatment response at both the short-term and 1-month follow-up as indicated previously (50% reduction of one headache clinical feature, and ≥ +5 on the GROC, or ≥ +5 in the level of satisfaction). Individual variables from self-report measures, the history and physical examination were all tested for univariate relationship with the reference criteria using independent samples t-tests for continuous variables and χ2 tests for categorical variables for both the short-term and 1-month follow-up. Variables with a significance level of P < 0.1 were retained as potential prediction variables (42). For continuous variables with a significant univariate relationship, sensitivity and specificity values were calculated for all possible cut-off points, and then plotted as a receiver–operator characteristic (ROC) curve (43). The point on the curve nearest the upper left-hand corner represented the value with the best diagnostic accuracy, and this point was selected as the cut-off defining a positive test (43). Sensitivity, specificity and positive and negative likelihood ratios (LR) were calculated for potential predictor variables.
Additionally, all potential predictor variables were entered into a step-wise logistic regression model to determine the most accurate set of variables for prediction of treatment success. A significance level of 0.1 was required for removal from the equation to minimize the likelihood of excluding potentially helpful variables (42). Variables retained in the regression model were obtained as the most optimal cluster for classifying patients with CTTH likely to benefit from manual therapy.
Results
Between February 2005 and September 2006, 35 subjects were recruited for the study. The total number of subjects screened, reasons for ineligibility and drop out can be seen in Fig. 2. From the included patients, there was no loss to follow-up during the entire study. Subject demographics and initial baseline variables from the headache history and self-report measures for the entire sample as well as for both the success and non-success groups at both the short-term and 1-month follow-up periods can be found in Table 1. Baseline clinical examination variables for the entire sample and both groups are found in Table 2. Nineteen patients (55%) were categorized as having achieved successful treatment and 16 (45%) as non-successes at both the short-term and 1-month follow-up periods. The number of days between the initial examination and short-term assessment was 55.3 (S.D. 18.8) and 55.4 (S.D. 1.9) (P = 0.9) for the success and non-success groups at the short-term follow-up, respectively. The mean number of days between the initial examination and 1-month follow-up assessment was 84.3 (S.D. 2.5) and 83.7 (S.D. 2.4) (P = 0.5) for the success and non-success groups at the 1-month follow-up, respectively.
Demographics, baseline self-report variables and baseline characteristics of subjects

χ2 tests.
Independent samples t-tests.
Physical examination variables from the baseline clinical examination

Analysed with independent t-tests.
CVA, Cranio-vertebral angle; PPT, pain pressure threshold; TrPs, trigger points.

Flow diagram of subject recruitment and retention throughout the course of the study. CDH, Chronic daily headache. ∗Five CTTH patients with successful or non-successful outcomes were not the same for immediate and 1-month follow-up periods.
The 12 potential predictor variables for predicting success at short-term (Table 3) that exhibited a significance level < 0.1 were entered into the logistic regression. The cut-off values determined by the ROC curves and the accuracy statistics for all 12 variables [and 95% confidence interval (CI)] are shown in Table 3. The positive LR ranged from 1.3 to 2.5, with the strongest predictor being the bodily pain domain of the SF-36 questionnaire less than 47. The following four variables were retained in the final regression model for predicting short-term response: headache duration (h/day) < 8.5 h, headache frequency (days/week) < 5.5, bodily pain < 47 and vitality < 47.5 (P < 0.001, Nagelkerke's R 2 = 0.82). These four variables were used to form the most parsimonious combination of predictors for identifying patients with CTTH likely to benefit at short-term period from TrP therapy. Accuracy statistics were calculated for the numbers of variables present (Table 4). The pretest probability for the likelihood of success with TrP therapy for this study was 54%. If the patient exhibited three of the four variables the positive LR was 3.4 (95% CI 1.4, 8.0) and the post-test probability of success increased to 80%. If the patient was positive on four of the four variables, the positive LR was 5.9 (95% CI 0.80, 42.9) and the post-test probability of success was 87.4% (Fig. 3).
Accuracy statistics with 95% confidence intervals (CI) for individual predictor variables for short-term response

PPT, Pain pressure threshold; the probability of success is calculated using the positive likelihood ratios and assumes a pretest probability of 54%.
Combination of predictor variables identified in the logistic regression analysis and associated accuracy statistics with 95% confidence intervals for the identification of short-term success

• Headache duration (h/day) (< 8.5)
• Headache frequency (< 5.5)
• Body pain (< 47)
• Vitality (< 47.5)
The probability of success is calculated using the positive likelihood ratios and assumes a pretest probability of 54%.
Accuracy statistics with 95% confidence intervals for individual variables for predicting success.

Post-test probability (87%) of immediate success if four predictor variables were present.
The 12 potential predictor variables for predicting success at 1-month follow-up that exhibited a significance level < 0.10 were entered into the logistic regression (Table 5). The cut-off values determined by the ROC curves and accuracy statistics for all 12 variables (and 95% CI) can be found in Table 5. The positive LRs ranged from 1.1 to 3.4, with the strongest predictor being cervical side-bending to the right > 31°. The following two variables were retained in the final regression model for predicting 1-month follow-up response: headache frequency (days/week) < 5.5, bodily pain < 47 (P < 0.001, Nagelkerke's R 2 = 0.52). These two variables were used to form the most parsimonious combination of predictors for identifying patients with CTTH likely to benefit from TrP therapy at 1-month follow-up. Accuracy statistics were calculated for the numbers of variables present (Table 6). If the patient exhibited one of the two variables the positive LR was 2.2 (95% CI 1.2, 3.8) and the post-test probability of success increased to 72%. If the patient was positive on two of the two variables the positive LR was 4.6 (95% CI 1.2, 17.9) and the post-test probability of success was 84.4% (Fig. 4)
Accuracy statistics with 95% confidence intervals (CI) for individual predictor variables for 1-month follow-up response
Combination of predictor variables identified in the logistic regression analysis and associated accuracy statistics with 95% confidence intervals for the identification of 1-month follow-up success

• Headache frequency (< 5.5)
• Bodily pain (< 47)
The probability of success is calculated using the positive likelihood ratios and assumes a pretest probability of 54%.
Accuracy statistics with 95% confidence intervals for individual variables for predicting success.

Post-test probability (84%) of 1-month success if two predictor variables were present.
Discussion
The present study has attempted to identify predictor variables for identifying patients with CTTH who subjectively responded to a muscle TrP therapy programme. In order to classify the treatment responders and non-responders, the combination of two criteria considered as clinically important was used. Several cut-offs for clinical changes on pain measures have been suggested, being a reduction of 50% the most widely used as prognostic factor in other pain conditions, e.g. neck pain (44). This criterion resulted in 54% of patients achieving a successful outcome.
The preliminary CPR identified in the present study can help clinicians to identify which CTTH patients may be most likely to benefit from muscle TrP therapy, although results should be taken with caution since the CPR is in a developmental stage. This preliminary clinical decision-making tool can assist with guiding patient interventions and result in improvement in their overall condition. Four variables, including headache duration (< 8.5 h/day), headache frequency (< 5.5), bodily pain (< 47) and vitality (< 47.5), were obtained. Interestingly, all of these variables appear to exhibit face validity, as it might be expected that CTTH patients with less duration and frequency of headaches as well as lower bodily pain may be more likely to experience a successful outcome, since higher headache frequency is associated with greater central sensitization (45). However, if patients exhibited lower vitality then they were also likely to be more responsive to the intervention, which may seem counterintuitive. Nevertheless, no previous studies have been conducted to confirm these hypotheses. Although no theoretical link between lower vitality scores and improved outcomes can be identified, if all four of these variables were present the probability of success was 84%.
An unexpected finding was that both number of active TrPs and tenderness were not identified as predictor variables in the CPR. We have previously reported that CTTH patients with active TrPs in the sternocleidomastoid, temporalis (33) or upper trapezius (32) muscle showed longer headache duration or frequency than those patients with latent TrPs. Nevertheless, no previous studies have investigated if the number of active TrPs is related to headache parameters. Future studies are required to investigate these relationships. Further to the fact that muscle tenderness also did not hold in the CPR, we agree with previous studies that have reported that muscle tenderness was not related to headache intensity, duration or frequency as reported by the patients themselves (46, 47)
Interestingly, Jull and Stanton were unable to identify any consistent predictors among a group of patients with cervicogenic headache and outcomes following an episode of physical therapy care (16). However, in the present study we did identify variables that were consistent between the immediate and 1-month follow-up periods. For example, both a lower headache frequency and lower bodily pain predicted immediate response for a successful outcome as well as at the 1-month follow-up period. Therefore, if a patient exhibited either the lower headache frequency or lower bodily pain than the chance of success was 73%. However, if both variables were present the patient had an 84% chance of achieving success. Perhaps as suggested by Jull and Stanton (16), their potential predictors might have not been sensitive enough, whereas the variables we used might be more responsive to identify those likely to achieve a successful outcome.
Perhaps using this preliminary CPR to classify CTTH patients into subgroups prior to carrying out a randomized clinical trial might lead to a strong effect size. However, this would require future controlled studies demonstrating consistent treatment findings since they will include more homogeneous samples. No studies have previously applied the TrP therapy programme, which was based on the clinical reasoning recently proposed by our group (12), used in this CPR. Future randomized controlled trials are now required in order to investigate the effectiveness of this therapy approach aimed at inactivating active muscle TrPs in CTTH patients.
There are a number of limitations to the study, including the small sample size. It is possible that the small sample size and the number of variables entered into the logistic regression may have resulted in over-fitting of the model, which can have led to spurious findings. However, in the development stage of a CPR it is important and necessary to include all the potential predictor variables, and any variable that may have fallen out as a predictor needs to be re-examined in future validation studies (48). Additionally, based on the prospective cohort design, it is also possible that the identified variables were simply predictors of the passage of time rather than contributing to the TrP therapy. Given the absence of previous investigation regarding the effectiveness of TrP therapy in CTTH patients, and the costs associated with high-quality clinical trials, a randomized trial was not the most sensible design at this time. The fact that it can be impossible to conclude definitively that the rule predicted a specific response to TrP therapy or the passage of time was known a priori is a question that would have to be answered in subsequent follow-on studies. Further, we should recognize that we used a generic questionnaire (SF-36) for assessing quality of life in our headache patients. Although it is one of the questionnaires most widely used in research, it would be recommended in future studies to include questionnaires evaluating perceived disability (49), anxiety (50) or depression (51) in headache populations.
Finally, future studies with larger sample sizes and a greater number of potential predictor variables (e.g. those previously discussed) are necessary to establish definitively a CPR for identifying CTTH patients who are likely to experience a successful outcome with a muscle TrP therapy programme. Additionally, this study should include a long-term follow-up and a comparison group to investigate further the predictive value of the variables in this preliminary CPR. Finally, if a rule is validated, an impact analysis of implementation and its effect on clinical practice patterns, outcomes, and costs of care should be investigated.
Conclusions
The present preliminary CPR provides the ability to identify CTTH patients who are likely to experience an immediate and 1-month follow-up success with a muscle TrP therapy programme. Future studies with larger sample sizes and a greater number of potential predictor variables are necessary.
