Abstract
This proof-of-concept study evaluated the efficacy of prednisone for the treatment of withdrawal symptoms in patients with medication overuse headache (MOH) in a randomized, placebo-controlled, double-blind design. Twenty patients were randomized and underwent in-patient withdrawal therapy. The total number of hours with severe or moderate headache within the first 72 and 120 h was significantly lower in the prednisone group. The results show that prednisone might be effective in the treatment of medication withdrawal headache.
Introduction
Chronic headache is a frequent disorder with a prevalence of 2–5% in the general population. Within the last 5 years epidemiological studies have shown that up to a third of these cases represent patients with medication overuse headache (MOH) or probable MOH (1–4) according to the classification of the International Headache Society, 2nd edn (ICHD-II, Code 8.2). The condition is defined as headache on > 15 days/month due to the frequent intake of antiheadache medication on 10–15 days/month (5). There is general agreement that all drugs used for the treatment of headache can cause MOH in patients with a primary headache disorder (6). Treatment of choice is the complete discontinuation and subsequent withdrawal from the overused headache medication. Discontinuation of overused medication usually leads to severe exacerbation of a headache called ‘withdrawal headache’ (7). In addition, most patients experience typical withdrawal symptoms such as nausea, vomiting, arterial hypotension, tachycardia, sleep disturbances, restlessness, anxiety and nervousness. Depending on the overused drugs, withdrawal symptoms may vary with respect to their intensity and may last up to 14 days, in special cases even longer (8). Due to a lack of treatment recommendations, therapeutical strategies to alleviate withdrawal symptoms vary among centres and countries. Among those agents suggested for withdrawal therapy are analgesics, tranquillizers, neuroleptics, antidepressant drugs (amitriptyline), antiepileptic drugs (valproate), intravenous dihydroergotamine, subcutaneous sumatriptan, oxygen and electric stimulation. None of these treatments has been investigated in a proper randomized, placebo-controlled trial. Few prospective studies have addressed the issue of the treatment of withdrawal symptoms during withdrawal therapy in patients suffering from MOH. An open non-placebo controlled study performing withdrawal on an out-patient basis has suggested that prednisone may be effective in reducing withdrawal symptoms (9). The aim of this study was therefore to evaluate the potential clinical efficacy of prednisone in a randomized, placebo-controlled, double-blind study for the treatment of withdrawal symptoms following withdrawal therapy in patients with MOH.
Methods
The study was approved by the local ethics committee (no. 03–2138) of the University of Essen. Written consent was obtained from all study participants prior to enrolment in the study.
Study population
Twenty patients were enrolled. Inclusion criteria were: men or women aged 18–65 years suffering from MOH according to the diagnostic criteria of the ICHD-II (5) caused by 5-HT1
Study design and protocol
This was a double-blind, placebo-controlled, randomized, single-centre pilot study. Patients with MOH underwent withdrawal therapy on an in-patient basis in the Department of Neurology, University Hospital of Essen. Placebo or prednisone 100 mg were given once daily for the first 5 days of withdrawal. The medication was blinded, randomized by standardized computer-based randomization software and delivered by the pharmacy of the University Hospital of Essen.
Severity of headache was documented every hour in a diary using a four-point scale (0 = none, 1 = mild, 2 = moderate, 3 = severe headache). Intravenous 1000 mg lysine acetylsalicylic acid (LAS) was given as rescue medication in case of severe withdrawal headaches. In Germany, LAS is the substance of choice for the treatment of severe migraine attacks and withdrawal headaches if there are contraindications for triptans or other oral antiheadache medication.
Outcome criteria
The primary end-point was the number of hours with severe or moderate headache within the first 72 h of withdrawal. Secondary end-points were the number of hours with severe or moderate headache within the first 120 h of withdrawal and the amount of rescue medication used in each group.
Statistical analysis
A Wilcoxon rank sum test was use to compare interval scaled (age, number of headache days per month and number of hours with severe or moderate headache and number of rescue medications during 120 h of withdrawal), and χ2 test for ordinal scaled data (gender) variables. The non-parametric test was chosen because of the relatively small number of patients. All statistics were calculated with the SPSS program (9.0.1; SPSS Inc., Chicago, IL, USA). The level of significance was set at 0.05.
Results
Patient population
Twenty patients were enrolled into the study (10 in each group). Complete sets of data were available for 18 patients (each group n = 9). One patient in the prednisone group was not suffering from any headaches in the days before starting treatment, the treatment or follow-up period. One patient in the placebo group did not record his headache severity exactly for every hour of the day. Because of missing data these two subjects could not be evaluated. Individual data of each patient are shown in Table 1.
Data of each individual patient
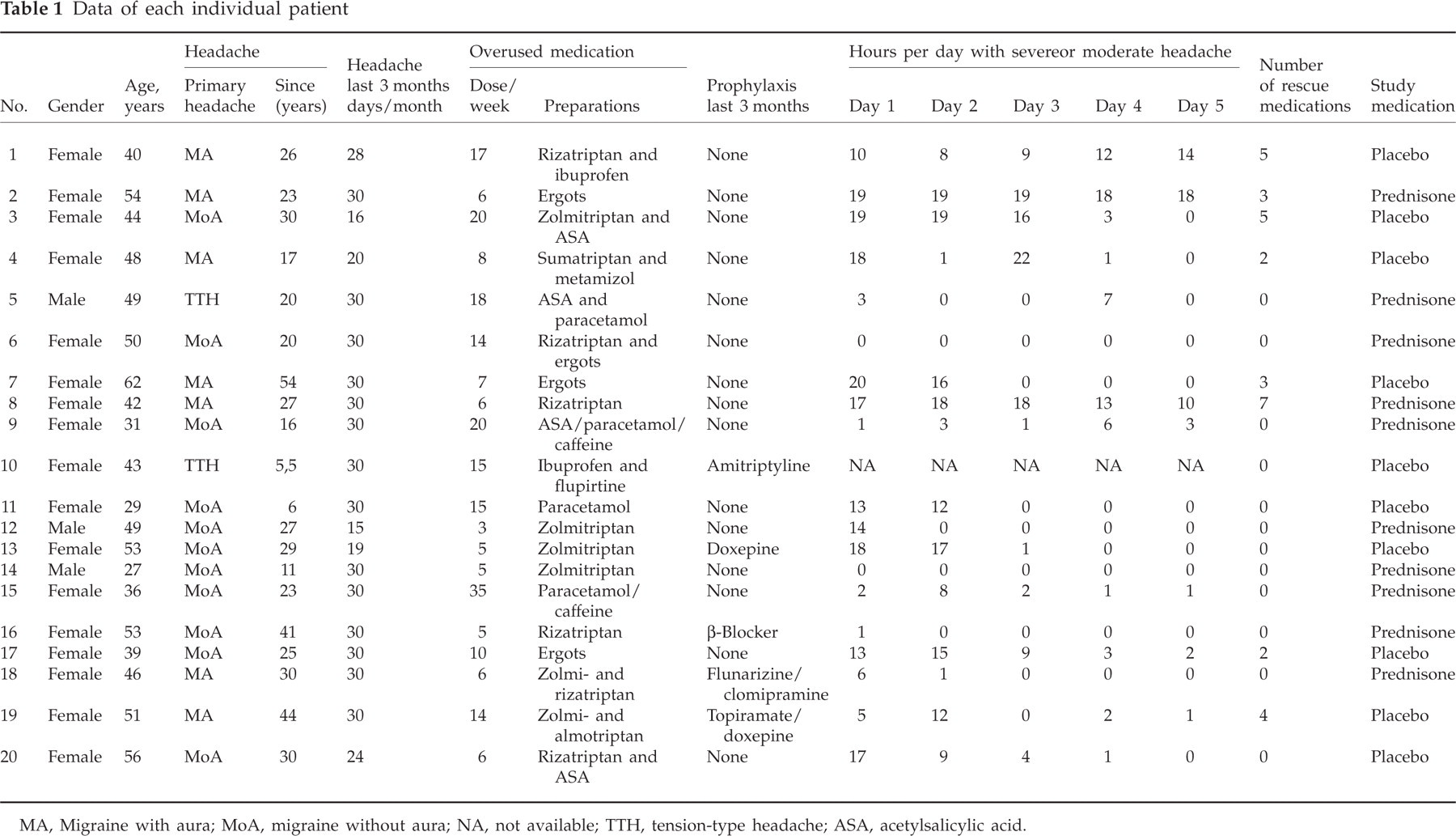
MA, Migraine with aura; MoA, migraine without aura; NA, not available; TTH, tension-type headache; ASA, acetylsalicylic acid.
No significant demographic and clinical differences were found between the two treatment groups (Table 2).
Clinical and demographic data of the patient population

Outcome criteria
The total number of hours with severe or moderate headache within the first 72 (primary end-point) and 120 h (Fig. 1) was significantly lower in the prednisone group than in the placebo group (18.1 vs. 36.7 h, P = 0.031, and 27.22 vs. 42.67 h, P = 0.05). Patients in the prednisone group requested rescue medication less frequently than those of placebo group, which however, did not reach significance (11 dosages vs. 21 dosages, P = 0.3). Adverse events were not reported.

Hours with severe or moderate headache within the first 72 and 120 h after withdrawal of headache medication.
Discussion
The main finding of this study was that 100 mg prednisone given daily for 5 days decreases the duration of withdrawal headache significantly compared with placebo-treated patients and is well tolerated. Significant adverse events were not reported by the participants. The duration and severity of withdrawal headache in the placebo group were in line with the results of former studies investigating withdrawal symptoms in patients with MOH undergoing withdrawal therapy. Since this trial was planned as a proof-of-concept study, only 20 patients were enrolled. Although the results are clearly in favour of prednisone therapy, this is a shortcoming of the study because the small number of patients does not allow subgroup analysis regarding the influence of the primary headache type, accompanying withdrawal symptoms, substance use, or gender. The issue of headache recurrence following sudden prednisone discontinuation (no tapering) was not addressed in this pilot trial and will be studied in a larger, future trial.
Our results suggest that prednisone could be effective in treatment of medication withdrawal headache and that the drug may facilitate withdrawal therapy. In contrast to our results, a recent Norwegian placebo-controlled study has shown that 60 mg prednisone given for 2 days and tapering the dose the following 4 days had no effect on withdrawal headache in patients with chronic daily headache and medication overuse (10). Typically, the intensity of withdrawal headache increases between days 2 and 4 after withdrawal of the previously used antiheadache medication and wanes again by days 6 and 8. Hence, it is tempting to speculate that a dosage of 60 mg for the first 2 days was simply not enough to achieve significant effects during withdrawal therapy (10).
Mechanisms leading to MOH are still little known. Down-regulation of serotonin-receptors (5-HT1B/D) or prostaglandin-synthesizing enzymes in anatomical structures that are involved in the transmission and modulation of nociceptive signals may lead to an impairment of antinociceptive activity and subsequently result in a permanent feeling of headache (11). The possible action of steroids in the treatment of MOH and other headaches such as cluster headache or status migrainosus still needs to be determined. The anti-inflammatory effect and the inhibition of cyclo-oxygenase, cyclo-oxygenase-II in particular, are possible mechanisms responsible for the positive effect of prednisone. As a consequence of this trial, a larger, multicentre, Phase III study has been initiated at various centres in Europe.
Footnotes
Acknowledgements
Supported by the Federal Ministry of Research and Education of Germany (BmBF) in the context of the German Headache Consortium (01EM0117).
