Abstract
The pathophysiology of cluster headache (CH) is supposed to involve the lower posterior part of the hypothalamus, the trigeminal nerve, autonomic nerves and vessels in the orbital/retro-orbital region. The exact connection of this hypothalamic-trigemino-autonomic-vascular axis is not fully understood. The presence of inflammation in the perivascular tissue of the retro-orbital region has been presented as a possible mechanism behind the pain and the sympatheticoplegia sometimes observed during headache attacks. In a previous study we found neither increased levels of erythrocyte sedimentation rate, C-reactive protein or acute-phase reactants nor clinical signs of a generalized inflammatory disorder. However, these tests may not be sensitive enough to detect a focal inflammatory process in the retro-orbital region. In the present study, we analysed serum levels of three soluble adhesion molecules; soluble intercellular adhesion molecule-1 (sICAM-1), soluble vascular cell adhesion molecule-1 (sVCAM-1) and soluble E-selectin (sE-selectin) in patients with episodic CH and in patients with biopsy-positive giant cell arteritis (GCA), a known vasculitic disorder of large and medium-sized arteries. A control group of healthy volunteers was also included. Within the CH group, sICAM-1, sVCAM-1 and sE-selectin showed an increasing trend in remission compared with the active CH period, but the difference was statistically significant for sE-selectin only. The mean sICAM-1 value was higher in patients with active GCA than in CH patients during the active cluster period. Compared with the healthy control group, the mean levels of soluble adhesion molecules in CH patients also tended to be higher, but statistically significantly so only for sVCAM-1. We hypothesize that CH is not a vasculitic disorder of the medium-sized arteries, but CH patients may have an immune response that reacts differently from that of healthy volunteers.
Introduction
Cluster headache (CH) is characterized by recurrent attacks of severe unilateral retro-orbital headache accompanied by restlessness and agitation (1), ipsilateral autonomic features, or both. The headache attacks occur both during daytime and at nights, often with a strikingly regular timing. Most CH patients suffer from the episodic variety of the disorder with intermittent periods of remission. In about 10% of the CH patients the disorder becomes chronic (2).
The pathophysiology of CH includes the participation of the hypothalamus, trigeminal nerve, autonomic nerves and vessels in the retro-orbital region (3, 4). Dilation of the ophthalmic artery has been observed accompanying the pain attacks (5–7). ‘Inflammation’ in the region of the cavernous sinus has been hypothesized to cause the pain (8–10). However, a recent study using single photon emission computed tomography (SPECT)/magnetic resonance imaging (MRI) techniques has not revealed evidence for extravasation of plasma proteins in the cavernous sinus during a CH attack (11).
Hannerz (12) has reported that patients with episodic CH during the active CH period presented laboratory signs of inflammation such as an increased erythrocyte sedimentation rate (ESR) or increased levels of orosomucoid and haptoglobin. Similar changes have also been reported in patients with recurrent Tolosa–Hunt syndrome. He therefore hypothesized a close pathophysiological relationship between these two conditions. Furthermore, he suggested that CH was actually a generalized inflammatory disorder, i.e. a systemic vasculitic disorder. In a previous study we were unable to confirm his hypothesis (13). The idea that a focal inflammatory reaction would be triggered by or even trigger the hypothalamic–trigemino–autonomic–vascular axis to cause vasodilation and the release of pain-inducing substances still remains a topic of interest.
Previous studies on patients with active systemic vasculitic disorders, including giant cell arteritis (GCA), have demonstrated elevated concentrations of circulating soluble adhesion molecules such as soluble intercellular adhesion molecule-1 (sICAM-1), soluble vascular cell adhesion molecule-1 (sVCAM-1) and soluble E-selectin (sE-selectin) (14, 15). E-selectin expression is preferentially detected in early infiltrates, whereas endothelial expression of ICAM-1 and VCAM-1 predominates at a later stage. Furthermore, E-selectin expression correlates with the presence of neutrophils, whereas in later stages mononuclear cells predominate.
Our aim was to study signs of inflammatory reactivity among patients with episodic CH, since clinically used routine laboratory tests may be too insensitive to detect signs of a focal inflammatory process. We chose to investigate the levels of soluble adhesion molecules, the first step of activation of proinflammatory cytokines (16), and compared the findings among CH patients with those among patients suffering from a vasculitic disorder, GCA, during active disease and a control group of healthy volunteers.
Materials and methods
Patients
Patients aged 18–65 years, with episodic CH according to the International Headache Society criteria (1), who consulted the Outpatient Department of Neurology at Söder Hospital, Stockholm, were asked to participate in the study. Twenty-seven patients, 21 men and six women, agreed and provided fully informed consent. The study was approved by the local Ethics Committee. The clinical data and results of clinically used laboratory tests of blood and urine samples aimed at detecting a generalized vasculitic disorder have been reported in a previous paper (13). In 21 of the patients serum levels of three soluble adhesion molecules, ICAM-1, VCAM-1 and E-selectin, were analysed. The blood samples were taken during an active CH period (between headache attacks) and repeated in remission at least 4 weeks after cessation of the headache attacks. The patients were not allowed to use corticosteroids, antibiotics, or non-steroidal anti-inflammatory drugs, but could treat their headache attacks with oxygen, codeine, paracetamol or sumatriptan during the study.
Control subjects
In 35 patients with biopsy-positive GCA blood samples were taken once in active but untreated disease. In 50 healthy control subjects also, blood samples were taken once. All GCA patients and healthy volunteers gave their informed consent.
Analyses
The blood samples were centrifuged and stored at −70°C until analysed. After thawing, the serum levels of sICAM-1, sVCAM-1 and sE-selectin were determined by commercially available enzyme-linked immunosorbent assay kits and standards (R&D Systems Ltd, Abingdon, UK). Duplicate analyses were performed and the arithmetic mean value was used for data analysis.
Statistical methods
All results are given as mean ± SD. Student's paired t-test was used in comparisons between CH during active cluster periods and CH in remission. A two-tailed P-value of <0.05 was considered to be statistically significant. A non-parametric test, Kruskal–Wallis
Results
The demographic and clinical data from the CH and GCA patients and the healthy volunteers are presented in Table 1. The mean age of the CH patients at inclusion was 44 years (range 30–60; n = 21). The GCA patients were generally older, mean age 70 years (range 52–79 years; n = 35). The mean age of the healthy control subjects was 50 years (range 46–61 years; n = 40). The mean age at CH onset was 27 years (range 17–55 years; n = 21) for all patients. Among female and male patients the corresponding mean values were 22 years (range 19–28 years; n = 3) and 28 years (range 17–55 years; n = 18), respectively. The few female patients tended to have longer CH periods and more CH attacks per day than the male patients.
Demographic and clinical data on patients with episodic cluster headache (CH), giant cell arteritis (GCA) and in healthy control subjects
The mean and SD values of sICAM-1, sVACM-1 and sE-selectin measured among CH patients during active cluster periods and in remission, GCA patients during active disease and healthy control subjects are presented in Table 2 and plotted in Fig. 1. The mean serum levels of sICAM-1, sVCAM-1 and sE-selectin during active CH periods were 251, 514 and 47 ng/ml, respectively. Corresponding mean values in remission were 270, 564 and 51 ng/ml, respectively, i.e. all three adhesion molecule values tended to be lower during active CH period than in remission. In 71% (n = 15) of the patients the sICAM-1 value was lower during active cluster periods than in remission. Similarly, sVCAM-1 and sE-selectin were lower in 67% (n = 14) and 67% (n = 14) of the patients in active periods, respectively. All three adhesion molecules were lower in active CH periods in 24% of patients. The decrease in active periods was statistically significant for sE-selectin (P = 0.027), but not for sICAM-1 or sVCAM-1 (Table 3).
Adhesion molecule levels in patients with episodic cluster headache (CH), giant cell arteritis (GCA) and in healthy control subjects

sICAM-1, Soluble intercellular adhesion molecule-1; sVCAM-1, soluble vascular cell adhesion molecule-1; sE-selectin, soluble E-selectin.
Results of the statistical analyses

CH, Cluster headache; GCA, giant cell arteritis; sICAM-1, soluble intercellular adhesion molecule-1; sVCAM-1, soluble vascular cell adhesion molecule-1; sE-selectin, soluble E-selectin. Mean and SD (standard deviation).
Statistical methods, two-tailed t-test for dependent samples when testing active CH vs. remission; Kruskal–Wallis

Mean levels (O) and standard deviations (vertical bars) of three different soluble adhesion molecules (sICAM-1, sVCAM-1, sE-selectin) in episodic cluster headache patients (CH) during active periods and in remission, in patients with biopsy-positive but untreated (active) giant cell arteritis (GCA) and in healthy headache-free control subjects (Controls). sICAM-1, Soluble intercellular adhesion molecule-1; sVCAM-1, soluble vascular cell adhesion molecule-1; sE-selectin, soluble E-selectin; ∗P-value < 0.05; ∗∗P-value < 0.01; ∗∗∗P-value < 0.001.
The mean sICAM-1 and sVCAM-1 values among GCA patients with active disease were 322 ng/ml and 610 ng/ml. sICAM-1 was significantly lower in CH patients during active periods than in GCA patients during active disease (P < 0.001), but sVCAM-1 and sE-selectin did not differ significantly between CH and GCA patients (Table 3).
The mean values of both sICAM-1 and sVCAM-1 among control subjects tended to be lower than among CH patients both during active periods and in remission. However, only the difference in sVCAM-1 levels among CH patients in remission compared with control subjects was statistically significant (P = 0.013) (Table 3).
Patients with active GCA had significantly higher mean values of sICAM-1 (P < 0001) and sVCAM-1 (P < 0001) compared with the control subjects (Table 3).
Mean values and SD of ESR and C-reactive protein (CRP) are reported in Fig. 2 (data on CH patients from Nilsson Remahl et al. (13)). The CH patients did not have pathologically increased ESR or CRP values either during active periods or in remission. GCA patients, in contrast, presented pathologically increased mean values of both ESR and CRP.
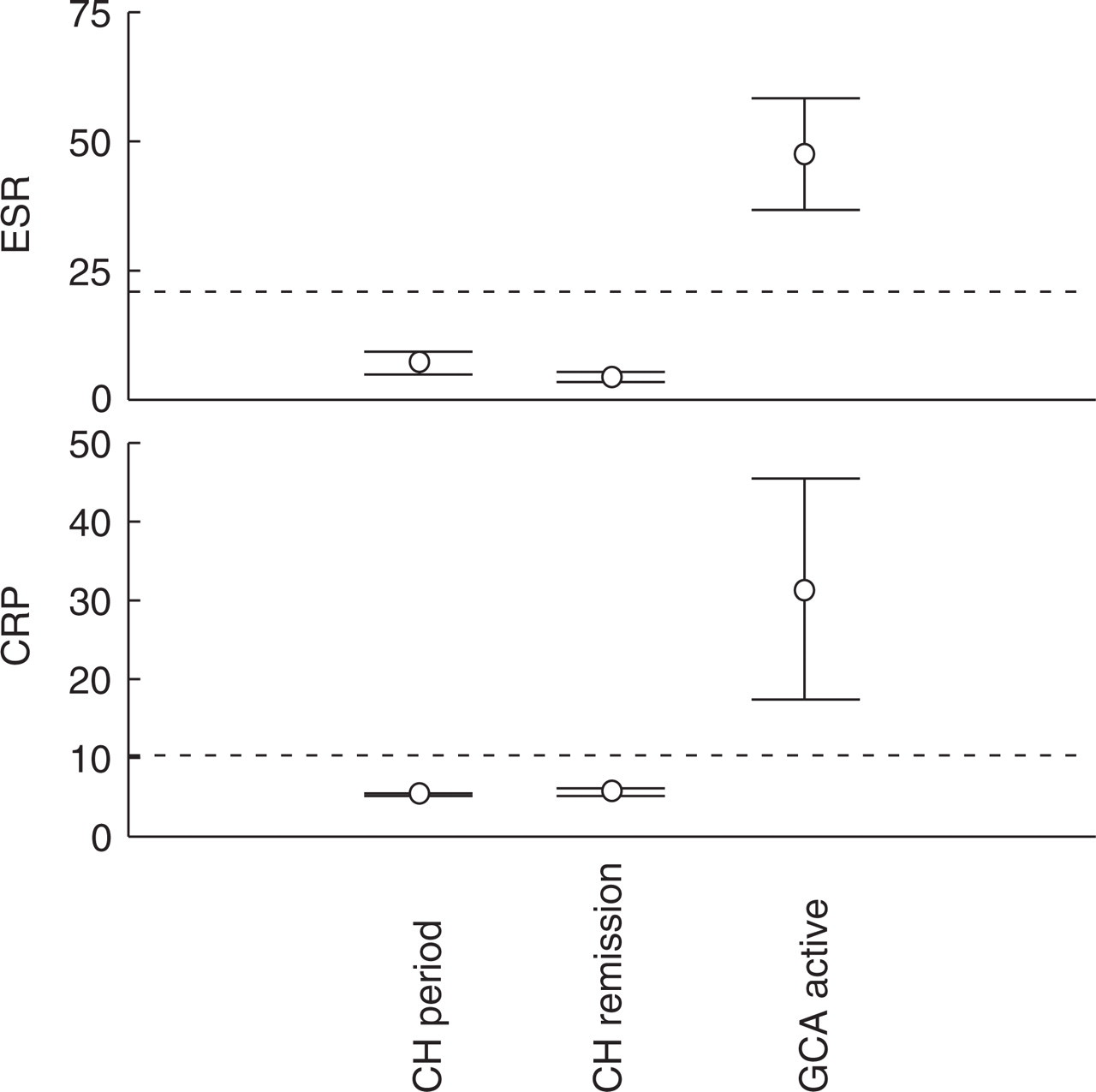
Plots of erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) in patients with episodic cluster headache during active periods and in remission and in patients with biopsy-positive but untreated (active) giant cell arteritis (GCA). The horizontal dotted line marks the upper level of the normal interval. Data on cluster headache patients from Nilsson Remahl et al. (13).
Discussion
Hannerz has presented the hypothesis that CH is caused by a vasculitic disorder affecting the retro-orbital vessels, presumably the venous circulation (8, 12). Especially amongst women, CH has been suggested to be one of many symptoms of a generalized inflammatory disorder, affecting several other organs (17, 18). In a previous study we could not repeat his observation of such a generalized disorder, either in clinical symptoms or in routine laboratory analyses (13). Gawel and coworkers, using gallium-67 citrate and SPECT technique, have found areas of increased activity in the ipsilateral parasellar region during active cluster periods between headaches in three of six investigated patients (19), indicating areas of active inflammation. Schuh-Hofer and coworkers have recently presented a report, based on observations in six CH patients, that a nitroglycerine-induced CH attack is not accompanied by extravasation of injected 99mTc-labelled human serum albumin in the cavernous region using SPECT and MRI techniques. This argues against leakage across the blood–brain barrier (11). Focal inflammation affecting the vessels (arteries or veins) in the retro-orbital region remains a possible pathophysiological mechanism causing the pain and the sympatheticoplegia during CH attacks.
In various vasculitic disorders, activated lymphocytes and macrophages produce cytokines, of which interleukin-1, tumour necrosis factor-alpha and interferon-gamma are the main inducers of adhesion molecule expression on endothelial cells (14, 15). Circulating adhesion molecules probably have a regulatory role, although their biological significance is unclear. Since soluble adhesion molecules are released by endothelial cells stimulated by cytokines, elevated concentrations have been considered a marker of endothelial activation. Furthermore, the adhesion molecules sICAM-1 and sVCAM-1 are expressed and released by activated lymphocytes and macrophages. Consequently, their increase may reflect immune activation.
In this study we have compared the levels of three adhesion molecules in CH patients, during active cluster periods and in remission, with GCA patients with a biopsy-verified active vasculitic disorder affecting large and medium-sized arteries in the trigeminal region, as well as with healthy control subjects. We have confirmed the findings by Coll-Vinent et al. (15), who found elevated levels of sICAM-1 in patients with clinically active GCA. Among CH patients, however, the sICAM-1-values were significantly lower both during active periods and in remission compared with active GCA, and sICAM-1-values did not differ between CH patients and control subjects. Furthermore, Coll-Vinent et al. have reported that sICAM-1 correlates with the disease activity, thus decreasing after treatment with corticosteroids. sICAM-1 mean values in CH patients showed a reverse trend with a non-significant increase in remission compared with active disease.
Our study has revealed a significant increase in sVCAM-1 among patients with active GCA compared with healthy control subjects, which is contrary to the findings of Coll-Vinent et al., who could not find such a difference. sVCAM-1 was also significantly increased in CH patients in remission, but not during active periods, compared with healthy control subjects.
sE-selectin was significantly increased among CH patients in remission compared with active CH periods, but did not differ compared with active GCA or control subjects.
We interpret the following results as evidence that CH is not a vasculitic disorder affecting the medium-sized arteries of the retro-orbital region: (i) the significantly lower mean values of sICAM-1 in CH patients during active cluster periods compared with GCA patients during active disease; (ii) the trend of down-regulation of sICAM-1 during active CH compared with in remission, which is the reversed pattern observed in active compared with corticosteroid-treated GCA (15); (iii) the observation that neither ESR nor CRP was increased in CH compared with active GCA. Our study thus supports the view that CH is mainly a disorder of the central nervous system (4).
In migraine patients sICAM-1 has been shown to be down-regulated both during spontaneous and nitric oxide-induced headache attacks compared with the attack-free state, which is in line with our results (20, 21). Our results of significantly increased sVCAM-1 values in remission compared with healthy controls, as well as the small but significant decrease in sE-selectin levels between active CH compared with in remission, have not previously been reported. These findings suggest a pattern of down-regulation of adhesion molecules, which indicates that changes actually take place in the immune system during the different states of CH, and that these changes indicate suppression of the immune system during active CH. These results may reflect that patients with vascular headache, migraine and CH, may have a differently reacting immune system compared with patients with vasculitic disorders such as GCA and healthy control subjects.
The following items may have affected the results of our study. First, the analyses were performed on blood from the cubital vein, which is far from the supposed focus in the retro-orbital region or the brain itself (20). However, it is possible to detect the inflammatory disorder GCA using peripheral blood samples, which may reflect that GCA is actually a generalized disorder. Second, the difference in mean age between the investigated groups of patients may have affected the results. Contradictory reports have been published on the issue of age-dependent changes in sICAM-1, sVCAM-1 and sE-selectin (22, 23). Third, the proportion of women was larger in the GCA group than in the CH group. One may speculate whether the immunological response with respect to the studied adhesion molecules differs between men and women (c.f. 24). The possible influence of age, and probably also gender, must be kept in mind when interpreting the results of this study. Fourth, the minor changes in levels of the studied adhesion molecules may reflect effects of pain and stress on the immune system (25). Obviously, further studies on the role of the immune system in vascular headache and specifically in CH are needed.
Footnotes
Acknowledgements
This study was supported by grants from Astra Zenica and Glaxo Smith Kline. The authors are grateful to Karl Ekbom for inspiring discussions and to Inger Vedin for expert technical assistance. Many thanks also to statistician Elisabeth Berg for her valuable help with the calculations.
