Abstract
Neuroimaging methods have been widely used in headache and migraine research. They have provided invaluable information on brain perfusion, metabolism and structure during and outside of migraine attacks, contributing to an improved understanding of the pathophysiology of the disorder. Human models of migraine attacks are indispensable tools in pathophysiological and therapeutic research. This review of neuroimaging methods and the attack-provoking nitroglycerin test is part an initiative by a task force within the EUROHEAD project (EU Strep LSHM-CT-2004-5044837-Workpackage 9) with the objective of critically evaluating neurophysiological tests used in migraine. The first part, presented in a companion paper, is devoted to electrophysiological methods, this second part to neuroimaging methods such as functional magnetic resonance imaging, positron emission tomography and voxel-based morphometry, as well as the nitroglycerin test. For each of these methods, we summarize the results, analyse the methodological limitations and propose recommendations for improved methodology and standardization of research protocols.
Functional magnetic resonance
D Magis, PS Sandór, M Sanchez del Rio, GG Schoonman & J Schoenen
We will focus on the following magnetic resonance (MR) techniques that have been used in migraine: blood oxygen level dependent functional magnetic resonance imaging (BOLD-fMRI), MR spectroscopy, diffusion-weighted and perfusion-weighted MRI.
Generalities on optimal method, personal experience
Until now the number of studies has been too small to establish an optimal protocol. Inclusion criteria should be identical with electrophysiological studies (see above), and as strict as possible, e.g. the timing of the study is critical, as neurophysiological patterns are likely to be different during premonitory, postdromal and interictal phases; a comprehensive discussion of these differences can be found in the electrophysiological paper.
There are no studies concerning the repeatability of functional MR techniques in migraine, and there are no studies correlating results with those obtained from, for example, electrophysiological techniques. Therefore, functional MR techniques are, until proven otherwise by means of proper methodological data, not to be regarded as diagnostic tests for individuals. The few studies on repeatability of electrophysiological methods in migraine (1, 2), suggest that, due to the weak signal-to-noise ratio, their results are of interest for the study only of groups, not of individuals. This might be due to the fact that the electrophysiological signal is obtained from the scalp surface. In contrast, fMRI techniques look non-invasively ‘into’ the brain and there is no such limitation; the probability is therefore higher that methodological improvements will result in a signal-to-noise ratio strong enough to be useful for diagnostic purposes.
BOLD-fMRI
Definition
The BOLD-fMRI signal, a surrogate marker of neuronal activation, was first described by Ogawa et al. (3). Changes in blood flow and the proportion of oxy- and deoxyhaemoglobin are responsible for local magnetic changes which can be detected (3). The signal increase in activated areas is thought to be related to the relative hyperaemia in these regions of neuronal activation, resulting from blood flow increase proportional to tissue oxygen demand. This technique is widely used to understand how the human brain responds to various conditions: when a brain region is used (or not), the blood flow is relatively increased (decreased) in this area.
Literature data
The literature concerning the BOLD technique is focused on the migraine attack and limited to a small number of studies (Table 1). Most of them have explored the visual cortex [visually triggered migraine (4, 5) and migraine with aura (MA) (6, 7)].
Main fMRI studies

The studies performed during spontaneous migraine attacks showed BOLD changes compatible with a brief activation followed by prolonged neuronal depression spreading over the cortical surface at a speed of 1–4 mm/min suggestive of the phenomenon of ‘cortical spreading depression’ (6) and, correlating with the migraine aura, brainstem activation for visually triggered MA or migraine without aura (MoA) (5). Interictally, Vincent et al. (7) have found enhanced reactivity of the visual cortex compared with the control population. Rocca et al. (8) have demonstrated in patients with MoA an extended area of cortical activation during a motor task, which was correlated with the amount of white matter abnormalities.
Limitations
There is an important limitation as far as temporal resolution of BOLD-fMRI is concerned: a 2–4-s haemodynamic delay has been found between neuronal activity changes seen by electrophysiology and imaging (9). This has to be taken into account particularly for future studies in migraine searching for correlations between electrophysiology and imaging. Nevertheless, this delay should not affect the recording of repetitive events, such as cortical spreading depression.
Moreover, the exact relationship (linear or not) between the magnitude of the BOLD signal and the underlying neuronal activation remains obscure.
As far as more ‘technical’ details are concerned, BOLD-fMRI recordings can be easily artefacted by head movements, which limits the protocols that can be studied. Artefacts are also often seen in regions close to air, e.g. the sinuses and the orbitofrontal cortex.
Magnetic resonance spectroscopy
Definition
Magnetic resonance spectroscopy (MRS) of the brain is a non-invasive technique providing biochemical information about metabolites and, especially if used with a functional paradigm, neuronal function. Whereas other MRI techniques listed in this review (BOLD-fMRI, diffusion-weighted MRI and perfusion-weighted MRI) are based on 1H ‘resonance’ (flip) in water molecules, in MRS 1H atoms in other molecules, or other atoms such as 31P, are being resonated. The principle of MRS is that nuclei in different chemical structures have different resonance patterns. The molecular composition of a given brain region (called a ‘voxel’) is displayed as a spectrograph where the quantity of a substance is positively correlated with the area under the peak value. 1H MRS allows the detection of the following compounds: N-acetylaspartate, lactate, glutamate, aspartate, GABA, choline, myoinositol and creatine, whereas 31P is able to measure intracellular pH, ATP metabolism and phospholipid metabolism.
Literature data
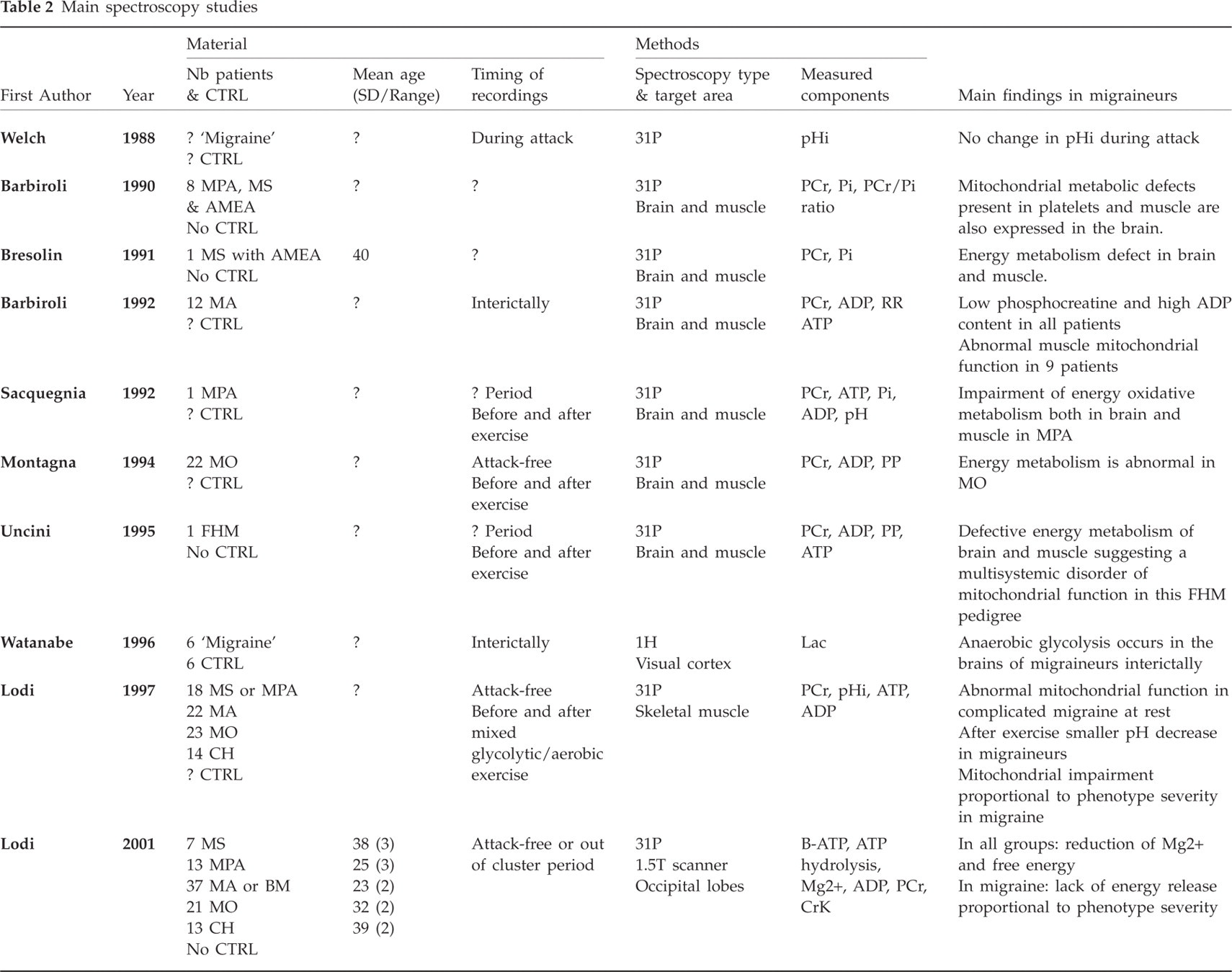
The available MR spectroscopy studies performed in migraine are listed in Table 2. The most common method used in migraine at present is 31P spectroscopy (10–19). More recently, 1H spectroscopy studies (20–23) have also been performed, some of them with a functional paradigm. Ictal and, more often, interictal recordings are available, and, as far as 31P spectroscopy is concerned, brain and muscle studies before and after an exercise. Most (11–18, 20–23), but not all (10, 19), studies are compatible with an abnormal energy metabolism, possibly due to mitochondrial dysfunction, i.e. a reduced phosphorylation potential. Abnormal magnesium Mg2+ concentrations have been found in all migraine types by one study (18). In another trial (19), a significant decrease of Mg2+ was seen in hemiplegic migraine patients only, with a trend to abnormality in other migraine subgroups. Although the studies on brain metabolism were performed by different research groups, all studies on muscle metabolism in migraineurs came from the same group and have yet to be confirmed by others. MR spectroscopy abnormalities are found in all migraine subgroups, including MoA (15, 17, 18), and seem proportional to the severity of the migraine phenotype (17–19, 21).
Main spectroscopy studies

Limitations
As the number of the molecules studied with MRS is much lower than that of water molecules, wider areas and longer periods are needed to detect a signal. There is consequently a limitation of spatial and temporal resolution in comparison with conventional MRI. However, new techniques are being developed to reduce this constraint. Moreover, MRS is limited to mobile compounds and therefore cannot be used for the study of neurochemical receptors.
Diffusion-weighted MRI
Definition
Diffusion-weighted (DW) MRI is based on random 1H movement in water molecules (24). The apparent diffusion coefficient (ADC) is the amount of water diffusion for a given pixel. Regions with low ADC will appear hyperintense. DWMRI is mostly used in the early diagnosis of acute cerebral ischaemia (24). It can detect a decrease in the ADC of the ischaemic brain, correlated with cytotoxic oedema and neuronal energy loss.
DWMRI can also be helpful in the detection of myelination (25).
Literature data
Most publications using DWMRI in migraine are case reports performed during spontaneous or persistent auras (26–28) and report normal findings (Table 3). Recently, Rocca et al. (8, 29), used diffusion-tensor imaging in migraine patients with and without MRI white matter lesions. They found that migraine patients had abnormalities of normal appearing white matter which were mild compared with other diseases associated with white matter abnormalities (29).
Main Diffusion-weighted and Perfusion-weighted MRI studies

Limitations
DWMRI provides information about the effect of a recent ischaemic insult, but does not give information concerning the integrity of the vascular bed.
Perfusion-weighted MRI
Definition
Perfusion-weighted (PW) MRI is used to assess the blood supply of the brain (blood flow, blood volume and tissue mean transit time), i.e. the rate at which blood is delivered to the tissue. There are two main approaches: bolus tracking (more frequently employed, using an injected gadolinium-containing compound) and arterial spin labelling (without contrast product infusion).
Literature data—method
The literature on PWMRI is limited to case reports of MA, prolonged aura or familial hemiplegic migraine (FHM); partially contrasting results have been reported (Table 3). Cutrer et al. (26) and Relja et al. (30) have found cortical hypoperfusion during the aura, whereas in the study of Smith et al. (28) prolonged aura was associated with hyperperfusion and vasogenic leakage. Gutschalk et al. (27) performed PWMRI during a prolonged attack of FHM and found no change in perfusion during hemiparesis. Sanchez del Rio et al. found no change of perfusion in MoA (31).
Limitations
The bolus-tracking approach using intravenous gadolinium infusion cannot be repeated as often as less harmful techniques. Moreover, in migraine, PWMRI seems to be of interest only during or immediately before/after the attack. In the bolus-tracking method, the presence of a reduced arterial diameter may cause bolus dispersion, which has been shown to introduce significant errors in cerebral blood flow (CBF) estimations. It remains to be determined if this could explain part of the contradictory results seen in migraine studies.
Positron emission tomography
S Afridi & PJ Goadsby
Definition
Positron emission tomography (PET) is a tomographic nuclear imaging procedure, which uses positrons as radiolabels and positron-electron annihilation reaction-induced gamma rays to locate the radiolabel. It can provide a quantitative measurement of blood flow. The PET scanner consists of multiple detector rings to scan a number of transaxial planes simultaneously. In H2 15O PET, water is labelled with a positron emitter (15O) and injected intravenously into the patient, who is scanned by the tomographic system. The PET images are coregistered with a structural scan, usually an MRI, to enable accurate anatomical localization.
Literature data
Main PET studies mentioned below are summarized in Table 4.
Main PET studies


Spontaneous migraine
In one of the earliest PET reports Woods et al. described a case report of a subject with migraine with no previous aura who unexpectedly developed a migraine during her participation in a visual activation paradigm whilst lying in a PET scanner (32). The subject described some visual blurring during one of the scans, but did not clearly describe any other features of typical aura. The migraine was associated with bilateral hypoperfusion starting in the occipital lobes and spreading anteriorly into the temporal and parietal lobes. The contiguous spread covered areas in the territories of the posterior and middle cerebral artery. In a later study, nine subjects were studied within 13 h of onset of MoA (33). They observed a 9.9% decrease in global CBF and a 5.2% decrease in cerebral blood volume persisting for at least 6 h. Oxygen metabolism and oxygen extraction remained unchanged. Most recently, a further study of spontaneous migraine has also reported bilateral posterior circulation hypoperfusion in MoA (34). These changes may represent hypoperfusion associated with cortical spreading depression (35), albeit without any clinical manifestations. Alternatively, they may represent a neurogenically mediated oligaemia, which may be seen in experimental animals with activation of the nucleus locus coeruleus (36) and is typically dominant in the posterior cortical structures (37).
The first PET study detailing regional activation during migraine was the study of Weiller and colleagues (38). Nine subjects with MoA presented with right-sided spontaneous headaches and were scanned within 6 h of onset of migraine, prior to having taken any medication. They were scanned during spontaneous migraine attacks, following sumatriptan and interictally. The study revealed brainstem activation during the migraine, which persisted after sumatriptan administration had relieved the pain. The resolution of the PET camera used was not high enough to identify specific nuclei, but the foci of maximum increase were around the dorsal midbrain and dorsolateral pons. Activation was also seen in the anterior cingulate, visual and auditory association cortices.
In a more recent study involving both MA and MoA, five patients were imaged in ictal and interictal states and the differences were analysed using statistical parametric mapping (39). Two patients had a typical migrainous aura prior to the onset of the headache. All the attacks studied fulfilled standard diagnostic criteria for migraine (40). Comparing the migraine scans with interictal scans there was significant activation in the dorsal pons, lateralized to the left, irrespective of the anatomical location of the pain. Activation was also seen in the right anterior cingulate, posterior cingulate, cerebellum, thalamus, insula, prefrontal cortex and temporal lobes.
Triggered migraine
A PET study by Andersson and colleagues (41) has investigated attacks provoked by red wine in 11 subjects with MA and MoA. They demonstrated reductions in regional CBF (23%) and in oxygen metabolism (22.5%) in an area corresponding to the primary visual cortex. They did not detect any significant increases in blood flow during aura or headache.
A case of a glyceryl trinitrate (GTN)-triggered migraine also revealed brainstem activation, on this occasion in the dorsolateral pons, which again persisted following abortion of the migraine (42).
The largest PET study to date involved 24 migraineurs (with and without aura) and eight healthy controls (43). The migraineurs were divided into three groups according to the site of their headache: right/left/bilateral. In each group a migraine was induced using a GTN infusion. The subjects were scanned at various points (preinfusion, during GTN, during migraine, post migraine). Significant brainstem activation was seen in the dorsal pons during the migraine state vs. the pain-free state when comparing migraineurs with controls. When each group was analysed separately to investigate laterality it was found that the dorsal pontine activation was ipsilateral in the right-sided and left-sided groups and bilateral in the bilateral headache group with a left-sided preponderance.
Chronic migraine
A PET study has examined eight patients with chronic migraine who had had bilateral suboccipital stimulators implanted. The authors reported activation in the dorsolateral pons that persisted when the stimulator was on and the patients had pain relief (44). The area was the same as demonstrated in the episodic migraine cases described above, suggesting that both chronic and episodic migraine share important common biological features in terms of areas of brain activation. Remarkably, the main difference between the pain state and the pain-free state was a change in the pattern of thalamic activation.
Limitations
Although the temporal and spatial resolution provided by PET is relatively good, it is lower than for fMRI. The spatial resolution of PET is dependent upon the position and type of detectors. The temporal resolution is dependent on the time taken for the tracer to reach the brain and on the half-life of the tracer (124 s for 15O). This enables a relatively good comparison of prolonged brain states such as migraine vs. pain free, but is not fast enough to follow, for example, progression of migraine aura.
Another limitation of PET is that during the scanning process ionizing radiation is administered, limiting the number of PET scans per subject.
Voxel-based morphometry
A May
Definition
Voxel-based morphometry (VBM) is a whole-brain technique capable of investigating subtle, regionally specific changes in grey matter by averaging across subjects. It gives information about density and volume. VBM has been cross-validated with region-of-interest measurements and functional data in a number of studies (45–47). This method is based on high-resolution structural T1-weighted 3D MR images, registered in a common stereotactic space, and is designed to find significant regional differences throughout the brain by applying voxel-wise statistics in the context of Gaussian random fields (48).
Literature data
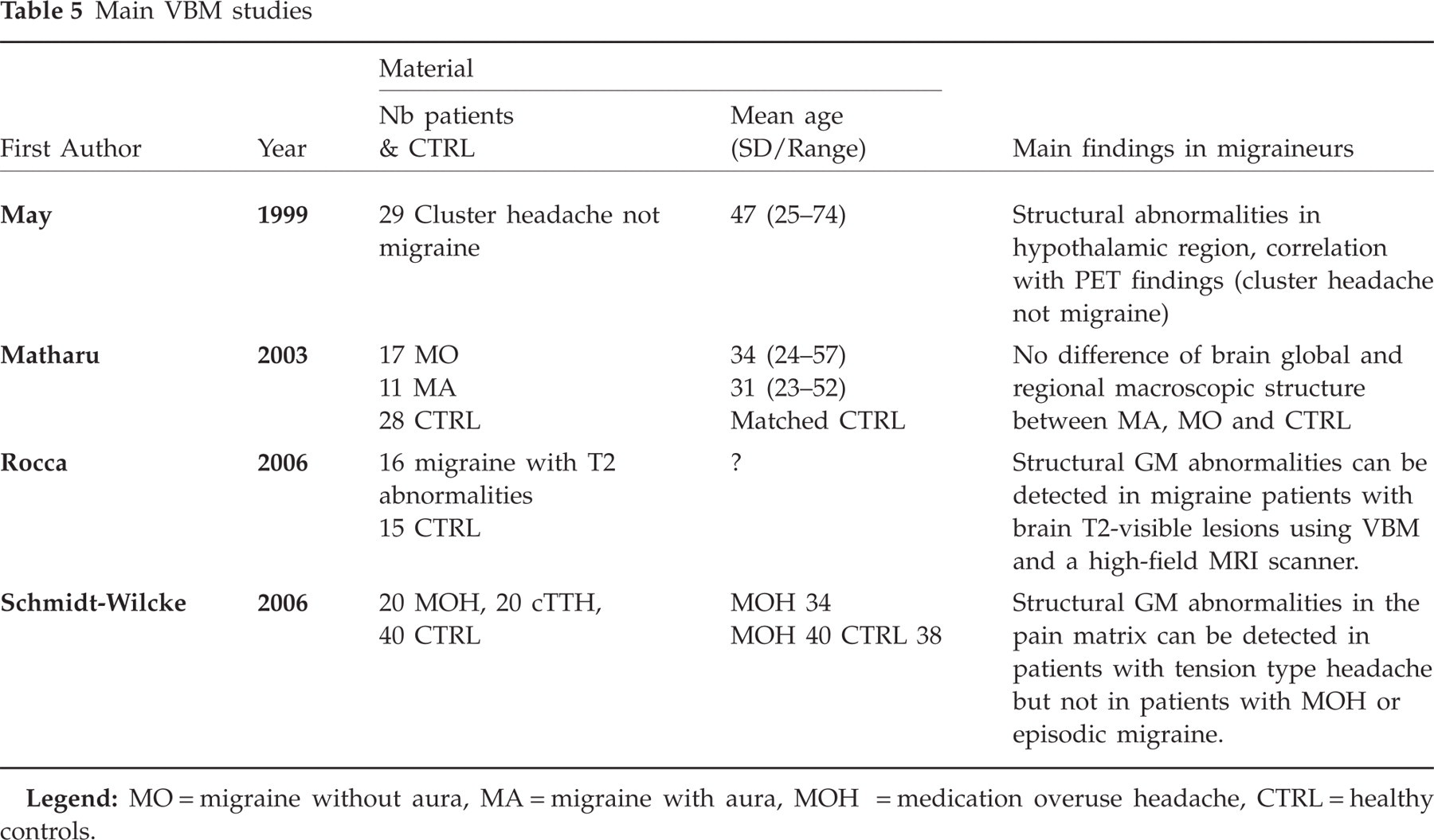
Unlike functional data using PET and fMRI, where a substantial amount of data is available, there are to date only four VBM papers on headache (49) (Table 5), two on chronic back pain (50, 51) and one on phantom pain (52). Of the headache studies, one studied patients suffering from cluster headache (47), one MA and MoA patients (53) and the third chronic tension-type headache (TTH) and medication overuse headache (54). All three studies compared patients with healthy controls (cohort studies). In cluster headache a significant unilateral increase of grey matter in the posterior inferior hypothalamus was found. In TTH, but not in patients with medication overuse headache, a significant negative alteration in grey matter was found in the pain matrix (cingulate cortex, anterior insulae, brainstem) (54). However, no significant morphometric changes were found in grey or white matter between patients suffering from episodic migraine and healthy controls. In contrast, a recent study of VBM in migraineurs with white matter T2 hyperintensities has shown decreased grey matter density in frontal and temporal areas, but increased densities in periaqueductal grey matter and dorsolateral pons. Reduced, but not increased, grey matter density was strongly related to age, disease duration and T2-visible lesion load (55).
Main VBM studies
Advantage of the method
Given that cluster headache and migraine have subtly different brain structures, the question arises whether other primary headache types may also be distinguishable on a morphological level. The advantage of VBM studies is that the method is non-invasive (T1-weighted MRI scans) and that it can be replicated in the same individual at different time points, e.g. during an attack or after pharmacotherapy.
Limitations of the method
VBM has still to prove that it generates reproducible results. VBM is for research purposes only and requires groups of at least 20 subjects per group to produce stable results. VBM is sensitive for changes in grey matter, and recent cross-sectional VBM studies have demonstrated learning-dependent changes in adult human brain and suggested anatomical correlates for navigation, arithmetic, linguistic, procedural and musical learning abilities (56–60). However, VBM is not sensitive for white matter changes, where another morphometric method, diffusion tensor imaging, would be the method of choice (61). Because of the limited knowledge regarding the cellular substrate detectable with VBM, we can only speculate about the nature of the structural changes. VBM would detect changes of grey matter concentration per voxel as well as changes of the classification of individual voxels, e.g. from white to grey matter (62) and is probably a combination of both. In general, an increase of grey matter could be due to a simple increase in cell size, neural or glial cell genesis, spining and even changes in blood flow or interstitial fluid. Finally, the need to average in Talairach's coordinates, i.e. the standard reference for brain locations in the scientific literature, may lead to include ‘mixed’ voxels containing grey and white matter as well as cerebrospinal fluid in certain areas. Independently of these limitations, there is a need for better standardization between different studies and centres (63).
Recommendations for improved methodology
To date, no standardized method allows comparisons between laboratories. If the software package Statistical Parametric Mapping (SPM) is used, the optimized protocol by Good et al. (62) is relevant.
The following information should be included in the Methods section of published studies:
Data acquisition: scanner type and voxel volume.
Data preprocessing: which SPM. Whether or not the optimized protocol by Good et al. (62) has been used. Choice of template. Information regarding modulation of volume changes. Smoothing factor.
Data analysis: if small volume correction is used: information about the choice of sphere volume.
Information about grey and white matter changes in both directions (increases or decreases).
Which optimal methodology can be proposed in headache studies?
In clinical practice, to obtain reliable and reproducible VBM data, the following method is recommended:
For cohort studies: n > 15 per group, better >20 per group
Age and gender matching (variables influencing cortical and subcortical anatomy)
Homogeneous groups, optimum: genetically determined cohorts
Always the same scanner with identical imaging parameters
Smoothing factor of 10 mm full width at half maximum
Longitudinal analysis (same patient over time) better than cohort analysis (patients vs. controls)
Additional regression analysis using external parameters (such as electrophysiological data, etc.) to better fit the model of the a priori hypothesis.
Studies needed
Migraine has a strong genetic component and, to render groups as homogeneous as possible, the ideal inclusion criteria could be based on genotype (cohort study) or response to treatment (longitudinal study including controls)
Standardization of protocols is needed
A way to compare directly groups investigated on different scanners is needed (65)
Histological data of the structures identified with VBM is needed
External parameters (neurophysiological data see evaluation and proposal for optimization of neurophysiological tests in migraine) need to be compared to changes in brain structure.
Migraine-provoking test
L Bendtsen, H Kaube & G Sandrini
Genuine migraine is difficult to study because of its attack-wise appearance. A large number of migraine-provoking agents have been described, including nitroglycerin (NTG), histamine, prostacyclin, reserpine, m-clorophenylpiperazine, adenosine, tyramine-containing foods, chocolate, sustained tooth clenching (for review see (66)), calcitonin gene-related peptide (CGRP) (67) and sildenafil (68). However, NTG is the only substance for which there is sufficient information on reproducibility and reliability.
Definition
GTN is widely used for its antianginal effect, and it has been known for >150 years that one of its most common side-effects is headache. It is believed that GTN is metabolized to nitric oxide and/or S-nitrosothiols, and that these metabolites cause the headache induced by GTN (66). GTN has been used as a migraine-provoking substance for more than five decades, and a specific human experimental model of migraine using GTN has been developed and tested in a series of studies by Iversen and colleagues within the last two decades (66, 69). More recently, other groups have adopted this model (43, 70–73).
Literature data
The main literature is listed in Table 6.
Main migraine-provoking tests studies



Iversen et al. have reported acceptable day-to-day variation in headache intensity experienced by healthy volunteers following intravenous GTN. Out of 50 paired infusions, only five differed by more than one score when scoring maximal headache intensity on an 0–10 scale (74). The standard deviation of the day-to-day difference of headache scores was 0.7, which allows detection of a difference of one headache score, with type 1 and 2 errors of 5%, in a crossover study of 12 subjects (66). Headache characteristics were also reproducible. In 10 healthy subjects headache quality did not differ and in one subject only the localization differed slightly between days (74). Thus, NTG induced headache with acceptable reproducibility in healthy subjects.
Olesen et al. compared GTN-induced headache responses in 17 sufferers with migraine, 17 headache-free subjects and nine patients with episodic TTH (75). Migraineurs developed an immediate headache within the first hour after infusion that most often did not fulfil the criteria for migraine, but 11 of the 17 migraineurs developed a delayed typical migraine within the next 24 h. Only one of the controls developed a delayed migraine. Thomsen et al. (76) infused GTN in 10 patients suffering from MoA. Eight patients developed immediate headache, only one of which fulfilled migraine criteria, whereas eight patients developed a regular migraine attack within 12 h. Peak migraine headache occurred at a mean of 5.5 h after the infusion. Pain characteristics and accompanying symptoms were very similar to the spontaneous attacks recorded by the patients. Christiansen et al. induced migraine attacks, but no aura, in six of 12 MA patients (77). Juhasz et al. have reported that sublingual NTG induced attacks of MoA in 10 of 15 patients with MoA and in two of eight controls (70). Afridi and colleagues (43) challenged 23 patients without and 21 patients with aura with GTN. Migraine attacks were triggered in 33 (75%) of the patients (83% in MoA and 67% in MA). Only one of these attacks was MA. Twenty-nine of the 33 patients who developed migraine were re-challenged at least a week later, and all but one developed a repeat attack. In one patient a visual aura was triggered on both occasions. Thus, NTG induced MoA with high reproducibility both in MoA and in MA.
Sances et al. have studied the reliability of NTG-induced migraine in a large study including 197 migraineurs, 175 without and 22 with aura (71). The study was methodologically well-performed. Thus, all diagnoses were made on the basis of a diagnostic headache diary kept during a 12-week run-in period, and the NTG test was performed by an observer blinded to the diagnosis. Nitroglycerin was given sublingually. The authors calculated sensitivity (the probability that a test will be positive in a subject affected by a given disease), specificity (the probability that the test will be negative in a subject not affected by a given disease) and accuracy (proportion of true-positive and true-negative tests out of total number of tests performed). A total of 138 of 175 patients with MoA (82.1%) developed MoA within the 8-h observation period, and the same was true for two of 53 healthy controls. In MoA the test sensitivity was 82.1%, specificity 96.2% and accuracy 85.5%. The reliability of the NTG test was less satisfactory in patients with MA, where only three of 22 patients developed MA (sensitivity 13.6%, specificity 96.2% and accuracy 72.0%). Thus, reliability of the NTG test was satisfactory in MoA but not in MA.
Advantages of the method and perspectives
There are a number of practical advantages of the NTG migraine-provoking test, together with characteristics of the induced attack very similar to the genuine attack that make it an interesting tool for the exploration of migraine pathophysiology. First, it is reliable, reproducible and critically evaluated by several independent groups. Second, it is able to trigger premonitory symptoms (yawning, tiredness, irritability, neck stiffness, frequency of urination, hunger and low mood) similar to those of the genuine attack (78). Third, the triggering aspect of the test bears resemblance to genuine attacks which are also often triggered, e.g. by stress, hormonal factors or alcohol. Fourth, it responds to acute (79) and prophylactic medications (80) known to have an effect in genuine migraine. Fifth, the efficacy of nitric oxide inhibitors in acute migraine (81) demonstrates that nitric oxide Key molecule in migraine. Thus, the NTG test provides excellent opportunities for prospective studies of the migraine attack, given that the limitations mentioned above are considered when interpreting the findings.
Premonitory symptoms can be studied prospectively (43) and central processes can be studied during the attack, e.g. central pain processing by means of the nociception-specific blink reflex (82), auditory evoked potentials (83), visual evoked potentials (84) and laser evoked potentials (72). Imaging studies may provide information about temporal and spatial activation of various pain-sensitive centres in the brain (43, 73), and biochemical studies may unravel the important changes in, for example, nitric oxide (85) and CGRP (86) concentrations during the attack. The mechanism of action of antimigraine drugs could be explored, e.g. by recording of plasma CGRP concentrations during sumatriptan treatment of NTG-induced migraine attacks (87). Genetic factors could be studied, e.g. by comparing the response to the NTG test between patients with and without a strong family history of migraine. The test can be used to evaluate the efficacy of acute (79) and prophylactic migraine medications (80). Rare forms of migraine, e.g. FHM or basilar type migraine, could be investigated in multicentre studies, as the test methodology can be strictly standardized. Furthermore, since the test can be applied also in animals, it provides opportunities to bridge the gap between animal and human experimental migraine models. Thus, there are wide perspectives for using the test in migraine as well as in other primary headaches in the future.
Limitations of the method
The major limitation of the NTG migraine-provoking test is that, although the induced migraine has many similarities with a spontaneous attack, it is not a genuine attack. This implies that it is unknown whether the changes observed, e.g. in imaging or biochemical studies, would also be true for spontaneous attacks. It should also be considered whether changes observed in the laboratory are caused by the attack itself or by laboratory-induced stress.
The long period needed to observe the delayed headache puts a burden on the patient and makes studies time-consuming and expensive. Moreover, the likely outcome of the provocation, the migraine attack, is not exactly what the normal migraine patient is imagining. Together, these factors impede the inclusion of a large number of subjects in the studies.
Recommendations for improved methodology
It is of crucial importance that headaches are classified meticulously before as well as during the NTG test, because NTG has a differential effect in MoA and in MA, as described above, and because NTG induces cluster headache in cluster patients (71, 88), and TTH in patients with chronic TTH (89). The NTG test should be performed by a trained observer who is blinded to the diagnosis.
The limited number of available studies indicates that the sensitivity of the NTG test for provoking an attack of MoA in MoA patients is approximately 80% whether provocation is done with intravenous infusion of GTN 0.5 µg/kg per min over 20 min (43, 76) or with 0.9 mg NTG sublingually (71). Using lower doses significantly decreases the sensitivity of the test (80). Higher doses of GTN cannot be recommended, since dose–response studies have demonstrated a ceiling effect at 0.5 µg/kg per min (74). Given the comparable sensitivity of intravenous GTN and sublingual NTG, it is unlikely that increasing the sublingual NTG dose will be beneficial, but this has not been examined. End-points should be predefined and may include maximal headache intensity, area under the headache curve or sum of pain scores and the type of headache provoked. At least a 2-day interval between GTN challenges is recommended in healthy subjects and longer, e.g. 1 week, in migraineurs (66).
Acknowledgements
The leading authors D.M. and J.S. are grateful to Mrs P. Gerard for help with literature search and referencing. This work is supported by the EU STREP EUROHEAD (LSHM-CT-2004-5044837) (Workpackage 9).
