Abstract
Background
Validated chronic migraine biomarkers could improve diagnostic, prognostic, and predictive abilities for clinicians and researchers, as well as increase knowledge on migraine pathophysiology.
Objective
The objective of this narrative review is to summarise and interpret the published literature regarding the current state of development of chronic migraine biomarkers.
Findings
Data from functional and structural imaging, neurophysiological, and biochemical studies have been utilised towards the development of chronic migraine biomarkers. These biomarkers could contribute to chronic migraine classification/diagnosis, prognosticating patient outcomes, predicting response to treatment, and measuring treatment responses early after initiation. Results show promise for using measures of brain structure and function, evoked potentials, and sensory neuropeptide concentrations for the development of chronic migraine biomarkers, yet further optimisation and validation are still required.
Conclusions
Imaging, neurophysiological, and biochemical changes that occur with the progression from episodic to chronic migraine could be utilised for developing chronic migraine biomarkers that might assist with diagnosis, prognosticating individual patient outcomes, and predicting responses to migraine therapies. Ultimately, validated biomarkers could move us closer to being able to practice precision medicine in the field and thus improve patient care.
Introduction
Chronic migraine (CM) affects 1% to 2% of the global population (1) and 2.5% of persons with episodic migraine (EM) progress to CM each year (2). Compared to EM, CM is associated with more headache-related disability, higher rates of comorbidities, and lower health-related quality of life (3,4).
According to the US Food and Drug Administration, biomarkers are characteristics that can be objectively measured and evaluated as an indicator of normal biological, pathological, or pharmacological processes (5). Biomarkers for migraine should be helpful in diagnosing the disease and its subtypes, prognosticating its progression, and predicting the outcome of a therapeutic intervention (6).
Despite the prevalence and the burden of the disease, there are no validated biomarkers for CM. The diagnosis of CM is purely clinical and based on the criteria established by the International Classification of Headache Disorders (ICHD-3) (7). Currently, there can be partial subjectivity when making migraine diagnoses, and substantial difficulty when trying to prognosticate patient outcomes and predicting responses to migraine therapies. The diagnostic process is more complicated when migraine becomes chronic, especially because a) the characteristics of pain change and are less specific, b) the diagnostic criteria for CM are debated, and in continuous evolution, and c) the differential diagnosis widens (8,9).
Improvement in the diagnostic process would be facilitated by the identification and development of diagnostic biomarkers in the migraine field. Prognostic and predictive biomarkers would also be of great value.
In this review, we summarise and interpret the main studies in the literature (based on PubMed searches for neurophysiology, imaging, and biochemical study methods combined with chronic migraine, treatment, medication overuse, or classification), that contribute to the identification and development of CM biomarkers using imaging, neurophysiology, and neuromolecular techniques.
Brain imaging: Structural and functional chronic migraine biomarkers
Structural and functional brain imaging data can be used to develop models for prognosticating migraine patterns, predicting treatment response, and classifying migraine. For these imaging biomarkers to be most useful, they need to have high accuracy and to be applicable at the level of the individual patient rather than at the level of the patient group. In addition, the imaging and data analysis techniques have to be highly replicable, so that the imaging biomarker performs across different healthcare settings.
Efforts towards the development of imaging biomarkers have utilised the large datasets that result from brain imaging studies, such as MRI, within machine-learning multivariate pattern and deep learning analyses that identify the optimal set of imaging measurements for accurate prognostication, prediction, or classification.
CM is associated with brain structure and function that differ from EM and non-migraine healthy controls, and greater headache frequency is positively correlated with the extent of structural and functional aberrations (10–12). For example, a functional MRI (fMRI) study demonstrated that interictal migraineurs have greater pain-induced brain activations of several pain processing regions compared to healthy controls (12). Similarly, another fMRI study demonstrated that, compared to EM, CM is associated with stronger activation of the hypothalamus in response to painful trigeminal stimulation (13). Resting state functional connectivity analysis of CM revealed altered connectivity of the default mode, salience, and central executive networks compared to healthy controls and a positive association between the frequency of moderate and severe headache days with the extent of abnormal connectivity in the salience and central executive networks (14,15). A resting-state functional connectivity study comparing CM to EM demonstrated that those with CM have stronger functional connectivity within the pain matrix (16).
Similar to the functional imaging studies, structural imaging studies demonstrate that higher headache frequency is associated with greater abnormalities in brain structure. Compared to healthy controls, migraineurs have less gray matter volume and thinner cortex of several pain and visual processing regions, and there are negative correlations between the structure of these regions with headache frequency (10,11).Furthermore, amongst those with CM, cortical thickness might correlate with specific clinical characteristics such as duration of time with CM, sleep quality, somatic symptoms and pain self-efficacy (17). CM is also associated with altered local and global structural network topology (18).
Prognostication of patient outcomes and predicting treatment response
A few publications suggest that the development of imaging-based predictive models for headache outcomes is possible. In one such study, right hippocampal volumes measured via MRI were positively associated with good patient outcomes 2 years later (defined as having at least a 50% reduction in headache frequency) (19). After adjusting for several variables that could impact patient outcomes (age, sex, anxiety/depression scores, baseline headache frequency), right hippocampal volume was associated with a 4.7 odds ratio for good outcomes. A longitudinal MRI study demonstrated that gray matter volume changes differed amongst those with EM who had worsening migraine over time versus those that had improved or showed stable migraine patterns (20). In an MRI study of individuals who had CM with medication overuse (MO), baseline orbitofrontal cortex volume predicted the response to various treatments (21). Further work is needed to determine if predictive models are accurate for the individual patient, are valid across similar patient populations, and if they can predict outcomes for individual modes of therapy.
Measuring early treatment response
Although not a study of CM, an investigation of pain-induced brain activations in EM demonstrated changes in activation patterns of the anterior cingulate cortex after 60 days of treatment with an external trigeminal neurostimulator (22). Migraineurs with more frequent migraine attacks had greater or more “hyper-reactive” pre- and post-treatment activations of the anterior cingulate. A MRI functional connectivity study of CM with MO demonstrated improvement in interregional intra-network connectivity of salience and executive networks following a series of treatments with sphenopalatine ganglion blocks (23). Brain imaging biomarkers for treatment response that could be used early after the initiation of treatment, prior to being able to determine response on clinical grounds alone, could help to increase the efficiency of screening new migraine therapies during their development. However, such a brain imaging biomarker is useful only if it measures or predicts treatment response earlier or more accurately than clinical indicators, such as headache frequency or severity.
Migraine classification using imaging data
Imaging data can be used for classification in at least two important ways. One method is useful for determining the specificity of imaging findings for individual headache diagnoses and for objectively testing the validity of subjective diagnostic criteria. With this method, imaging data are obtained from patients with known headache diagnoses, multivariable models of imaging data are developed, and the accuracy of those models for assigning an individual brain MRI (i.e. individual patient) to the correct diagnosis is determined. This approach has been taken in several published studies (24–26). The classification accuracies for migraine, EM, and CM have ranged from about 65% to 95% depending on the study (26–31). The most accurate models are those that classify individuals with more severe migraine burden (i.e. those with higher headache frequency/CM and more years with migraine). The higher classification accuracy for CM compared to EM further supports the observation that CM is associated with greater aberrations in brain structure and function compared to EM, thereby making it easier to classify. A useful imaging biomarker for CM classification would not only differentiate CM from EM and healthy controls, but also from other headache types. Although much more work is needed in this regard, studies have identified differences in brain structure or function between migraine and tension-type headache, cluster headache, and post-traumatic headache (32–37).
Another method of using imaging data for classification purposes is to use “automated” data-driven clustering techniques to identify sub-groups of patients based upon imaging data alone. Once sub-groups are identified, similarities and differences in patient characteristics are examined. This approach could contribute to identification of new headache types and/or migraine sub-types. Automated MRI classification of 66 migraineurs and 54 healthy controls divided the group into two sub-groups based upon measures of brain structure (31). The migraineurs in one sub-group had more severe symptoms of allodynia during migraine attacks, more years with migraine, and greater migraine-related disability, suggesting that these clinical characteristics are associated with differences in brain structure.
Neurophysiology: Functional chronic migraine biomarkers
Several research groups have investigated possible neurophysiological biomarkers of CM. It is worth mentioning that most of the CM studies included patients with medication overuse headache (MOH). Among various possible candidates, electrophysiological markers of central sensitisation and defective central pain control systems are the most promising.
Chronic migraine
In CM, a larger amplitude in cortical responses related to pain has been detected (38,39). The frequent recurrence of pain promotes neural plasticity, and remapping of the pain process over the anterior cingulate cortex has been described in CM (40).
While investigating simultaneous non-painful somatosensory evoked potentials (SSEP) habituation and thalamo-cortical loop activation in CM, researchers have observed a remarkable neurophysiological pattern similar to that of ictal EM. In fact, both groups of patients were characterised by higher initial amplitudes and by diminishing response (habituation) over sequential block averages, resulting in a “transient” central sensitisation. In ictal recordings of CM and EM patients, the decrease in interictal episodic thalamo-cortical activity (reflected by reduced early high-frequency oscillations [HFOs] embedded in the common broad-band SSEP) normalised, whereas the amplitude of the primary cortical component (late HFOs) consistently increased compared with those of healthy controls and interictal EM patients (41). Central sensitisation in CM is not due to a deficit of lateral inhibition in the parietal cortex (42) but a contribution of subcortical inhibitory circuits cannot be excluded (43). These neurophysiological abnormalities of CM are proportional to the duration of the headache chronification phase (44).
Contradictory results were obtained studying the brainstem using the blink reflex response (39,45). Higher initial mean block amplitude followed by a normal response decrement (i.e. habituation) was also observed in recordings of magnetic visual evoked potentials (P100m) in CM patients compared with EM patients recorded interictally (46). This is consistent with the SSEP studies mentioned above (39,43). Using a test of cortical inhibition known as transcranial magnetic suppression of perceptual accuracy, researchers observed that CM patients (with or without MO) exhibited the lowest suppression index compared with controls, with EM falling in between (47,48). Cosentino et al. (49) reported an inhibitory response in CM patients during trains of TMS at 5 Hz, instead of a progressive physiological facilitation. Prolonged inhibition was confirmed recording the transcranial magnetic stimulation (TMS)-induced cortical silent period (CSP) in a group of patients with CM, but most of them were taking prophylactic medication at the time of testing (50).
Migraine evolved in medication overuse headache
In patients with MOH, the initially higher SSEP amplitudes were further increased during stimulus repetition, resulting in a “persistent” sensitisation (51). Nonetheless, patients experiencing cutaneous allodynia exhibited greater SSEP sensitisation compared with those without allodynia (44). As for CM, the neurophysiological abnormalities of MOH are proportional to the duration of the headache chronification phase (51).
Nonetheless, despite the overall lack of habituation in MOH patients, SSEP amplitudes differed according to the drug of overuse, because amplitudes were smaller in triptan overusers than in patients overusing non-steroidal anti-inflammatory drugs (NSAIDs) or both medications combined (51). These abnormalities appear to be influenced by genetic factors (52). Moreover, MOH patients exhibited deficient habituation during contingent negative variation cognitive potentials (53) and laser evoked potentials (LEP) recordings (54).
Exploring inhibitory circuits, Currà et al. (55) measured the CSP in a group of MOH patients. Despite the overall similarity in CSP duration between MOH patients and healthy controls, subgroup analysis revealed that CSP duration was significantly shorter in triptan overusers than in the NSAID or triptan-plus-NSAID overuser groups. In NSAID and triptan-plus-NSAID MOH subgroups, CSP duration correlated positively with monthly tablet intake, while triptan overusers exhibited a negative correlation (55). In a study using trains of motor cortex TMS at 5 Hz, which enhanced motor evoked potential amplitudes in healthy controls and pure CM patients, depressed MEP amplitudes in MOH patients, helping to differentiate these two forms of chronic headache (56).
Perrotta et al. (57) explored spinal cord pain processing by studying the reflex threshold, area, and temporal summation threshold (TST) of the lower limb nociceptive withdrawal reflex in a group of MOH patients before and after drug withdrawal. A significantly lower reflex threshold, higher amplitude, and lower TST were detected in MOH patients before drug withdrawal compared with EM and healthy controls. All of these neurophysiological abnormalities tended to recover to normal values after withdrawal from acute medication overuse (57) in parallel with an elevation in endocannabinoid system activity (58).
In sum, in patients with CM, neurophysiological abnormalities showed a pattern similar to that of ictal episodic migraine; that is, initial sensitisation and delayed habituation. For this reason, CM was previously defined as a condition of “never-ending migraine attack” (59). Electrophysiological responses differ from those of ictal migraine only when the chronicity is due to drug abuse. In patients with MOH, SSEPs suggested a persistent cortical sensitisation; that is, initial sensitisation and delayed lack of habituation. Moreover, the recorded neurophysiological abnormalities appear to vary according to the overused drug.
Prognostication of patient outcomes and predicting treatment response
Although not explicitly performed in CM, some studies observed that fluctuations of several neurophysiological responses depend on the spontaneous clinical fluctuations in attack frequency occurring in the 6 months following the testing session. Between attacks of EM, the degree of intracortical somatosensory inhibition, the activation of the thalamocortical loops and the degree of somatosensory habituation were significantly reduced in patients experiencing a spontaneous worsening of the frequency of the attacks (60–62). In noxious cortical evoked potentials using CO2 laser, researchers observed reduced habituation of the main N2-P2 vertex response after both cephalic and extra-cephalic stimulation in MOH patients who did not improve after preventive treatment (54). However, MOH patients who had clinically improved, habituation was significantly more pronounced after treatment.
Measuring early treatment response
Researchers have used different neurophysiological tools to investigate biomarkers of response to treatment in CM and MOH.
In MOH patients with larger amplitude cortical responses related to pain, acute medication withdrawal and successful therapeutic intervention with onabotulinumtoxinA can both restore normal cortical activation (54,63,64).
In a study involving patients who remitted from CM to EM after preventive treatment with topiramate, the abnormally higher visual evoked magnetic response pattern shifted toward a form of EM interictally (i.e. decreased initial amplitude with subsequent deficient habituation) (65). Similarly, an anesthetic block of bilateral greater occipital nerves as treatment for CM+MOH resulted in the visual and auditory neurophysiological patterns reverting to those characterising interictal EM (i.e. deficient VEP habituation and flat intensity dependence of auditory evoked cortical potentials slope) and, in a subgroup of patients, in parallel with resolution of MO (66). The lowest degree of cortical inhibition detected using TMS in CM normalised after 1-month preventive treatment with topiramate (47).
Migraine classification using neurophysiological data
Neurophysiological data were used for reliably predicting the occurrence of EM attacks. The classification model using resting-state EEGs showed a mean accuracy of 76% for classifying interictal and preictal phases (67). Similarly, steady-state VEPs also detected pre-ictal state in EM with a 73% of accuracy (68). In a recent paper, the classification tasks were performed using a single-channel SSEP signal, and researchers attained a relatively high accuracy of above 88% in migraine ictal or interictal versus healthy controls discrimination, and above 80% in classification of migraine ictal versus migraine interictal states (69).
Although no studies have been done in CM and the number of studies is limited, the integration of new EP-EEG analytical methods to classify preictal, ictal, and interictal neurophysiological responses might be used further in predicting the evolution from episodic to CM, promoting the use of preventive treatment.
Neurochemistry: Biochemical chronic migraine biomarkers
Trigeminovascular system neuropeptides
Migraine is a complex disorder in which changes in the central modulating nociceptive inputs activate the trigeminovascular system. This activation is thought to release a number of vasoactive, potentially pain-producing neuropeptides. Considering that in CM there should be a permanent, or almost permanent, activation of the trigeminovascular system, it is possible that some of these neuropeptides could theoretically become biological and diagnostic markers of CM. Previous attempts to find a biomarker of the prodromal and aura phases have not been successful and sometimes, as in the case of glutamate levels as a potential marker of migraine aura, totally contradictory (71,72). Therefore, these will not be reviewed here.
Sensory neuropeptides
Calcitonin-gene-related peptide (CGRP), substance P, neurokinin A, amylin and cholecystokinin-8 are the vasodilator peptides found in the trigeminal cell bodies of the afferent arm of the system (72,73). To the best of our knowledge, the role of substance P and neurokinin A has not been studied in patients with CM. Substance P, an endothelium-dependent vasodilator, was suggested to be involved in nociceptive transmission. The 13- preprotachykinin that produces substance P may be differentially cleaved to yield a decapeptide with a similar profile of action and localisation in the trigeminal system, neurokinin A. Blockade of substance P, though effective in blocking plasma extravasation, has not demonstrated efficacy in migraine prevention. In addition, studies testing substance P in plasma as a potential migraine biomarker have provided contradictory results (74). Amylin is a 37 amino acid peptide structurally related to CGRP, with vasodilatory and pronociceptive actions (75,76). A very recent study has shown that interictal amylin levels are elevated in peripheral blood in a series of CM patients, suggesting that this peptide could play a role in migraine chronification (77).
The powerful vasodilator CGRP is a very abundant peptide in the trigeminal system and is able to induce migrainous pain when injected in migraine sufferers, which indicates a crucial role in the pathophysiology of migraine pain (78). CGRP seems to play a relevant role in the development of sensitisation of the trigeminal pain pathways, which leads to migraine pain chronification (79). CGRP is the only neurotransmitter reliably shown to be released acutely during the migraine attack (80–82). In addition, peripheral plasma (83,84), saliva (85–87) and cerebrospinal fluid (88–91) CGRP levels have been shown to be increased in interictal, headache-free periods in CM patients as compared to controls without a history of migraine and EM patients (80–83). Current results indicate that interictal concentrations of CGRP in serum from peripheral blood samples and saliva have a good sensitivity and specificity for CM detection (79,80). Increased CGRP levels have been shown to correlate with the response to onabotulinumtoxinA in CM (84,92) and this treatment significantly decreases CGRP levels as measured 1 month after pericranial injections (93). Therefore, interictal CGRP concentration from peripheral blood samples might be a specific and sensitive biomarker in CM in the context of a patient with a compatible clinical history. However, such a role must still be taken with caution. A recent study did not find differences in interictal serum CGRP levels in EM, CM and healthy controls (94). Even though the numbers of CM patients included in this study are lower compared to those which found increased CGRP levels (83,84) and CGRP levels in healthy controls are higher than those found in all previous studies, it is clear that there are a number of clinical and methodological doubts that should be clarified before CGRP levels could be considered a formal CM biomarker. For instance, the very short half-life of the heterogeneous ELISA tests currently in the market might influence results. It is unknown whether increased CGRP levels are the actual consequence of trigeminovascular system activation or if they are a chronic pain or chronic headache marker, or even a marker for a specific comorbidity of CM, though preliminary data indicate CGRP levels do not change in chronic tension-type headache (95), cervicogenic headache (96) or due to fibromyalgia (83,97). In addition, the use of peripheral CGRP as a CM biomarker is limited by the high interpersonal variability. Thirty percent of CM patients have a peripheral level of CGRP that is comparable to those in the healthy controls. It is possible that these patients suffer from other headaches mimicking CM. Alternatively, these data could suggest that CGRP is not the only neuropeptide involved in pain generation and maintenance, and that in some patients it might not be the most relevant.
Parasympathetic neuropeptides
Besides the trigeminal nerve, activation of the cranial efferent parasympathetic arm of the trigemino-vascular system also plays a role in migraine pathophysiology (72). The autonomic nervous system is involved in migraine as reflected by the “general” (nausea, vomiting, polyuria or diarrhea) and “cranial” (conjunctival injection, lacrimation, nasal congestion, etc) symptoms and signs appearing during attacks. A recent study has shown that around 80% of women with CM have at least one cranial autonomic parasympathetic symptom coinciding with pain (98). It has been suggested that parasympathetic outflow to cephalic vasculature may trigger activation and sensitisation of perivascular sensory afferents and thereby contribute to migraine pain chronification (99).
Goadsby and Edvinsson showed, more than 20 years ago, an increase in vasoactive intestinal peptide (VIP) during cluster headache attacks as a reflection of the intense parasympathetic activation (100). Few studies have tested VIP levels in migraine and such information is limited in CM. Interictal serum VIP levels have been shown to be increased in CM, though this increase was less pronounced and consistent as that seen for CGRP (92,101). VIP, but not CGRP, levels correlate with the presence and degree of cranial parasympathetic symptoms as measured by an ad-hoc quantitative scale (102). These data suggest that VIP levels could be an indication of the degree of activation of cranial parasympathetic system in CM patients.
Experimental data support a role for pituitary adenylate-cyclase-activating polypeptide (PACAP) in migraine pathophysiology. Unlike VIP, PACAP localises to both parasympathetic and sensory systems (103), which could explain why PACAP infusion produces a migrainous headache (104). However, data on PACAP levels in migraine patients are scarce and inconsistent. For instance, while PACAP levels were seen to be increasing in jugular samples during acute migraine attacks (105), decreased interictal PACAP levels (as compared to non-headache subjects or tension-type headache patients), which normalise during attacks, have been shown in EM patients (106,107). Contrary to CGRP and VIP; interictal serum levels of PACAP have been shown to be in the range of controls in a large series of CM (108).Therefore, in spite of the evidence suggesting a relevant role of PACAP in the pathophysiology of migraine pain and in its chronification, and in contrast to CGRP and VIP, serum levels of PACAP, as measured peripheral and interictally by ELISA techniques, do not seem to be a useful biomarker in CM, though again current data should be interpreted with caution.
Glial neuropeptides
Glial cells are abundant in the trigeminovascular system and mainly in the trigeminal ganglion. S100B is a calcium-binding protein, found in the cytoplasm of glial cells in the central nervous system, which is released in response to inflammatory stimuli. Previous works analysing S100B in migraineurs have offered contradictory results. The first study in CM has shown S100B levels in the range of healthy controls (109).
Discussion
There is supporting evidence that CM is associated with structural and functional changes in the brain that are measurable using imaging, neurophysiological, and neurochemical techniques. In addition to yielding insights into CM pathophysiology, the quantification, combination, and modeling of these changes can contribute to the development of diagnostic, prognostic, and predictive biomarkers (see Tables 1, 2, 3).
Brain imaging results that could contribute to developing chronic migraine biomarkers.

BOLD: blood oxygen level detection; CM: chronic migraine; CTTH: chronic tension-type headache; EM: episodic migraine; ETTH: episodic tension-type headache; HC: healthy control; TTH: tension-type headache.
Neurophysiology results that could contribute to the development of chronic migraine biomarkers.
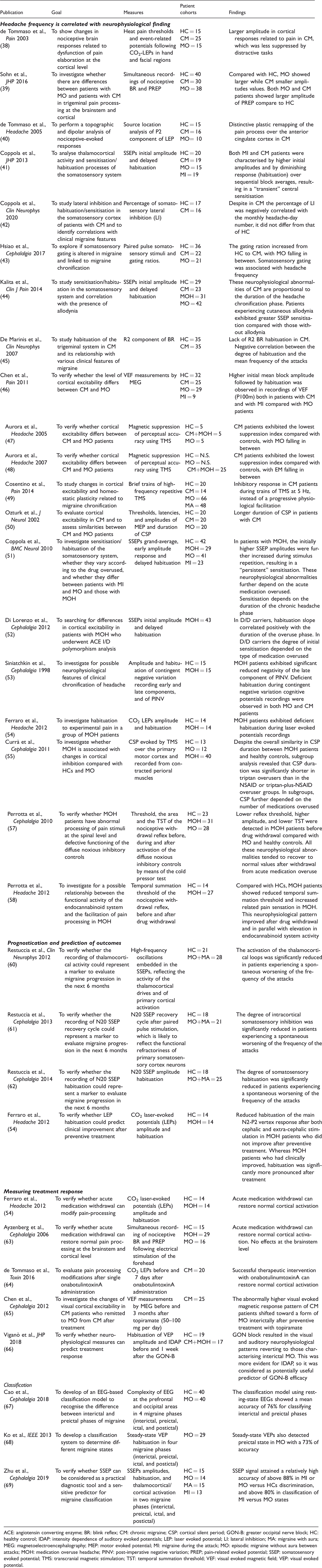
ACE: angiotensin converting enzyme; BR: blink reflex; CM: chronic migraine; CSP: cortical silent period; GON-B: greater occipital nerve block; HC: healthy control; IDAP: intensity dependence of auditory evoked potentials; LEP: laser evoked potential; LI: lateral inhibition; MA: migraine with aura; MEG: magnetoelectroencephalography; MEP: motor evoked potential; MI: migraine during the attack; MO: episodic migraine without aura between attacks; MOH: medication overuse headache; PINV: post-imperative negative variation; PREP; pain-related evoked potential; SSEP: somatosensory evoked potential; TMS: transcranial magnetic stimulation; TST: temporal summation threshold; VEF: visual evoked magnetic field; VEP: visual evoked potential.
Interictal trigeminovascular neuropeptides in different biological fluids that could contribute to the development of chronic migraine biomarkers.
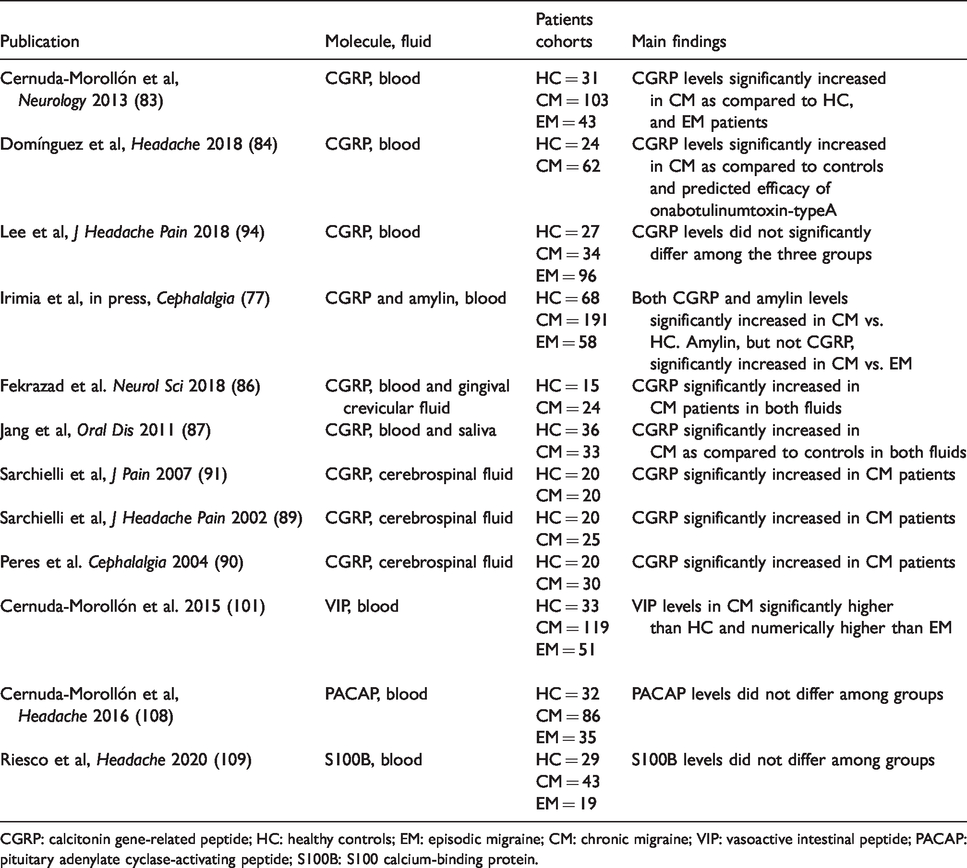
CGRP: calcitonin gene-related peptide; HC: healthy controls; EM: episodic migraine; CM: chronic migraine; VIP: vasoactive intestinal peptide; PACAP: pituitary adenylate cyclase-activating peptide; S100B: S100 calcium-binding protein.
Imaging data have shown promise for contributing to CM biomarkers. However, they need to be optimised and then validated in independent datasets. This should include testing model accuracy on completely independent cohorts and data collected from different scanners at different institutions.
Neurophysiological data have also demonstrated the presence of functional changes in the migraine brain. Normal habituation, central sensitisation, and higher amplitude of non-noxious (SSEP and VEP) and noxious (LEP and PREP) evoked responses may play important roles in the chronic nature of migraine with or without MO.
At a neurochemical level, there is evidence of changes that occur in the brain when migraine becomes chronic. Technical issues with measuring these neuropeptides must be resolved. If resolved, the future might see a practical neurochemical biomarker for migraine.
To optimise the accuracy of CM biomarkers, future models should include multiple data types such as structural and functional brain imaging, results of neurophysiologic studies, genetic profiles, patient demographics, migraine symptoms, neuropeptide levels, medical comorbidities, and treatment responses. However, a clinically useful biomarker needs to not only be highly accurate, but it also must consist of data that are relatively easy and practical to obtain.
In the future, CM biomarkers might include epigenetic and deep clinical phenotyping data alone or in combination with imaging, neurophysiological and biochemical data. Whether these biomarkers will eventually become useful for further defining migraine and for practicing precision medicine will depend upon their ultimate accuracy, their generalisability, and the practicality of collecting the data that contribute to the biomarker. Identification and development of biomarkers with utility to diagnose CM, to prognosticate and evaluate treatment response would greatly advance the field.
Conclusions
Existing studies show feasibility for developing diagnostic, prognostic, and predictive CM biomarkers based on imaging, neurophysiological, and neurochemical data.
Article highlights
Validated chronic migraine biomarkers could improve diagnostic, prognostic, and predictive abilities, as well as increase knowledge on pathophysiology. Structural and functional changes that occur in the brain with the progression from episodic to chronic migraine might be useful for developing biomarkers. Chronic migraine biomarkers would help us move closer to practising precision medicine and improve patient care.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PPR has received honoraria for participation in clinical trials and contribution to advisory boards or presentations from Allergan, Almirall, Amgen, Biohaven, Chiesi, Electrocore, Eli Lilly, Medscape, Novartis, and Teva. Financial support for research projects was provided by Allergan and Novartis. Headache research is supported by la Caixa Foundation, MINECO, AGAUR, Fundacion La Marato TV3, Instituto Investigacion Carlos III, Migraine Research Foundation, Mutual Medica, and PERIS. She serves as an Associate Editor for Cephalalgia, Frontiers in Neurology (Headache Section), Editor for Revista de Neurologia, and on the Advisory Board of The Journal of Headache and Pain.
GC has received contribution to advisory boards or presentations from Novartis, Teva, and Eli Lilly. He serves as an Associate Editor for Cephalalgia, BMC Neurology – Pain section, and on the Advisory Board of The Journal of Headache and Pain.
JP has served on the Advisory Boards of Allergan, Lilly and Novartis-Amgen. Financial support for research projects was provided for Instituto de Investigación Carlos III. He serves as an Associate Editor for Headache.
TJS receives research support from the American Migraine Foundation, Henry Jackson Foundation, National Institutes of Health, Patient-Centered Outcomes Research Institute, US Department of Defense, and Amgen. Within the past 12 months, he has received personal compensation for serving as a consultant or advisory board member for Alder, Allergan, Amgen, Biohaven, Cipla, Click Therapeutics, Eli Lilly, Equinox, Weber & Weber, and XoC Pharmaceuticals. He holds stock options in Aural Analytics and Nocira. He serves as an Associate Editor for Headache and Cephalalgia.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author contributions
All authors contributed equally to the draft and final version of the paper.
