Abstract
Migraine patients often complain of sleepiness, a problem that manifests both during and outside an attack, may impair the quality of life and can lead to potentially harmful situations. Findings from an uncontrolled study suggest that a high percentage of migraineurs experience excessive daytime sleepiness (EDS). We investigated EDS in a case-control study on 100 patients with episodic migraine and 100 age- and sex-matched healthy controls and also assessed sleep quality, anxiety and depression. Although it was found that EDS was more frequent in migraineurs than in controls (14% vs. 5%; odds ratio 3.1; 95% confidence interval 1.1–8.9), the frequency was lower than previously reported. EDS correlated with migraine disability, sleep problems and anxiety. EDS in patients with migraine probably stems from the full constellation of headache-sleep-affective symptoms resulting from the complex clinical burden of the disease.
Introduction
Excessive daytime sleepiness (EDS), defined as difficulty in maintaining a desired level of wakefulness (1), can be a disabling symptom. Besides impairing a person's quality of life, it may also lead to potentially harmful situations such as loss of attention and sleep attacks. The prevalence of EDS in the general population ranges from 10% to 20% and increases in the young and very old (2, 3). EDS is generally attributed to poor sleep quality, depression, anxiety, medications, sleep disordered breathing, obesity and metabolic syndrome (4).
Considerable attention has focused in recent years on the problem of EDS in several common neurological diseases such as Parkinson's disease, epilepsy and, more recently, migraine (5–7). Whether EDS in migraine arises from migraine itself, migraine treatment or both is unclear. Nearly one-quarter of patients with migraine complain of sleepiness during various phases of a migraine attack including the pain and postdrome phases (8, 9). In addition, patients may complain of sleepiness after taking drugs used for acute therapy, such as indomethacin and triptans (10), as well as those for preventive treatment (e.g. flunarizine, antiepileptics and amitriptyline) (11). Sleepiness is therefore a potential source of disability not only during migraine attacks but also at times when patients are headache free.
In an uncontrolled study including patients with episodic and chronic migraine, Peres et al. found that a high percentage of migraineurs reported experiencing EDS (7). This observation prompted us to investigate EDS in a case–control study in a homogeneous group of patients, all of whom had episodic migraine. To determine whether EDS is a primary phenomenon or arises from other factors, concomitant diseases, including anxiety and depression, use of medications and sleep quality were investigated.
Methods
A case–control study was conducted, designed to compare patients who had episodic migraine (12) consecutively recruited at our Headache Clinic from 1 January to 30 June 2006 and population healthy controls paired for sex and age (±1 year), consecutively recruited in the same period from non-consanguineous relatives of out-patients attending our neurological unit.
All participants underwent a complete physical and neurological examination. Patients were screened for factors known to interfere with the sleep–awake cycle, such as use of coffee or alcohol, smoking, type of work, body mass index (BMI), restless legs syndrome (RLS) (13), medication, anxiety or depression, or both, and concomitant cardiovascular, pulmonary or urological diseases.
Detailed information on the clinical features of migraine (disease duration, family history of migraine, presence of aura, presence of sleepiness as a prodromal, accompanying or postdromal symptom, frequency and duration of attacks, location, quality and intensity of pain, acute or preventive treatment or both, and concomitant headaches) was gathered with face-to-face interviews using structured questionnaires (14). Migraine disability was assessed using the Migraine Disability Assessment Scale (MIDAS) (15). All patients were assessed at least 5 days after the last migraine attack.
To measure EDS we applied the Epworth Sleepiness Scale (ESS), a simple, self-administered questionnaire which correlates with sleep latency as measured during the multiple sleep latency test and overnight polysomnography (16). EDS was defined as an ESS score ≥10 (16). The quality of sleep was measured using the Pittsburgh Sleep Quality Index (PSQI) (17), a self-rated questionnaire scored on a 0–3 scale assessing various factors related to sleep quality, including estimates of sleep duration and latency, frequency and severity of specific sleep-related problems, then grouped into seven component scores. An overall score of > 5 distinguishes poor (PSQI > 5) from good (PSQI < 5) sleepers.
Symptoms of depression were rated with the Beck Depression Inventory (BDI) and anxiety was rated with the State-Trait Anxiety Inventory (STAI-1 and STAI-2) questionnaires.
The χ2 test was used to analyse differences in the frequency of categorical variables. Differences in the means of continuous measurements were determined with Student's t-test and checked by the Mann–Whitney U-test. To determine the relationships between the studied variables, Pearson and Spearman correlation coefficients were calculated. A logistic-regression model was run to evaluate the relation between ESS and episodic migraine adjusted for gender, PSQI and medications, and odds ratios (ORs) with relative 95% confidence intervals (CIs) were estimated. Values are expressed as mean ± SD. P-values of < 0.05 were considered to be statistically significant. All statistical analyses were performed with the statistical package SPSS (version 13.0; SPSS Inc., Chicago, IL, USA).
Results
We studied 100 migraine patients (M/F: 16/84; mean age 36.9 ± 12.1 years; migraine without aura 86, migraine with aura: 8, migraine without and with aura: 6) and 100 healthy controls (M/F: 16/84; mean age 36.7 ± 11.7 years).
There were no differences in smoking, or coffee or alcohol intake between patients and controls. More controls than patients reported working on nightshifts (15.9% vs. 6.1%; P < 0.05). No difference emerged in BMI, cardiovascular, pulmonary or urological diseases between the two groups. Conversely, anxiety and depression, as well as the use of anxiolytics and antidepressants, were more common in patients than in controls (Table 1). RLS was reported by six patients, but by none of the controls.
Depression, anxiety and use of antidepressants/anxiolytics in patients and controls
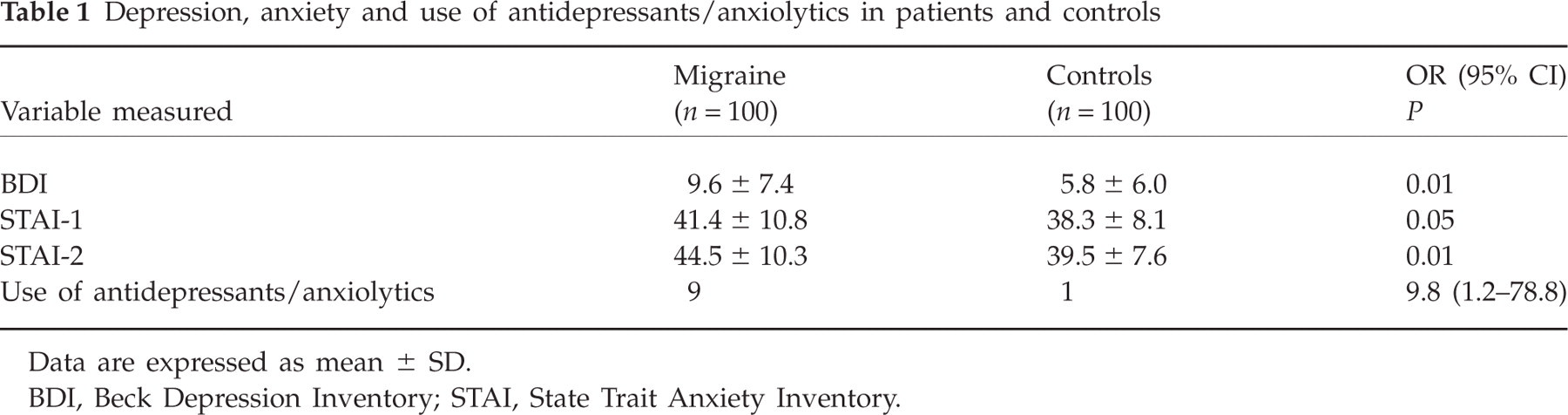
Data are expressed as mean ± SD.
BDI, Beck Depression Inventory; STAI, State Trait Anxiety Inventory.
Among the 100 patients, migraine duration was 18.9 ± 11.6 years, attack frequency 5.1 ± 4.6/month and duration of attacks was < 24 h in 46 patients, 24–48 h in 37 patients and > 48 h in 17 patients. Pain intensity was rated as mild in four patients, moderate in 37 and severe in 59. The MIDAS score was 23.3 ± 27.3. Nine patients also had episodic tension-type headache. Of the 100 patients studied, 41 complained of sleepiness during migraine attacks. Sleepiness manifested at various phases: in 10 patients during the prodrome, in 13 during the pain phase, in 11 during the resolution and in seven patients during all three phases. No clinical or demographic differences emerged between patients with and those without sleepiness during the attack. According to the questionnaire, all 100 patients studied took medications for acute treatment of the attack: 60 patients usually took non-steroidal anti-inflammatory drugs, 36 triptans and four opioids. Of the 100 patients studied, only 21 were currently receiving migraine prophylaxis (seven with amitriptyline, eight with flunarizine and six with anticonvulsants). One patient took antiemetics (metoclopramide) during the attack.
There were no significant differences in mean ESS scores between patients and controls (Table 2). In contrast, pathological ESS scores (≥ 10) were more frequent in migraineurs than in controls (Table 2). The overall PSQI score was significantly higher in patients than in controls (P < 0.01) and PSQI scores ≥5 were more frequent in migraineurs than in controls (Table 2). PSQI subscale analysis showed that patients had significantly higher scores than controls for the items regarding sleep quality, sleep efficiency and sleep disturbances (component scores 1, 4 and 5) (P < 0.01). Logistic regression analysis confirmed that ESS scores for gender, PSQI and antidepressant/anxiolytic drugs were higher in patients than in controls (Table 3).
Daytime sleepiness and sleep quality in patients and controls

Data are expressed as means ± SD.
ESS, Epworth Sleepiness Scale; ESS ≥ 10, excessive daytime sleepiness (EDS); PSQI, Pittsburgh Sleep Quality Index; PSQI ≥ 5, bad sleeper.
Logistic regression analysis to evaluate the relationship between Epworth Sleepiness Scale (ESS) and episodic migraine adjusted for gender, Pittsburgh Sleep Quality Index (PSQI) and use of antidepressant/anxiolytic drugs

ESS ≥ 10, Excessive daytime sleepiness (EDS); PSQI ≥ 5, bad sleeper.
Patients who reported experiencing sleepiness during the attack had lower PSQI scores than those without, but no difference was found between the two groups for ESS, alcohol, smoking, coffee, RLS, workshift, clinical features of migraine, symptomatic and prophylactic therapies, or other concomitant diseases or medications.
In the analysis investigating the relationships of ESS and PSQI scores with clinical variables among patients, the ESS score was directly correlated with MIDAS (P < 0.05), PSQI (P < 0.05), STAI-1 and STAI-2 scores (P < 0.05) and inversely with age (P < 0.05). The PSQI score correlated with RLS (P < 0.01), headache frequency (P < 0.05), MIDAS (P < 0.001), STAI-1, STAI-2 and BDI scores (P < 0.001).
Discussion
In our case–control study in a diagnostically homogeneous group of patients, it was found that although patients with episodic migraine and healthy, age- and gender-matched controls had similar mean ESS scores, pathological ESS scores (≥ 10) were more frequent in patients than in controls (14% vs. 5%). Hence, we have confirmed in a case–control study the increased prevalence of EDS in migraine described by Peres et al. in an uncontrolled study (7). A distinctive finding of our study was the relatively low frequency of EDS in migraine patients (14% vs. the reported 36.3% in episodic and 55.1% in chronic migraine) (7), implying that EDS has a less important impact in episodic migraine than others have suggested (7). This discrepancy probably arose because we included a control group, studied a larger number of patients with migraine (100 vs. 72) and investigated concomitant diseases, including anxiety and depression, therapeutic drugs and sleep quality, factors which are known to contribute to the pathogenesis of EDS.
Assessing the quality of sleep and the presence of anxiety and depression proved useful, because it again confirmed that EDS is a complex problem profoundly influenced by affective disorders and alterations in the sleep–wake cycle (3). As expected, our patients' ESS scores correlated not only with migraine disability, as rated by MIDAS, but also with sleep problems (56% of patients reported being bad sleepers) and anxiety (49% had pathological scores on the STAI-1 and 54% on the STAI-2 questionnaires). The high scores for anxiety and depression found in our patients confirm the need to investigate the presence of these disorders in patients with migraine (18). Interestingly, some studies have reported an even greater relationship between migraine and anxiety than between migraine and depression (19, 20). Also, the high frequency of bad sleepers among the patients with migraine is in keeping with the notion that migraine is a chronobiological disorder characterized by a striking association with sleep and other circadian biorhythms. Migraine attacks are more likely to occur during periods of rapid eye movement (REM) sleep with morning arousals associated with larger amounts of stages II, III and REM sleep. Parasomnias, such as somnambulism and night terrors, are more frequent in migraineurs than in healthy controls (21, 22).
Our EDS results and, most important, the lack of correlation between ESS scores and the presence or absence of sleepiness during the attack weaken the hypothesis that the hypothalamus is the mediator of EDS in migraineurs (7). Our data are therefore in keeping with the idea that rather than being mediated by the hypothalamus, EDS is probably a consequence of the complex clinical burden of the disease (migraine disability, anxiety, sleep problems) stemming from the full headache–sleep–affective symptom constellation. The idea that the hypothalamus might be involved in migraine derives from the observation that the cerebral circuits involved in adaptative homeostatic mechanisms are altered in migraine (23). The hypothalamus is wired to rostral and caudal areas involved in sleep mechanisms and pain control and probably represents the neural site responsible for the chronobiological features of some headaches, including migraine (23). Other elements pointing to a role of the hypothalamus in the pathophysiology of migraine are the circadian rhythm of migraine attacks that peak in the early morning, the quality of some prodromal, accompanying and postdromal symptoms (sleepiness, thirst, food craving) and the abnormal pattern of hypothalamic hormonal secretion reported in chronic form (23).
A possible limitation of this study is the fact that we considered the major risk factors for EDS (4) without taking into account other variables which can interfere with the sleep–awake cycle such as menstruation, premenstrual syndrome and premenstrual dysphoric disorder. In addition, the assessment of sleep and EDS was purely clinical. In the large study sample, EDS was assessed with the ESS, a quick, inexpensive, easy, self-administered questionnaire that correlates with the more time-consuming multiple sleep latency test and overnight polysomnography (24). To evaluate sleep the PSQI was used, a simple test that discriminates healthy subjects from depressed patients and patients with sleep disorders, is sensitive to the effect of treatments, may be used in general medical disorders and correlates with quality-of-life questionnaires (25–28).
Notwithstanding the lack of neurophysiological testing, including multiple sleep latency test and overnight polysomnography, our clinical data obtained with the ESS and PSQI should help in assessing the importance of EDS in patients with episodic migraine. Whether these findings can be generalized to patients with chronic migraine awaits the results of future studies.
