Abstract
The aim of this study was to investigate the effect of painful conditioning stimuli on pain and blink reflexes to supraorbital nerve stimulation. Electromyograph activity was recorded bilaterally from the orbicularis oculi muscles in 13 normal participants in response to low (2.3 mA) and high-intensity (18.6 mA) electrical stimulation of the left supraorbital nerve before, during and after the application of ice to the left or right temple or immersion of the left hand in ice-water for 60 s. The pain evoked by the high-intensity electrical stimulus was greater during painful conditioning stimulation of the ipsilateral temple than during the recovery period afterwards, and was greater than during painful conditioning stimulation of the contralateral temple. These findings imply that spatial summation of nociceptive signals across different divisions of the trigeminal nerve can heighten pain. However, painful conditioning stimulation, particularly to the right temple, strongly suppressed the R2 component of the blink reflex to the low-intensity stimulus, and also suppressed R2 to the high-intensity stimulus. Thus, an inhibitory influence (e.g. diffuse noxious inhibitory controls) appeared to mask ipsilateral segmental facilitation of R2 during ice-induced headache. This finding contrasts with recent electrophysiological evidence of trigeminal sensitization in migraine.
Introduction
The blink evoked by periorbital cutaneous stimulation is considered to be a protective reflex with several components. The initial ipsilateral (R1) component has a brief latency and is mediated in the pons by fibres that project from the principal sensory nucleus of the trigeminal nerve to the ipsilateral facial nucleus via one or two interneurones. The second bilateral (R2) component involves a multisynaptic medullary pathway that descends to the caudal part of the spinal trigeminal nucleus and adjacent reticular formation before ascending to the ipsilateral and contralateral facial nuclei (1). Painful infrared laser stimulation of the face evokes a bilateral blink reflex at a latency corresponding to R2, but does not evoke an early ipsilateral component (2). Since painful pulses of heat emitted from CO2 lasers selectively activate nociceptive fibres (3), Aδ fibres apparently form part of the afferent limb of R2 but not R1. The third late (R3) component habituates rapidly and is suppressed when the stimulus is announced; thus, it may form part of a startle response (4–6).
Both noxious and innocuous stimuli provoke the blink reflex, probably due to convergence of nociceptive and non-nociceptive signals on wide dynamic range neurones in brain-stem trigeminal nuclei. Noxious stimulation anywhere in the body inhibits activity in spinal and trigeminal wide dynamic range neurones through a process of ‘diffuse noxious inhibitory controls’ (DNIC) (7). DNIC originates in neurones in the caudal medulla that appear to have ‘whole-body’ receptive fields for noxious stimulation, and that project to trigeminal nuclei and to the dorsal horn at all levels of the spinal cord. DNIC may improve the signal-to-noise ratio of nociceptive signals emanating from wide dynamic range neurones by inhibiting background activity induced by innocuous stimulation or by mild pain in another part of the body (7–10). Noxious stimulation of the limbs inhibits the second component of the blink reflex. For example, Pantaleo et al. (11) reported that R2 amplitude to supraorbital nerve stimulation decreased during ischaemic arm pain and persisted at low levels for 15 min afterwards. Similarly, Ellrich and Treede (12) found that painful thermal stimulation of the forearm inhibited R2 to innocuous stimulation of the supraorbital nerve.
Despite inhibitory influences such as DNIC, stimuli applied ipsilaterally near to or within the same dermatomal segment as a primary nociceptive stimulus can facilitate nociceptive reflexes. For example, Willer and Albe-Fessard (13) reported that painful electrical stimuli applied to the sural nerve in the ankle facilitated the RIII flexion reflex to a sural nerve stimulus in the calf. Painless radiant heat stimuli applied to the sural nerve territory in the foot also facilitated the RIII flexion reflex to electrical stimulation of the sural nerve in the ankle (14), presumably due to convergence of nociceptive and non-nociceptive signals on spinal neurones that mediate the reflex. Ellrich et al. (15) investigated the convergence of nociceptive and non-nociceptive signals in trigeminal pathways. The conditioning stimulus was a 300-ms pulse of radiant heat applied to the forehead that evoked a painful pricking sensation but not a blink reflex. The test stimulus was a 0.2-ms square wave electrical pulse applied to the supraorbital nerve at an intensity below the pain threshold but that still provoked a blink reflex. The interstimulus interval was adjusted so that subjects perceived only one sensation when they received both forms of stimulation. This was successful for about one-third of trials. During these trials, R2 was> 30% greater than during trials where the electrical stimulus was presented alone.
Although brief conditioning stimuli provoke ipsilateral segmental facilitation of withdrawal and blink reflexes, broad inhibitory influences may mask the facilitatory effects of more persistent noxious conditioning stimulation. For example, recruitment of an inhibitory mechanism (probably DNIC, see 16) appears to mask spatial summation of pain during protracted thermal stimulation of progressively larger segments of the arm (17).
The aim of the present study was to investigate the effect of persistent painful conditioning stimuli applied to the temples (thus inducing a brief headache) and the hand on pain and the blink reflex evoked by supraorbital nerve stimulation. The temple is innervated by the temporal branch of the zygomatic nerve (maxillary division of the trigeminal nerve) and by the superficial temporal branches of the auriculotemporal nerve (mandibular division of the trigeminal nerve). Low threshold mechanoreceptive neurones within brain-stem trigeminal nuclei have small receptive fields limited to one trigeminal division; however, nociceptive neurones within the trigeminal nucleus caudalis and the subnucleus interpolaris generally have large receptive fields, and many of these neurones respond to a wide range of stimulus intensities from more than one trigeminal division (18, 19). Since the supraorbital nerve extends only as far as the superior temporal line (20, 21), a noxious stimulus applied to the temple should not sensitize primary sensory afferents in the supraorbital nerve (a possible outcome of prolonged noxious stimulation within the supraorbital nerve distribution). Instead, facilitation of pain and the blink reflex to supraorbital nerve stimulation by a conditioning stimulus applied to the ipsilateral temple would be consistent with summation of signals converging on wide dynamic range neurones in brain-stem trigeminal nuclei. However, this facilitation might be superimposed on an inhibitory effect, mediated by DNIC. To delineate the extent of this inhibitory influence, in some trials ice was applied to the contralateral temple and the hand during supraorbital nerve stimulation.
Methods
Subjects
The sample consisted of five males and eight females aged between 18 and 29 years (mean age 23 years). Each subject provided informed consent prior to participating in the study, which was approved by the Murdoch University Human Research Ethics Committee.
Blink reflexes
Electromyograph (EMG) signals were recorded bilaterally from the orbicularis oculi muscles with surface electrodes attached to the mid-lower eyelids. Reference electrodes were attached over the outer part of each orbit, and a ground electrode was attached to the wrist. Blink reflexes were elicited by electrical stimulation of the left supraorbital nerve delivered via 5 mm diameter surface electrodes. The cathode was placed over the supraorbital foramen and the anode was placed approximately 2 cm rostrally on the forehead. The stimuli were monopolar square wave pulses (pulse width 0.3 ms, 2.3 ± 0.3 mA or 18.6 ± 0.3 mA) delivered at an interstimulus interval of approximately 10 s to minimize habituation. These stimulus intensities were selected on the basis of pilot studies that indicated that the 18.6-mA stimulus evoked painful sensations whereas the 2.3-mA stimulus did not.
EMG signals were amplified by biopotential amplifiers (Grass Instrument Co., Quincy, MA, USA), and were sampled at 1000 Hz and stored for later off-line analysis on a personal computer via an MP100 Biopac Systems Analogue/Digital Channel Receptor and AcqKnowledge software (Biopac Systems, Goleta, CA, USA). Signals were rectified and integrated prior to analysis of the R2 component of the blink reflex. The area under the curve (AUC) was assessed in the time window 27–87 ms after the stimulus (12).
Procedure
The electrical stimuli were applied in four series, each 14 min long, separated by rest periods of 5 min. The timing of electrical stimulation was the same in each series. In the first two series (the baseline), electrical stimuli were presented while the subject sat quietly. In the third and fourth series, electrical stimuli were presented during and after cold-pain stimulation (Fig. 1). The sequence began by applying an ice block (contact area 3 × 4 cm) to the left (six subjects) or right temple (seven subjects) lateral to the orbit for 60 s. In pilot studies on 10 participants, pain quickly increased during the first 30–40 s of stimulation, then persisted at moderately high levels during the remaining period of stimulation. Therefore, electrical stimuli were administered after 45 s and 55 s of cold stimulation when pain was expected to peak. Blink reflexes and pain ratings to the low (2.3 mA) and high (18.6 mA) intensity stimuli were obtained during each cold stimulus, and during the 60-s recovery period after each cold stimulus. In six subjects the low-intensity stimulus was presented first, whereas in the other seven subjects the high-intensity stimulus was presented first. After 60 s of recovery, the ice block was applied to the temple again for 60 s. Two minutes later the subject immersed the fingers and palm of their left hand to the level of the thumb in ice-water at 2 °C for 60 s to investigate heterotopic effects of cold stimulation on the blink reflex. After a second immersion of the hand, the cycle was repeated for the temple not previously stimulated with ice (Fig. 1).

Experimental sequence. Electrical stimuli were applied in four series, each 14 min long and separated by rest periods of 5 min. (a) In the third and fourth series, low- and high-intensity electrical stimuli were applied during and after 60-s blocks of cold stimulation (represented by the black rectangles). (b) An exploded view of the sequence of electrical stimuli during two consecutive minutes (the first without and the second with concurrent cold stimulation). Electrical stimuli were administered at the 45th and 55th seconds of each minute.
Subjects rated cold-pain at 20, 40 and 60 s, and rated the intensity of each electrical stimulus on numerical rating scales with descriptors of ‘no sensation’ or a ‘painless sensation’ (coded as 0), ‘mild pain’ (1–2), ‘moderate pain’ (3–5), ‘somewhat severe pain’ (6–7), ‘severe pain’ (8), and ‘extremely severe pain’ (9).
Data reduction and statistical analysis
The mean R2 AUC was calculated separately for the 12 low-intensity (2.3 mA) stimuli and the 12 high-intensity (18.6 mA) stimuli administered during the two baseline series. In the third and fourth series, four low- and four high-intensity stimuli were administered during cold stimulation of the hand and the left and right temples; in addition, another four low- and four high-intensity stimuli were administered after cold stimulation of each site. The mean AUC to each set of four stimuli was expressed as a percentage of the mean response during the two baseline series. Electrical-pain ratings were averaged across the 12 stimuli in the baseline series, and across the four stimuli in each set during and after cold stimulation. Ratings of cold-pain were averaged at each time point (20, 40 and 60 s) across the four trials at each site.
To simplify the interpretation of findings, the effects of low- and high-intensity electrical stimulation were investigated separately in statistical analyses. Electrical-pain ratings were investigated in a Cold-stimulation (during, recovery) × Site (ice on the left temple, ice on the right temple) analysis of variance. Analyses that investigated changes in the R2 AUC contained an additional factor of Laterality (the ipsilateral vs. the contralateral component of the blink reflex). Since immersion of the hand in ice-water induced more pain than application of ice to the temples, results for the hand are reported separately. Data are presented as the mean ± SE.
Results
Pain ratings
Cold-pain increased rapidly during application of ice to the temples and remained in the ‘moderately painful’ range during the final 20 s of stimulation (mean pain rating 4.1 ± 0.4). Pain disappeared almost completely during the first 20 s of the recovery period. Cold-pain increased equally during stimulation of the left and right temples (mean pain ratings 4.0 ± 0.5 and 4.1 ± 0.5, respectively). Pain was rated as ‘somewhat severe’ when the hand was immersed in ice-water (mean rating after 60 s of immersion 6.2 ± 0.4).
During the two baseline series, only two of the 13 subjects ever described the 2.3-mA stimulus as painful (mean ratings 0.13 and 0.04, respectively, indicating that the pain was negligible). Similarly, during cold stimulation only one subject described the 2.3-mA stimulus as painful (the mean rating for this subject was 0.25). Ratings to the 18.6-mA stimulus averaged 3.1 ± 0.6 during the two baseline series (i.e. moderately painful). Ratings to the 18.6-mA stimulus increased slightly when ice was applied to the left temple (i.e. the side that was stimulated electrically). However, as shown in Fig. 2, pain ratings to the high-intensity stimulus fell during the recovery period after cold stimulation, and were lower during cold stimulation of the right temple than the left (Cold-stimulation × left vs. Right temple interaction, F(1,12) = 7.6, P < 0.05). Pain ratings also fell slightly during and after immersion of the hand in ice-water (mean decrease 0.31 ± 0.19 and 0.25 ± 0.18, respectively; difference not significant). Exploratory analyses indicated that the pain evoked by the high-intensity (left-sided) stimulus was greater when ice was applied to the left temple than when the hand was immersed in ice-water [t(12) = 3.83, P < 0.01]. In contrast, the effect on shock-induced pain of applying ice to the right temple did not differ from the effect of immersing the hand in ice-water.

Pain ratings to 18.6-mA shocks during and after cold stimulation. Pain ratings to high-intensity shocks were greater during cold stimulation of the left temple than during cold stimulation of the right temple [∗t(12) = 2.18, P < 0.05]. In addition, pain ratings to high-intensity shocks were greater during cold stimulation of the left temple than during the recovery period [∗t(12) = 2.62, P < 0.05]. Error bars represent standard errors, ▪, Ice on the temple; □, recovery.
Blink reflex
The R2 AUC to the 2.3-mA stimulus decreased substantially during and after cold stimulation of the temples compared with responses at baseline (mean decrease 29.4 ± 6.0%, F(1,12) = 24.4, P < 0.001). As shown in Fig. 3a, decreases in R2 AUC were greatest when ice was applied to the right temple (Cold-stimulation × left vs. Right temple interaction, F(1,12) = 6.12, P < 0.05). The R2 AUC also decreased during and after immersion of the hand in ice-water (mean decrease 25.9 ± 8.1%, F(1,12) = 10.1, P < 0.01). Decreases were symmetrical and did not differ between the immersion and recovery periods.
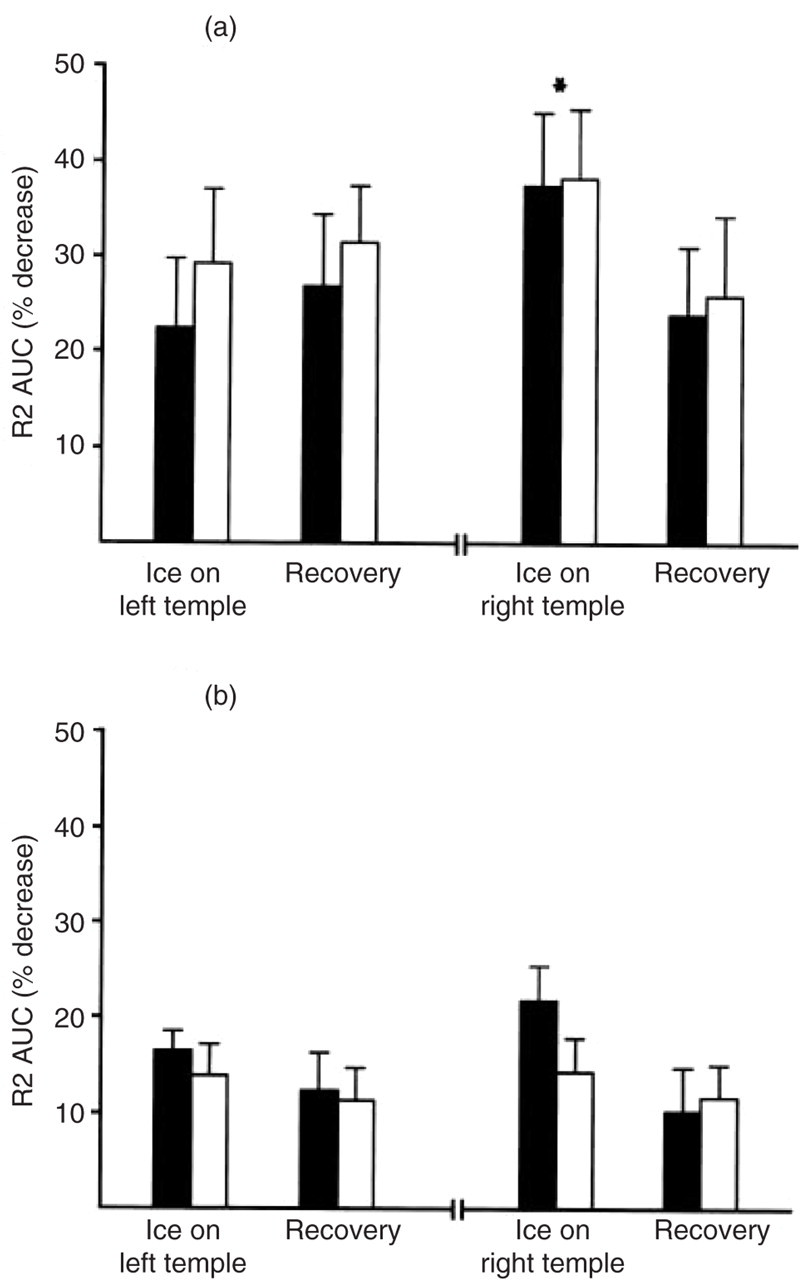
Decreases in the area under the curve (AUC) of the R2 component of the blink reflex during and after cold stimulation. (a) The R2 AUC to low-intensity shocks decreased to a greater extent during cold stimulation of the right temple (contralateral to supraorbital nerve stimulation) than during the recovery period (∗t(12) = 2.37, P < 0.05); in contrast, the decrease in the AUC during cold stimulation of the left temple did not differ from the decrease during the recovery period. (b) Decreases in R2 AUC to high-intensity shocks were greater during cold stimulation than during the recovery period afterwards. Decreases were similar when ice was applied to the left and right temples. Error bars represent standard errors. ▪, Ipsilateral R2; □, contralateral R2.
The R2 AUC to the 18.6-mA stimulus was lower than baseline during cold stimulation of the temples and during the recovery period (mean decrease 14.0 ± 2.3%, F(1,12) = 37.4, P < 0.001) (Fig. 3b). In addition, the AUC was smaller during cold stimulation than during the recovery period (mean decrease below baseline 16.6 ± 2.2% vs. 11.4 ± 2.9%, F(1,12) = 4.92, P < 0.05). Although decreases in the AUC were greatest when ice was applied to the right temple, this effect did not achieve statistical significance (Cold-stimulation × left vs. Right remple interaction, F(1,12) = 2.72, not significant). The R2 AUC decreased 13.8 ± 2.5% during and after immersion of the hand in ice-water (F(1,12) = 30.9, P < 0.001). Decreases were symmetrical and did not differ between the immersion and recovery periods.
The mean decrease in R2 AUC (averaged over the temples and hand during and after cold stimulation) was greater for the low- than the high-intensity supraorbital stimulus (28.3 ± 6.4% vs. 13.9 ± 2.0%, t(12) = 2.21, P < 0.05).
Discussion
It was hypothesized that painful stimulation of the temple ipsilateral to supraorbital nerve stimulation would facilitate electrically evoked pain and R2 due to convergence of nociceptive signals in the brain-stem trigeminal nucleus. However, painful stimulation of the temple contralateral to supraorbital nerve stimulation and immersion of the hand in ice water should inhibit pain and R2 evoked by the supraorbital nerve stimulus due to DNIC. The present findings only partly support these hypotheses. In particular, the facilitatory effect of ipsilateral conditioning stimulation outweighed inhibitory influences on pain, whereas inhibitory influences predominated for R2.
Consistent with expectations, the pain evoked by strong electric shocks to the supraorbital nerve was greater during painful conditioning stimulation of the ipsilateral temple than during the recovery period afterwards, and was greater than during painful conditioning stimulation of the contralateral temple or hand. These effects were small, possibly because of central interaction between input from different modalities recruited by the electrical stimulus. For example, activation of low threshold mechanoreceptive neurones that inhibit central nociceptive transmission segmentally (22–24) might have reduced spatial summation of nociceptive signals. In addition, activation of nociception-specific neurones that do not respond to DNIC (25) could have masked the inhibitory influence of contralateral conditioning stimulation. Despite such influences, the findings imply that spatial summation of nociceptive signals across different divisions of the trigeminal nerve overshadowed counter-irritation effects on pain.
The R2 component of the blink reflex was strongly suppressed during and after painful conditioning stimulation. General inhibitory mechanisms such as DNIC appear to suppress R2 during painful stimulation of the limbs (11, 12), and probably account for the attenuation of R2 during and after painful conditioning stimulation of the hand and contralateral temple in the present study. The attenuation of R2 during and after painful conditioning stimulation of the ipsilateral temple suggests that DNIC may also operate across different divisions of the trigeminal nerve. It is interesting to note that R2 suppression was greater for low- than high-intensity supraorbital nerve stimulation. This finding is consistent with the view that the major role of DNIC is to suppress background activity elicited by innocuous stimuli in wide dynamic range neurones (7).
If noxious and innocuous signals within the trigeminal nerve territory converge upon wide dynamic range neurones in the brain-stem (15, 18, 19), noxious stimulation of the ipsilateral temple should facilitate the R2 component of the blink reflex to innocuous stimulation of the supraorbital nerve. The dominant influence of the conditioning stimulation on R2 clearly was inhibitory. This inhibitory effect was greater during painful stimulation of the right temple (contralateral to the supraorbital nerve stimulus) than during the recovery period immediately afterwards. In contrast, the inhibitory effect did not intensify during painful stimulation of the left temple. One interpretation of this finding is that a local facilitatory effect induced by the conditioning stimulus competed with general inhibitory influences on R2. In support of this interpretation, Ellrich et al. (15) found that a brief painful pulse of heat to the forehead clearly facilitated R2 to innocuous supraorbital nerve stimulation. Presumably facilitation was greater in Ellrich's study than in the present experiment because convergence was greater within than across divisions of the trigeminal nerve; in addition, the conditioning stimulus employed in the present study probably mobilized inhibitory influences more effectively than the brief pulse of heat used by Ellrich et al.
The site of painful conditioning stimulation did not influence the extent of R2 suppression to the high-intensity stimulus. The painful quality of the high-intensity stimulus indicates that it activated a wider range of trigeminal afferents than the low-intensity stimulus. Possibly ‘gate control’ effects (22–24) due to the simultaneous activation of low- and high-threshold sensory afferents in the supraorbital nerve masked any facilitatory influence of conditioning stimulation on R2. Participation of nociception-specific neurones in encoding painful sensations, or convergence of nociceptive signals in the thalamus or cortex, might account for the different effects of painful conditioning stimulation on shock-induced pain and R2. In any event, the dissociation between the effects of the conditioning stimuli on shock-induced pain and R2 suggests that R2 does not by itself provide a simple electrophysiological index of trigeminally mediated pain.
Competition between segmental facilitation and broader inhibitory influences on trigeminal nerve activity might be important in migraine. Burstein et al. (26, 27) suggested that sensitization of wide dynamic range neurones in trigeminal nuclei mediates scalp tenderness within the distribution of migraine headache. Spread of sensitization to wide dynamic range neurones in thalamic nuclei may then result in cutaneous allodynia elsewhere in the body. The present findings suggest that spatial summation of nociceptive signals across different divisions of the trigeminal nerve can facilitate pain. Thus, during attacks of migraine, spatial summation of nociceptive signals from diverse sources in cranial tissues could intensify headache and scalp tenderness.
Kaube et al. (28) recently reported that the R2 component of the blink reflex evoked by nociceptive stimuli delivered from a concentric electrode was over six times greater during migraine than during the headache-free interval, consistent with sensitization of cutaneous nociceptive afferents or second-order neurones in the trigeminal nucleus caudalis. Trigeminal sensitization may also persist subclinically during the headache-free interval (29). It is difficult to compare the present findings with those of Kaube et al., because different stimulation parameters and electrode types were used. Nevertheless, the facilitation of the blink reflex during migraine appears to differ qualitatively from the R2 inhibition associated with ice-induced headache. Whether the failure of inhibitory processes such as DNIC contributes to trigeminal sensitization in migraine sufferers (28, 29) requires further investigation.
Footnotes
Acknowledgements
This study was supported by grants from the National Health and Medical Research Council of Australia and the UK Migraine Trust. I wish to thank Ms Shiree Hassard for technical assistance.
