Abstract
SUNCT (Short-lasting Unilateral Neuralgiform headache attacks with Conjunctival injection and Tearing) and SUNA (Short-lasting Unilateral Neuralgiform headache attacks with cranial Autonomic symptoms) are rare primary headache syndromes, classified as Trigeminal Autonomic Cephalalgias (TACs). Hypothalamic involvement in the TACs has been suggested by functional imaging data and clinically with deep brain stimulation.
Fifty-two patients (43 SUNCT, 9 SUNA) were studied to determine the clinical phenotype of these conditions and response to medications. A functional imaging study explored activation of the posterior hypothalamus in attacks of SUNCT/SUNA.
The clinical study characterised SUNCT and SUNA in terms of epidemiology, phenotype and clinical characteristics.
Indomethacin is ineffective on single-blind testing. Intravenous lidocaine was effective in all cases. Open-label trails showed the effectiveness of lamotrigine, topiramate and gabapentin.
On functional imaging there was hypothalamic activation bilaterally in 5/9 SUNCT patients, and contralaterally in two patients. Two SUNCT patients had ipsilateral negative activation. In SUNA the activation was bilaterally negative. There was no hypothalamic activation in a patient with SUNCT secondary to a brainstem lesion.
The data suggests that there should be revised classification for SUNCT and SUNA, with an increased range of attack duration and frequency, cutaneous triggering of attacks, and a lack of refractory period. The concept of ‘attack load’ is introduced. The lack of response to indomethacin and the response to intravenous lidocaine, are useful in diagnostic and therapeutic terms, respectively. Preventive treatments include lamotrigine, gabapentin and topiramate. The role of hypothalamic involvement in SUNCT and SUNA as TACs is considered.
Introduction
SUNCT (short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing) is a rare primary headache syndrome that is grouped together with cluster headache and paroxysmal hemicrania as a trigeminal autonomic cephalalgia (TAC) (1), based on the now classical clinical combination of head pain and activation of cranial autonomic efferents (2). SUNCT is defined by the International Headache Society as being characterized by unilateral orbital or temporal pain that is stabbing or throbbing in quality and of moderate severity. There should be at least 20 attacks, lasting for 5–240 s and ipsilateral conjunctival injection and lacrimation should be present (3). SUNCT syndrome may be a subset of SUNA, short-lasting unilateral neuralgiform headache attacks with cranial autonomic features, although case reports of pure SUNA are rare (4). SUNCT is usually a primary headache syndrome. However, there are a number of cases that are secondary to intracranial lesions, typically either in the posterior fossa or in the pituitary gland. SUNCT has until recently been considered refractory to treatment. However, open-label trials of neuromodulatory treatments such as lamotrigine (5–9), carbamazepine (10, 11), topiramate (11–13) and gabapentin (14–16) have been successful in the preventive treatment of some cases of SUNCT. Non-pharmacological treatments include hypothalamic deep brain stimulation (17) and greater occipital nerve injection, which have been effective in a wide variety of headache conditions (18). Functional imaging work has shown that activation of the posterior hypothalamus is linked in positron emission tomography (PET) to attacks of cluster headache (19, 20) and PH (21). Hemicrania continua, which is neither migraine nor currently classified as a TAC, shares symptoms of both. A PET study in HC shows activation in the posterior hypothalamus, which correlates with hypothalamic activation in the TACs, and also in the dorsal rostral pons, which corresponds with loci for migraine (22). Functional MRI shows hypothalamic activation in SUNCT (23–25). Deep brain electrical stimulation of the posterior hypothalamus has had beneficial effects in both intractable CH (26) and SUNCT (17).
Aims and objectives of the thesis
A difficulty in defining the clinical characteristics of a rare syndrome is to obtain sufficient experience in one place and case histories to make comparisons and see common themes. This has not been hitherto possible with SUNCT/SUNA. Three main studies make up the body of the thesis: (i) clinical aspects of the phenotype of SUNCT and SUNA; (ii) treatment of SUNCT and SUNA; and (iii) functional imaging in SUNCT and SUNA with an a priori hypothesis that there is activation in the region of the posterior hypothalamus during attacks of SUNCT and SUNA, and that this activation increases parametrically with increasing levels of pain.
Clinical aspects of the phenotype of SUNCT and SUNA
The aim of this prospective study is to ascertain and further clarify the clinical and phenotypic characteristics of SUNCT and SUNA, in terms of the International Headache Society Classification of these syndromes (3), and to make comments about these syndromes based on a large clinical population of 52 patients with SUNCT and SUNA.
Methods
A cohort of 43 SUNCT and nine SUNA patients was assembled over 5 years. They were interviewed by phone or in the Outpatient Department.
In the course of the history taking, each patient was asked the same questions about: the side, site and character of their pain; duration, frequency and periodicity of their attacks; triggering and relieving factors; and the presence of a refractory period after which one attack could not be triggered following another. Most patients had attacks witnessed in clinic; the remainder were asked to keep prospective diaries recording the frequency and duration of their attacks, and the presence of autonomic symptoms. Patients' relatives also provided a corroborative history.
Results
Three attack types were identified: stabs, groups of stabs, and saw-tooth patterns (Fig. 1). The patients described the saw-tooth attacks as a single attack each, despite consisting of a volley of several shorter stabs, as the pain did not return to baseline for the duration of the saw-tooth attacks, which could last for up to hours at a time.

The three types of attack in SUNCT/SUNA.
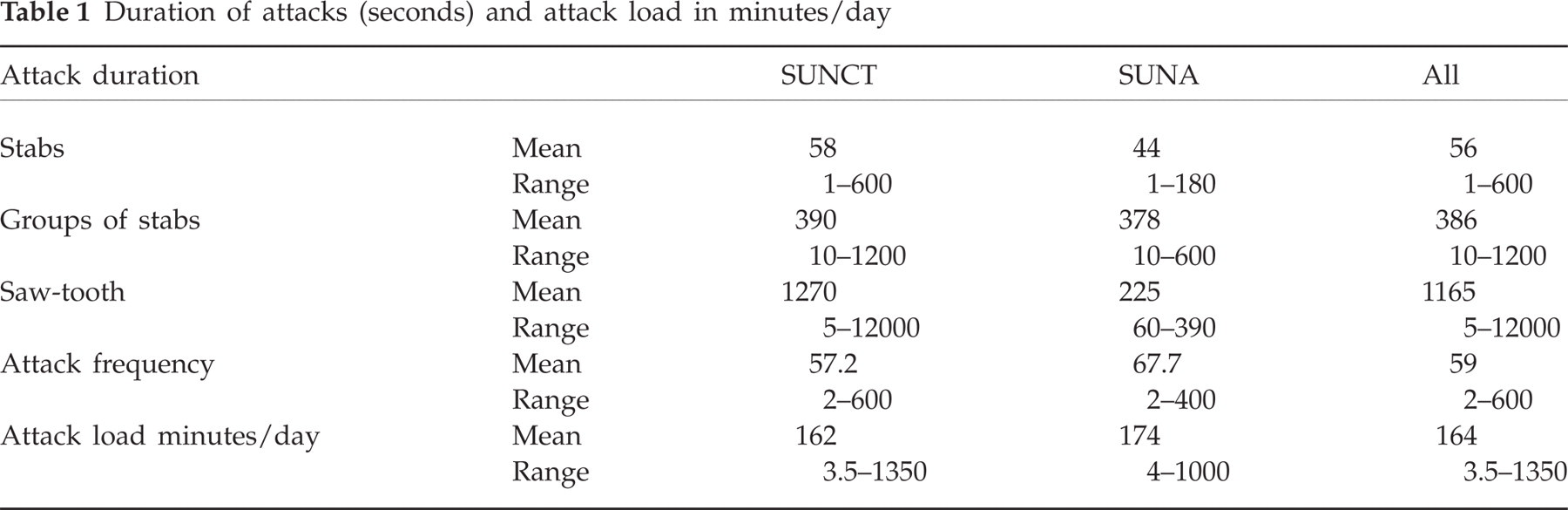
Mean duration of stabs was 56 s (1–600 s), stab groups 386 s (10–1200 s), and saw-tooth 1165 s (5–12000 s). Mean attack frequency was 59 attacks/day (2–600), dependent largely on the type of attack (Table 1).
Duration of attacks (seconds) and attack load in minutes/day
Pain was orbital, supraorbital or temporal in 38 (88%) SUNCT and seven (78%) SUNA, and also occurred in the retro-orbital region, side, top, back of head, V2, V3, teeth, neck and ear.
Twenty SUNCT (47%) and two SUNA (22%) patients experienced background pain; that is, they were not entirely pain free between attacks.
Eighty-eight per cent SUNCT and 78% SUNA patients experienced pain within the IHS-defined location. In others the pain was as in Table 2.
Site of attacks

All SUNCT patients had conjunctival injection and tearing. Two SUNA patients had conjunctival injection, four had tearing, but none had both. Other cranial autonomic symptoms included nasal blockage, rhinorrhoea, eyelid oedema, facial sweating/flushing, and ear flushing.
Cutaneous stimuli triggered attacks in 79% of SUNCT but only 33% of SUNA patients.
The majority (95% SUNCT and 89% SUNA) had no refractory period between attacks.
Thirteen SUNCT patients (30%) and one SUNA patient (11%) had abnormal findings on neurological examination. Twelve SUNCT patients and one SUNA patient had abnormal findings on intracranial imaging (Table 3).
Abnormal examination and intracranial imaging in SUNCT/SUNA

Discussion
This study found that SUNCT forms the majority subset of SUNA. Symptomatic forms of SUNCT and SUNA exist with pituitary lesions, vascular loops compressing the trigeminal nerve and other intracranial lesions. We suggest that the IHS criteria include the following: wider range of attack length, multiple types of attacks (stabs, groups of stabs and saw-tooth attacks), a wider distribution of cranial pain, cutaneous triggering, and lack of refractory period between attacks. Given the wide interpatient variation in frequency and duration of attacks, the concept of attack load in minutes of pain per day is introduced.
Treatments in SUNCT and SUNA
SUNCT/SUNA have been thought until recently to be refractory to treatment, although there have been case reports for lamotrigine (7), topiramate (12), gabapentin (15) and carbamazepine (11). There have also been reports of a good response to intravenous lidocaine (11). In terms of other TACs, an abortive treatment for CH is high-flow inhaled oxygen (27), and the standard treatment for PH is indomethacin (28).
Methods
A cohort of 43 SUNCT and nine SUNA patients was studied. As outpatients they received preventive medications in an open-label fashion as follows: lamotrigine up to 400 mg daily, topiramate up to 400 mg daily, and gabapentin up to 3600 mg daily.
Ten SUNCT and four SUNA patients were admitted for inhaled oxygen (100%, 12 l/min), the modified Indotest (100 mg indomethacin with saline placebo), and intravenous lidocaine 1.5–3.5 mg/kg/ h. Two SUNCT patients had modified Indotests at other centres. Eight SUNCT patients and one SUNA patient underwent injection of 2% lidocaine and 80 mg depomedrone in the region of the ipsilateral greater occipital nerve (GON).
For the abortive therapy (oxygen), a positive result was cessation or shortening of the attack. Preventive therapies were considered positive if the attacks were mostly or entirely suppressed for 12–24 h following the Indotest, or for the duration of the lidocaine infusion up to several months afterwards, or for the duration of the preventive.
Results
Inhaled oxygen was ineffective to abort attacks in 100% of patients. Indomethacin 100 mg intramuscularly was ineffective in 100% of patients.
Intravenous lidocaine had a moderate to good effect in all patients who recorded their response. Seven SUNCT patients were pain free for times varying between the duration of the infusion to 6 months. Three SUNCT patients had reduced attack frequency or severity, and one was lost to follow-up. All SUNA patients were pain free for 2 days to 12 weeks. These are illustrated in Table 4.
Preventive medications and greater occipital nerve block (GON) in SUNCT/SUNA
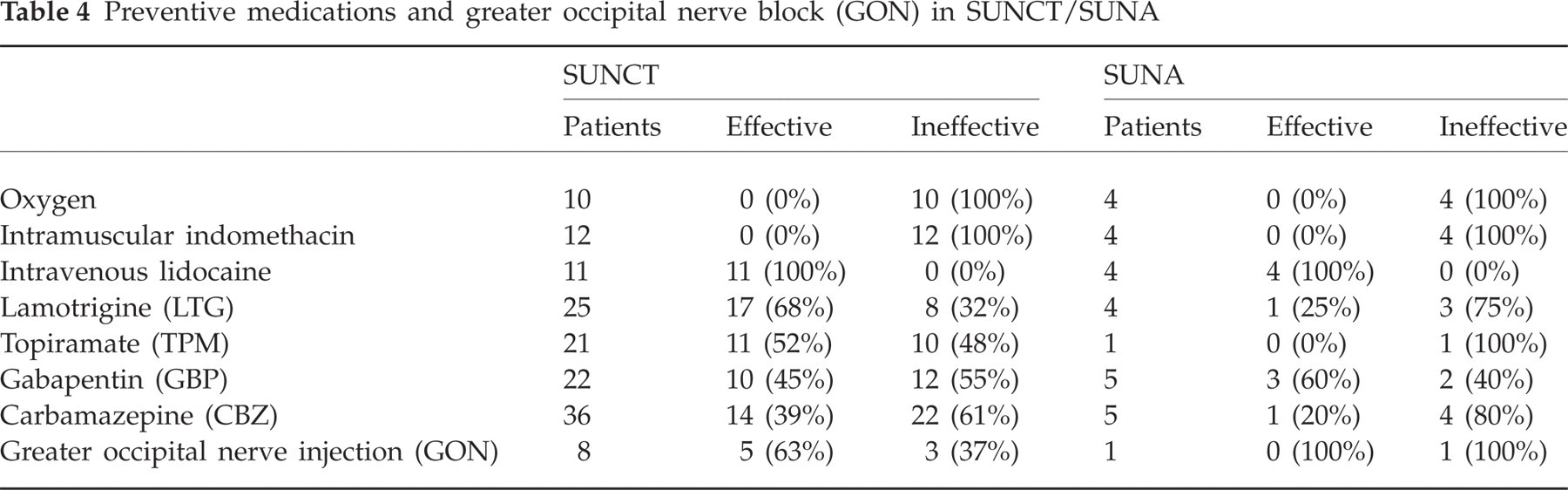
The following preventives were effective: lamotrigine (to 400 mg daily), 68% SUNCT, 25% SUNA; topiramate (to 400 mg daily), 52% SUNCT, 0% SUNA (800 mg, one patient); gabapentin (to 3600 mg daily), 45% SUNCT, 60% SUNA.
GON blockades had a good effect in five SUNCT patients (63% with pain free times ranging from 1 week to 6 months). In one of these patients the attacks returned after 2 weeks but on the contralateral side to the injection. The SUNA patient had a minimal transient effect (Table 4).
Discussion
To aid diagnosis of SUNCT/SUNA, we suggest an indomethacin test and oxygen trial, which will rule out the differential diagnoses of PH and CH, respectively. Intravenous lidocaine is effective for short-term prevention, although the effects can last for up to 6 months after the infusion. Lamotrigine and topiramate are recommended as preventives, and gabapentin may have a beneficial effect in SUNA. Greater occipital nerve injections may play a role as a neuromodulatory procedure, either acting at the trigeminocervical complex or more centrally.
Functional MRI in SUNCT and SUNA
Functional imaging studies have shown activation in the hypothalamus in cluster headache (19, 20), SUNCT (23), and in a trigeminal autonomic cephalgia not otherwise specified (29). Clinical studies with deep brain hypothalamic stimulation give further evidence for the role of the hypothalamus in cluster headache (26) and SUNCT (17).
We report BOLD (blood oxygen level dependent) changes in the hypothalamus in nine cases of primary SUNCT and two cases of SUNA, which showed activation corresponding to spontaneous attacks, and increased activation with increasing pain rating.
Methods
Nine patients with primary SUNCT, two with primary SUNA and one with a symptomatic SUNCT secondary to a brainstem lesion, underwent functional MRI scanning in four 10-min slots, during which there would be spontaneous or triggered attacks amongst attack-free periods. There was an auditory stimulus every 4 s, after which they would rate their pain (0 for no attack, 1 for mild attack, 2 for moderate and 3 for severe attack).
The images were analysed with SPM2 (http://www.fil.ion.ucl.ac.uk/spm/), by which a parametric analysis was set up for differential activation with respect to pain ratings. Brain activation in a region of interest in the hypothalamus was predicted to vary parametrically with pain ratings. Activated voxels are reported at P ≤0.001 uncorrected.
Results: BOLD changes
In patients with primary SUNCT there was positive activation in the region of the posterior hypothalamus, which was bilateral in five patients and contralateral to the side of the attack in two patients. Figure 2 shows an example in a patient with left-sided attacks. There was negative activation ipsilateral to the side of the pain in two patients. Figure 3 shows an example in a patient with right-sided attacks.

Bilateral positive hypothalamic activation during attacks of SUNCT.

Ipsilateral negative hypothalamic activation in SUNCT.
The patient with symptomatic SUNCT showed no activation in this area.
In the SUNA patients there was negative activation bilaterally.
Discussion
Previously, hypothalamic action has been described as ipsilateral to the side of the pain in SUNCT (23) and cluster headache (19), or bilateral in SUNCT (25), but contralateral in PET studies of paroxysmal hemicrania (21) and hemicrania continua (22). This study shows activation that can be contralateral to the pain, or bilateral, or negative activation ipsilateral to the side of the pain. In SUNA the activation was negative, and in symptomatic SUNCT there was no such activation. It strengthens the theory that the hypothalamus is a central generator for primary TACs, although this may not be true for symptomatic (secondary) SUNCT. This poses questions about the laterality of the attacks, in that some patients may be predisposed to contralateral symptoms. Patients with SUNA appear to have negative activation, and this may reflect the differences in phenotype between SUNCT and SUNA in terms of first division trigeminal (V1) cranial autonomic symptoms.
Discussion and conclusions
SUNCT and SUNA are rare primary autonomic headache syndromes that have been poorly understood due to the small numbers of patients in previous studies. Their pathophysiology has been speculated to involve the posterior hypothalamus, according to some of their shared clinical characteristics with other TACs such as CH and PH, and with evidence of functional imaging studies and deep brain stimulation in the posterior hypothalamic region.
As well as from functional imaging work, evidence for hypothalamic involvement stems from the fact that SUNCT/SUNA are TACs and related to CH. For example, the hypothalamus is known to regulate circadian (and seasonal) rhythms, and there are biochemical studies that demonstrated abnormal levels of serum testosterone, cortisol, prolactin, melatonin and thyroptropin in CH patients, suggesting hypothalamic dysfunction (30). However, these have not been done in SUNCT.
It is interesting to observe that the clinical feature that is common to the four primary headache syndromes in which posterior hypothalamic activation has been reported (i.e. PH, CH, SUNCT and HC) is prominent cranial autonomic features in association with the headache. It has been suggested that the pathophysiology of these syndromes revolves around the trigeminal-autonomic reflex (1). It has also been suggested that the cranial autonomic symptoms may be prominent in these syndromes due to a central disinhibition of the trigeminal-autonomic reflex by the hypothalamus (31). Indeed, there are direct hypothalamic-trigeminal connections (32), and the hypothalamus is known to have a modulatory role on the nociceptive and autonomic pathways, specifically trigeminovascular nociceptive pathways (33).
This project has undertaken a prospective clinical study in a large group of 52 SUNCT and SUNA patients. The phenotype of these two conditions has been characterized, and suggestions have been proposed to the current classifications by the International Headache Society, including: a wider variation of attack character, frequency and duration; the concept of attack load in minutes of pain per day; the ability to trigger attacks by cutaneous stimuli; and the lack of refractory period. These results build on previous work in which the character of the attacks and the lack of refractory period have been documented in case series (34–36).
Clinical studies have shown that SUNCT and SUNA do not respond to inhaled oxygen or intramuscular indomethacin, in contrast to CH and PH, respectively. Intravenous lidocaine, lamotrigine, topiramate and gabapentin have been shown to be useful in SUNCT and SUNA, as have GON injections in SUNCT.
The differences between SUNCT and SUNA have been addressed with regard to clinical phenotypes, response to medication, and the finding of negative activation on fMRI in SUNA. It is speculated that SUNA and SUNCT have differing hypothalamic activation, which may result in the lack of V1 cranial autonomic symptoms in SUNA and a reduction in cutaneous triggerability. The nomenclature suggests that SUNCT forms a subset of SUNA (which encompasses any combination of autonomic symptoms), yet it is recognized that the pathophysiology of SUNA may be different from that of SUNCT in terms of hypothalamic activation. Moreover, a variation in hypothalamic activation may lead to a spectrum of disorders from the relatively V1-poor SUNA to the striking degree of conjunctival injection and lacrimation that characterize SUNCT.
A fundamental tenet of primary headache syndromes is the lack of underlying structural abnormality in the brain. However, TACs are known to exist as a result of other disorders, including pituitary and posterior fossa lesions. This study presents new cases of symptomatic SUNCT and SUNA, and speculates on the pathophysiological differences as a result of one case of symptomatic SUNCT with no significant hypothalamic activation. Although the pathophysiology of primary and symptomatic SUNCT/SUNA may be different and clearly the primary pathology may warrant treatment of its own accord, the response to treatment of the attacks with preventive medications and intravenous lidocaine, and lack of response to indomethacin, were similar in both groups.
It is possible that although all TACs share some aspects of their phenotypes, and also both CH and SUNCT respond to deep brain stimulation at the posterior hypothalamus, the pathophysiology of these syndromes is different. This may be manifest by a more uniform activation of the ipsilateral posterior hypothalamic region in CH and PH, as compared with activations in SUNCT and SUNA over a wider area. SUNCT and SUNA are also phenotypically different to the other TACs in that the attacks are less stereotyped within the condition, with a wide range of attack duration and frequency, and with striking features such as triggering by cutaneous stimuli and lack of a refractory period between attacks. They also have a remarkable response to intravenous lidocaine, which can aid not just in the diagnosis of these conditions, but also afford a pain-free (and therefore drug-free) respite period for up to 6 months after cessation of the infusion. It is possible that longer attacks of SUNCT, particularly the saw-tooth attacks, which can last for up to hours at a time, may have been previously misdiagnosed as CH or PH.
Nosologically, the TACs can be divided into episodic or chronic forms of the disease for CH and PH (3). In this series both episodic and chronic SUNCT exist, although patients with primary chronic SUNCT and SUNA can go into remission for protracted periods (37), thus making this method of classification complex. SUNCT and SUNA may well be classified best according to the phenotype of their attacks, with details of the periodicity of their disease as an addendum.
The hypothesis that SUNCT and SUNA are associated with activation in the posterior hypothalamic region is upheld on an individual patient basis, but more evidence is needed that these are as strongly related to the hypothalamus as was previously thought. There is a need for biochemical evidence of hypothalamic derangement in these conditions, and from a clinical perspective a group of patients with SUNCT and SUNA should be followed-up for deep brain hypothalamic stimulation in the long term. Pre-clinical studies to ascertain the mechanism of action of lidocaine, topiramate and lamotrigine in these particular syndromes are needed. However, the prospect of taking a potentially ineffectual placebo for several weeks in such devastatingly painful conditions has put many patients off entering double-blind placebo-controlled studies, and this may prove even more difficult in such rare syndromes with already small numbers of patients.
In the meantime, the suggestion is to widen the range of diagnostic criteria, and treat SUNCT and SUNA with intravenous lidocaine, lamotrigine, topiramate, and gabapentin, especially in SUNA. Greater occipital nerve injections emerge as a new concept in treatment. Deep brain hypothalamic stimulation remains a possibility in the future, but with the reservation that the previously proposed hypothalamic activity in all TACs may not be the only mechanism for the pathophysiology of SUNCT and SUNA.
