Abstract
The aim of this study was to evaluate the efficacy and tolerability of topiramate for the prevention of chronic migraine in a randomized, double-blind, placebo-controlled trial. Chronic migraine is a common form of disabling headache presenting in headache subspecialty practice. Preventive treatments are essential for chronic migraine management, although there are few or no controlled empirical trial data on their use in this patient population. Topiramate is approved for the prophylaxis of migraine headache in adults. Patients (18–65 years) who experienced chronic migraine (defined as ≥15 monthly migraine days) for ≥3 months prior to trial entry and had ≥12 migraine days during the 4–week (28–day) baseline phase were randomized to topiramate or placebo for a 16–week, double-blind trial. Topiramate was titrated (25 mg weekly) to a target dose of 100 mg/day, allowing dosing flexibility from 50 to 200 mg/day, according to patient need. Existing migraine preventive treatments, except for antiepi-leptic drugs, were continued throughout the trial. The primary efficacy measure was the change in number of migraine days from the 28–day baseline phase to the last 28 days of the double-blind phase in the intent-to-treat population, which consisted of all patients who received at least one dose of study medication and had one outcome assessment during the double-blind phase. Health-related quality of life was evaluated with the Migraine Specific Quality of Life Questionnaire (MSQ, Version 2.1), the Headache Impact Test (HIT-6) and the Migraine Disability Assessment (MIDAS) questionnaires, and tolerability was assessed by adverse event (AE) reports and early trial discontinuations. Eighty-two patients were screened. Thirty-two patients in the intent-to-treat population (mean age 46 years; 75% female) received topiramate (mean modal dose ± SD = 100 ± 17 mg/day) and 27 patients received placebo. Mean (±SD) baseline number of migraine days per 4 weeks was 15.5 ± 4.6 in the topiramate group and 16.4 ± 4.4 in the placebo group. Most patients (78%) met the definition for acute medication overuse at baseline. The mean duration of treatment was 100 and 92 days for topiramate- and placebo-treated patients, respectively. Study completion rates for topiramate- and placebo-treated patients were 75% and 52%, respectively. Topiramate significantly reduced the mean number of monthly migraine days (±SD) by 3.5 ± 6.3, compared with placebo (-0.2 ± 4.7, P < 0.05). No significant intergroup differences were found for MSQ and HIT-6. MIDAS showed improvement with the topiramate treatment group (P = 0.042 vs. placebo). Treatment emergent adverse events were reported by 75% of topiramate-treated patients (37%, placebo). The most common AEs, paraesthesia, nausea, dizziness, dyspepsia, fatigue, anorexia and disturbance in attention, were reported by 53%, 9%, 6%, 6%, 6%, 6% and 6% of topiramate-treated patients, respectively, vs. 7%, 0%, 0%, 0%, 0%, 4% and 4% of placebo-treated patients. This randomized, double-blind, placebo-controlled trial demonstrates that topiramate is effective and reasonably well tolerated when used for the preventive treatment of chronic migraine, even in the presence of medication overuse.
Introduction
Migraine is an episodic brain disorder that affects about 15% of the population (1, 2), can be highly disabling (3) and has been estimated to be the most costly neurological disorder in the European Community at more than €27 billion per year (4). Patients with the highest frequency of attacks are the most disabled (5). Patients with daily or near-daily headache, typically defined as ≥15 days of headache per month (6), are often referred to as having chronic daily headache, and the subgroup with migrainous biology as having chronic migraine (7) or transformed migraine (8). Such patients may use acute attack medicines frequently (9–11). It is textbook teaching that such patients are refractory to preventive treatments until their acute attack medicines are stopped (12). This placebo-controlled trial of chronic migraine assessed whether topiramate would be effective in its preventive management in the practical clinical setting of including patients with medication overuse.
Topiramate is a sulphamate-substituted monosaccharide with a range of pharmacological actions on neurons (13). It has been shown to be an effective and reasonably well-tolerated preventive treatment of episodic migraine with or without aura (14–16). In these previous studies patients with >15 headache days a month were excluded, as were patients with medication overuse. The exclusion of these patients was, in part, a reflection of the lack of diagnostic criteria and considerable discussion around the biological nature of headache in migraineurs with an attack frequency exceeding 15 days per month, especially where there is medication overuse. The term chronic migraine was defined in the second edition of The International Classification of Headache Disorders (7) to address this issue. During the development of the revised criteria there was open discussion of the proposals (17) so that work began on the entity of chronic migraine with which the senior authors were familiar. This double-blind study concerned topiramate patients with chronic migraine, ≥15 days per month of migraine, whether or not they were overusing acute attacks medicines.
The data suggest the current textbook view that patients with medication overuse need to withdraw from their acute treatment(s) may be incorrect. The results of this trial have been presented in preliminary form at the 6th Meeting of the European Headache Federation (Valencia, Spain, 26 April 2006) (18).
Methods
Study design
This was a randomized, double-blind, placebo-controlled, parallel-group, multicentre trial of topiramate for the prevention of headache in patients with chronic migraine with and without medication overuse (Fig. 1) conducted in accordance with Good Clinical Practice (19). A prospective, 4-week baseline phase was followed by a 16-week, double-blind treatment phase, which consisted of a 4-week titration and 12-week treatment period. The taper-down phase (Fig. 1) lasted up to 7 weeks. Computer randomization was used prior to study start, medications were randomized in blocks of four, two topiramate and two placebo, and numbered. Each study centre received at least two blocks: one for subjects without medication overuse and one for subjects with medication overuse. Subjects were assigned to the next available medication number within the block. Topiramate and placebo, identical tablets produced by the manufacturer, were titrated to the target dose of 100 mg/day (50 mg twice daily) at a rate of 25 mg/week. Study physicians could increase or decrease the target dose (within a range of 50–200 mg/day) during the first 12 weeks of the double-blind phase, depending on efficacy, tolerability, or both. The dose of topiramate was required to remain stable during the last 4 weeks of the double-blind phase at the time when data for the primary end-point were collected. Last dose and modal dose of topiramate (or matching placebo) were assessed per patient, and the means ± SD presented per treatment group. Medication overuse, allowed in this trial, was operationally defined according to the revised International Headache Society (IHS) criteria (20) as the intake of ergotamine, triptans, or opioids, or combination analgesics for ≥10 days per 4 weeks, or the intake of simple analgesics for ≥15 days per 4 weeks. Randomization was stratified according to presence or absence of medication overuse in the baseline period.
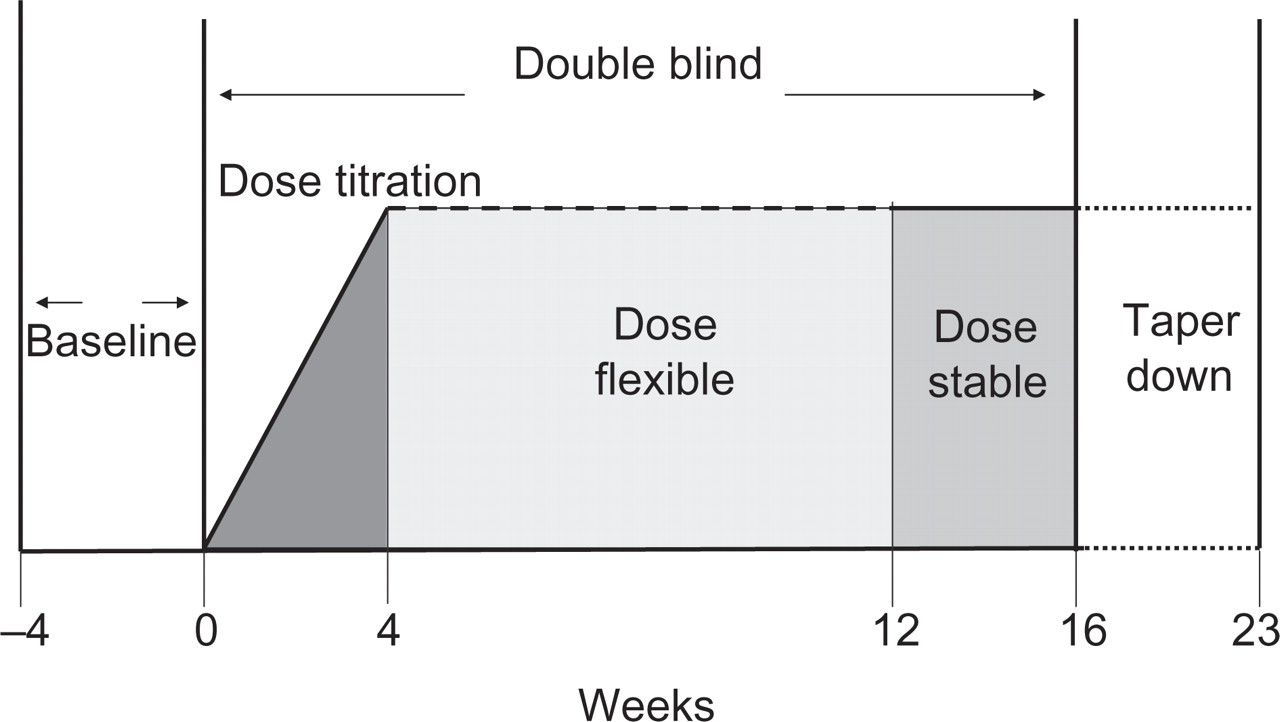
Trial design. A prospective baseline period was followed by a 4-week titration period, with increases of topiramate doses of 25 mg/week. Data for the primary end-point were collected in the last 4 weeks of treatment.
Inclusion criteria
Patients (18–65 years of age) were required to have a diagnosis of chronic migraine that satisfied the second edition of The International Classification of Headache Disorders criteria of ≥15 migraine headache days per 4 weeks, at least during the last 3 months prior to trial entry, with an established migraine history for at least 1 year (7). Patients could be included if they had ≥12 migraine days in the prospective baseline period.
Exclusion criteria
Patients were excluded if they presented with another primary chronic headache or any secondary headache except medication overuse headache (MOH). Patients who experienced onset of migraine after age 50 and those who were severely depressed [Beck Depression Inventory (BDI) scale score ≥30] were also excluded. Patients taking antidepressants were excluded unless the antidepressant was used at a stable dose for at least 3 months prior to trial entry and the patient intended to continue the antidepressant throughout the trial. In addition, patients taking any migraine prophylactic drug were excluded unless the drug had been used for at least 3 months (at a stable dose for at least 1 month) prior to trial entry and was continued throughout the trial. Other exclusion criteria included a prior history of topiramate use, use of other anticonvulsants within 30 days of trial entry and use of a carbonic anhydrase inhibitor.
Concomitant therapies
Patients were allowed to take acute rescue medications such as analgesics, non-steroidal anti-inflammatory drugs (NSAIDs), triptans, opioids and ergot derivatives during any phase in the trial as usual. The use of acute rescue medication had to be specified, next to the migraine attack information, in the trial-specific patient diary.
Outcome measures (efficacy)
The primary efficacy variable was the change in the mean number of monthly migraine days from baseline to the last 4 weeks of the double-blind phase. A migraine day was defined as a calendar day with symptoms of a migraine attack lasting at least 30 min. Secondary outcome measures included: change in monthly migraine days from baseline to the entire double-blind phase; the percentage of patients with ≥50% reduction in the mean number of monthly migraine days (categorical responder rates); change from baseline in the mean number of days of acute medication intake; patient satisfaction ratings with the efficacy and tolerability of the treatment they received; and mean changes from baseline on the Migraine-Specific Quality of Life Questionnaire (MSQ, Version 2.1) (21), Headache Impact Test (HIT-6) (22) and Migraine Disability Assessment (MIDAS) questionnaire (23) scores. All three questionnaires were administered at start and end of the double-blind treatment phase; the MSQ and HIT-6 were also administered at 4 and 8 weeks in the double-blind phase.
Tolerability and safety measures
Spontaneously reported treatment-emergent adverse events (TEAEs) were recorded. Vital signs, body weight changes and laboratory parameters, including bicarbonate, sodium, potassium and chloride, were measured at the start of the double-blind phase and at weeks 8 and 16. Fewer bicarbonate estimations were done compared with others since the bicarbonate measurement was added after the study had commenced.
Statistical analyses
Since the effect size of topiramate is unknown in subjects with chronic migraine, the following assumptions were made based on the results obtained in subjects with episodic migraine (24). First, the average number of migraine days would be between 15 and 28 at an average number of 20. Second, there would be a 45% reduction in the number of migraine days on topiramate. Third, there would be a 25% reduction in the number of migraine days on placebo, so the estimated effect size over placebo was four migraine days per month. Fourth, we estimated the SD of the change in the number of migraine days per month to be 5. Under these assumptions two treatment groups of 29 subjects each would be needed to show a statistically significant difference between topiramate and placebo with a power of 0.80 and α = 0.05 (two-sided). Efficacy analyses were performed on the intent-to-treat (ITT) population, which consisted of all randomized patients who received at least one postbaseline efficacy evaluation (25). Differences between treatment groups (topiramate vs. placebo) were compared using the Wilcoxon two-sample test for ordinal/continuous data, and interpreted at the 5% significance level (two-tailed comparison). Differences within a treatment group were tested using the Wilcoxon signed rank test (ordinal/continuous data). Fisher's exact test was used to assess differences between nominal data. For patients who dropped out, data from the last visit available were carried forward only for the end-point visit. Data have not been corrected for multiple comparisons.
Results
Patients were recruited between 10 November 2003 and 17 July 2005. The total ITT population (n = 59) consisted of 32 patients receiving topiramate and 27 patients receiving placebo (Fig. 2). There were no significant differences in demographics and baseline characteristics between the topiramate-treated and placebo-treated patients (Table 1). The trial was completed by 75% of the topiramate-treated and 52% of the placebo-treated patients (Table 2). The modal dose of topiramate was recorded for each individual patient; the treatment group mean modal dose ±SD was 100 ± 17 mg. Ten patients were on a stable dose of a medicine that might have had a concomitant prophylactic effect. Three patients in the topiramate arm were on a β-blocker and five in the placebo arm, while one in each arm took a calcium channel blocker (not flunarizine).

Disposition of patients. Primary reason for NO RANDOMIZATION (‘screen failure’) of 23 subjects (82 screened−59 randomized):
Demographics and baseline characteristics, intent-to-treat population

Reasons for discontinuation

Comments: ‘did not believe she was on active treatment’; ‘patient has no effectiveness of the trial medication’.
Medication overuse
The subgroup of patients who were overusing acute medication (n = 46) consisted of 23 patients receiving topiramate and 23 receiving placebo. There were no significant differences in demographics and baseline characteristics between the topiramate-treated and placebo-treated patients. It appeared, however, that triptans were the most commonly overused acute medications in the placebo group (96%, vs. 61% in the topiramate group), whereas the topiramate group had a higher rate of analgesic overuse (30%, vs. 9% in the placebo group). The modal dose of topiramate was assessed for each individual in this treatment subgroup. From these values, the calculated mean modal dose was 102 ± 17 (mg/day ± SD).
Efficacy
Topiramate significantly reduced the mean number of monthly migraine days (primary end-point) in patients with chronic migraine from 15.5 ± 4.6 at baseline by 3.5 ± 6.3 in the last 4 weeks of the double-blind phase, compared with an increase of 0.2 ± 4.7 days from 16.4 ± 4.4 at baseline for patients receiving placebo (P = 0.02; Fig. 3). For the subgroup of patients overusing acute medication, topiramate also significantly reduced the mean number of monthly migraine days by 3.5 ± 7.1 from baseline in the last 4 weeks of the double-blind phase, compared with an increase of 0.8 ± 4.8 days for patients receiving placebo (P = 0.03). A significantly higher percentage of patients receiving topiramate experienced a ≥50% reduction in mean monthly migraine days at end-point (22%), compared with placebo (0%, P = 0.012; Fig. 4).
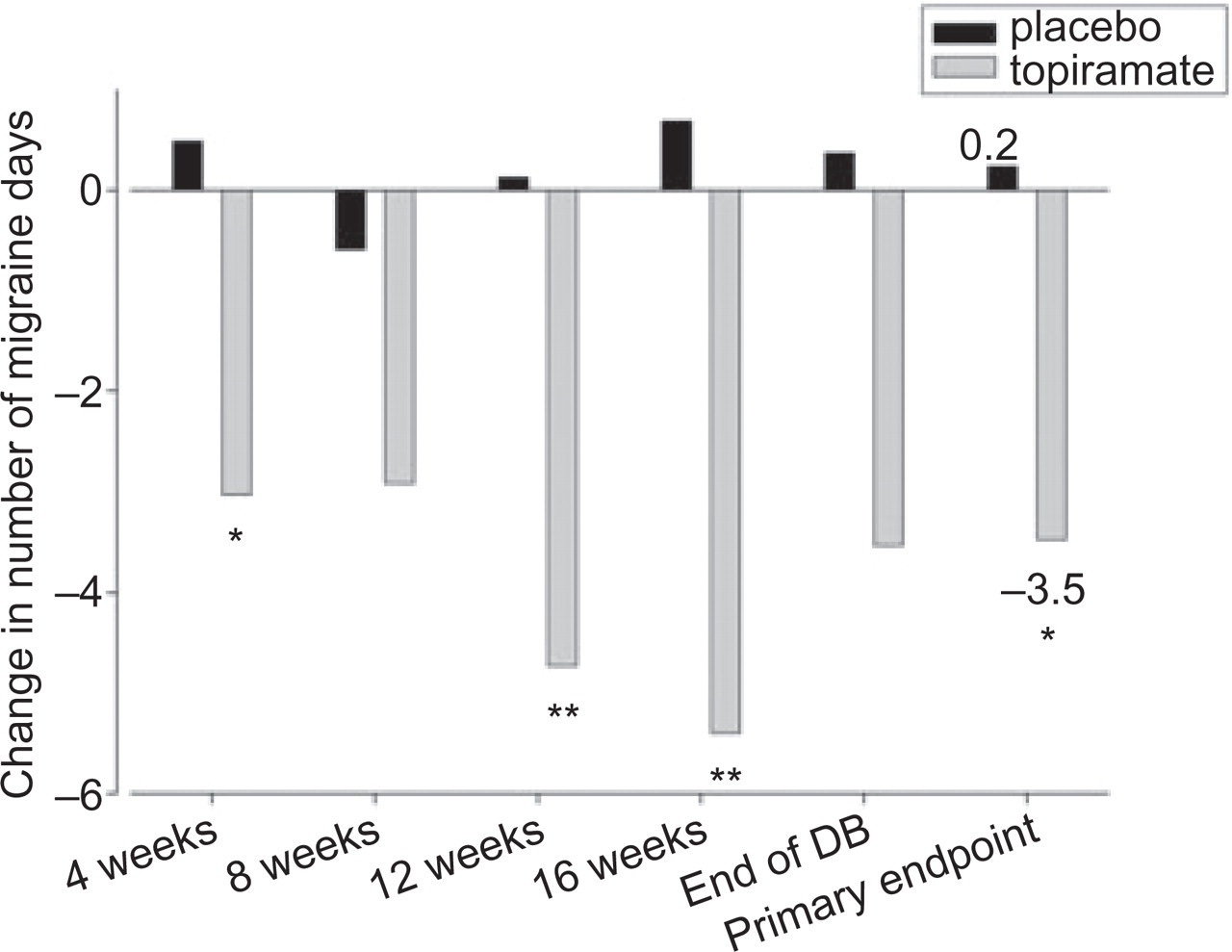
Mean change from baseline to monthly migraine days, intent-to-treat population. ▪, Placebo; □, topiramate. ∗P < 0.05; ∗∗P < 0.01 vs. placebo.

Rate of responders (50% reduction in migraine days). ▪, Placebo; □, topiramate. ∗P < 0.04 vs. placebo.
Medication intake
Topiramate therapy did not significantly reduce the mean number of days per month of acute medication intake, including all analgesics, when compared with the effect of placebo (P = 0.11). The baseline rate was 13.3 ± 6.8 days/month and the reduction was 3.0 ± 5.9 days/month, compared with placebo baseline of 14.7 ± 6.5 days/month and reduction of 0.7 ± 6.2 days. In the medication overuse subpopulation, topiramate also did not significantly reduce the mean number of monthly days of acute medication (reduction 3.7 ± 6.7) from baseline to the end of the double-blind phase, compared with placebo (0.5 ± 6.5 days/month; P = 0.07).
Disability
The baseline MIDAS total scores (± SD) were 67 ± 87 for topiramate (n = 25) and 61 ± 99 for placebo (n = 14). At end-point, the MIDAS scores were reduced by 26 ± 61 in the topiramate group, while the scores increased by 3 ± 21 in the placebo group (P = 0.04). There were no significant between-group differences in MSQ and HIT-6 scores at any point assessed during the double-blind phase or at trial end-point (data not shown).
Satisfaction
Of the topiramate-treated patients, 37% (n = 11) rated their satisfaction level with the effectiveness of the medication as ‘good’ or ‘very good’, compared with 10% (n = 2) of patients who received placebo (P = 0.04).
Tolerability and safety
Table 3 lists all treatment-emergent adverse events that occurred in two or more patients. Paraesthesia, a known side-effect of topiramate (24), was the most frequent adverse event with topiramate (53%, n = 17). No major safety concerns with the use of topiramate were reported in this trial. Two serious AEs were observed: one patient on topiramate was hospitalized for surgery for carpal tunnel decompression and one placebo patient was hospitalized because of neurogenic muscle spasm.
Treatment-emergent adverse events n (%) with incidence ≥2 subjects or of clinical importance

Slight decreases in systolic (decrease of 5.0 mmHg topiramate vs. increase of 1.5 placebo, P = 0.06) and diastolic (decrease of 4.3 mmHg topiramate vs. decrease of 0.3 placebo, P = 0.45) blood pressures were observed in the topiramate group. Body weight was significantly reduced during the double-blind phase in topiramate-treated compared with placebo-treated patients (P < 0.01). Topiramate-treated patients lost 2.05 ± 2.90 kg of weight by the end of the double-blind phase, while placebo-treated patients gained 0.09 ± 2.67 kg. There were no statistically significant or clinically relevant between-group differences in the results of clinical laboratory tests except for chloride levels, which were raised in the topiramate group compared with the placebo group (end-point + 3.9 ± 4.0 mmol/l for topiramate vs. −0.5 ± 4.6 mmol/l for placebo; P < 0.01).
Discussion
The data demonstrate that topiramate at a mean modal dose of 100 mg reduces migraine days in patients with chronic migraine when compared with placebo. Furthermore, this effect is seen in patients with medication overuse. Although our study was small, the result is robust and challenges the current textbook teaching on the optimal use of migraine preventives. When considering the therapeutic gain and the very small placebo response, the observed 50% response rate is in close agreement with published preventive studies in episodic migraine (24). The current trial suggests that chronic migraine is a tractable therapeutic target that requires further study. Moreover, the results suggest the need for a fundamental re-evaluation of the standard approaches to medication overuse. Additional well-designed, controlled and adequately powered trials will be required to understand fully the optimal timing for the use of migraine preventives when medication overuse is present.
Migraine is currently defined by the IHS as episodic if there are <15 affected days a month and chronic if there are ≥15 affected days per month, over at least a 3-month interval (7). Our inclusion criteria allowed patients with ≥12 days of migraine in the baseline to be entered, which is compatible with the latest revision of the classification that mandates essentially 8 days (20). Topiramate is effective for the treatment of episodic migraine (14–16). In a meta-analysis of controlled trials, the 50% responder rate, defined as the proportion of patients with a ≥50% reduction in migraine days, demonstrated a 24% therapeutic gain over placebo for the 100-mg dose of topiramate (24). Our current trial demonstrated a comparable value (22%) for the therapeutic gain achieved with topiramate compared with placebo. In a small placebo-controlled study, Silvestrini and colleagues (26) compared 14 patients in each of two groups, placebo and topiramate 50 mg daily, who had chronic migraine. They reported a 71% responder rate (50% improvement) for topiramate against a 7% placebo response rate. The current results from our larger scale trial support this finding and the small placebo rates are concordant. Furthermore, our results are consistent with those observed in a recent blinded parallel-group study of patients with chronic migraine that showed a reduction in headache frequency and improvement on the MIDAS scale with topiramate or valproate treatment (27). Similarly, we saw an improvement in MIDAS in our study. We saw no change in HIT-6 or MSQ. Disability scores have not been widely used in placebo-controlled trials of preventive treatment in migraine. MIDAS seems more sensitive here, and this may relate to its explicit real-world link to days of disability, which translates well in a preventive study.
An unusual result in our trial was the lack of a placebo response. Placebo effects are generally robust in pain trials (28) and well characterized in migraine studies (29). In the chronic migraine trial by Silvestrini and colleagues, a similar flat response to placebo was also observed (see Fig. 1 (26)). The randomization process in our trial seems to have resulted in treatment groups with similar baseline characteristics, therefore the possibility exists that some unblinding may have occurred. Various factors could have contributed to this, including differential tolerability profiles, such as weight loss being greater in the topiramate group. Additionally, paraesthesia was more common in the topiramate group compared with the placebo group. These differences are difficult to avoid unless one uses an active treatment that produces the same symptom. Interestingly, the same high rate of paraesthesia in topiramate-treated patients was observed in several trials of episodic migraine, yet a robust placebo effect was still reported (25). Our design may have contributed to the low placebo effect, in that while we had a run-in baseline, we did not require any change in treatment, either preventive or acute attack, so the patients were clinically stable. An important area of future investigation will be to examine whether or not there is a plausible biological explanation for the low placebo response rate in chronic migraine trials.
If the outcome of the study, a reduction in headache frequency in chronic migraine among patients given topiramate, was not clinically believable, speculating on a mechanism for the low placebo response may be fanciful. However, it seems from widespread clinical experience that the result of our study is correct insofar as topiramate is often used for the preventive management of chronic migraine, so could the low placebo response be specific to chronic migraine? The placebo effect appears to be an active process that alters the experience of pain (30) by engaging the same neural substrate as, for example, opioid analgesia: the rostral anterior cingulated gyrus and the brainstem (31). Alterations in these brain regions are well documented and associated with the early part of the brain response (32). The placebo effect is complex and it is accepted that not all patients/subjects have prominent placebo effects (33). Patients with chronic migraine have, by definition, daily or near-daily pain. It has been shown in chronic migraine studies using positron emission tomography imaging techniques, that brainstem regions are important in migraine. Areas of the pain-processing matrix, particularly the dorsolateral pons, are persistently active in chronic migraine (34). If areas of the brain normally contributing to the placebo effect are persistently active (i.e. ‘saturated’) in chronic migraine, this may obviate any observable placebo effect. An alternative explanation involves expectation of success, which is crucial for the placebo effect (33). In general, this expectation is low in chronic migraine sufferers and may therefore contribute to the lack of effect seen in our trial. Since few cohorts with chronic migraine have been studied using placebo-controlled methodologies, our data do at least invite careful consideration of these issues in larger trials in the future.
Perhaps the most interesting finding of this study is the positive effect seen in patients with medication overuse. Although medication use did not decrease in the topiramate group compared with the placebo group, the differences were notable and the lack of a statistically significant effect may have been due to a lack of power. It is taught and written often that for preventive agents to be successful, medication overuse must first be controlled (12). This view is at least 20 years old (35). It is widespread practice to withdraw patients from medication overuse on either an out-patient or in-patient basis with either some adjunctive treatment, such as NSAIDs, or with no analgesia at all. Our study suggests that this may not be necessary prior to the initiation of preventive treatment. Topiramate treatment resulted in a robust reduction in the number of monthly migraine days in the medication overuse subpopulation. A certain proportion of patients could immediately begin preventive therapy without the need for the possible discomfort associated with medication withdrawal. Clearly, not everyone responded to treatment. However, it seems crucial to investigate this issue further, since it would be unethical to continue to insist on medication withdrawal without additional treatment unless it is necessary. Our data do not advocate an immediate change in clinical practice, but they certainly demand a careful re-evaluation and prospective well-powered controlled trials of clinical practice in order to define how best to manage this very disabled group of patients.
In summary, we report a randomized, placebo-controlled trial demonstrating that topiramate is superior to placebo in reducing migraine days in patients with chronic migraine. We saw a clear effect that was present in the subgroup of patients with medication overuse. The study had a very small placebo effect, which may represent an unblinding issue or may suggest some interesting pathophysiological differences between patients with very frequent and relatively infrequent migraine. Trials that study clinical practice are sorely needed. Patients with chronic migraine and medication overuse are common in referral practice, highly disabled, and burdensome to society. It is imperative that the optimal strategies for their management are evaluated with well-controlled clinical trials.
Authors' contributions
The trial idea and initial protocol were developed by H-C.D. and P.J.G. The study was sponsored and the data analysed by the sponsor. H-C.D. and P.J.G. wrote the first draft of the manuscript.
Acknowledgements
J.C.V.O., M.L. and S.S. are employees of Janssen-Cilag.
