Abstract
Eletriptan 40 mg and 80 mg have shown greater efficacy in acute migraine than oral sumatriptan 100 mg and naratriptan 2.5 mg. This study continues the systematic series of active comparator trials in the eletriptan clinical development programme. In a multicentre double-blind, double-dummy, parallel-groups trial, 1587 outpatients with migraine by IHS criteria were randomised in a 3 : 3 : 3 : 1 ratio to eletriptan 80 mg, eletriptan 40 mg, zolmitriptan 2.5 mg or placebo. Of these, 1312 treated a single migraine attack and recorded baseline and outcome data to be included in the intention-to-treat population. The primary analysis was between eletriptan 80 mg and zolmitriptan. For the primary efficacy end-point of 2-h headache response, rates were 74% on eletriptan 80 mg, 64% on eletriptan 40 mg, 60% on zolmitriptan (P < 0.0001 vs. eletriptan 80 mg) and 22% on placebo (P < 0.0001 vs. all active treatments). Eletriptan 80 mg was superior to zolmitriptan on all secondary end-points at 1, 2 and 24 h, in most cases with statistical significance. Eletriptan 40 mg had similar efficacy to zolmitriptan 2.5 mg in earlier end-points, and significantly (P < 0.05) lower recurrence rate and need for rescue medication over 24 h. All treatments were well tolerated; 30-42% of patients on active treatments and 40% on placebo reported all-causality adverse events that were mostly mild and transient. On patients' global ratings of treatment, both eletriptan doses scored significantly better than zolmitriptan.
Introduction
With seven triptans now marketed for the acute treatment of migraine, the question has been asked whether the pharmacokinetic and pharmacodynamic differences between them result in clinically relevant differences in efficacy (1). Such questions go to the heart of evidence-based medicine; they are answered at best partially, and at worst misleadingly, by meta-analyses of trials in which direct comparisons were not the original intent but are made nonetheless between groups drawn from different populations without random allocation between them (2–5). The definitive answer, according to the authors who posed the question, could only come from ‘a wide range of randomised clinical trials comparing triptans’ (1).
Eletriptan is a second-generation triptan with rapid and consistent absorption following oral administration, high bioavailability and potent agonist activity at 5-HT1B/1D receptors (6–8). In a series of randomised controlled trials eletriptan has shown dose-related efficacy against acute migraine in the range 20–80 mg. In head-to-head comparative trials, eletriptan 40 mg and 80 mg have been superior on a range of standard efficacy measures to sumatriptan 100 mg (9–11), oral Cafergot (12) and naratriptan 2.5 mg (13).
Zolmitriptan, usually in a dose of 2.5 mg, is a commonly prescribed alternative to sumatriptan (2). Clinical trials have produced little evidence of greater efficacy from a dose of 5 mg (2, 14–16). Since the 2.5-mg dose is associated with a somewhat lower incidence of adverse events (14), this appears to be the optimal dose in terms of benefit/risk ratio. In many countries, 2.5 mg remains the only licensed dose of zolmitriptan. It was therefore the appropriate comparator dose.
This study, conducted as part of the clinical development programme for eletriptan, continues the series of direct comparisons with widely used treatments for acute migraine. The primary objective was to compare eletriptan in a dose of 80 mg with zolmitriptan 2.5 mg for efficacy and tolerability, with eletriptan 40 mg included as an additional comparator. A placebo control was added for external validation in line with recommendations of the International Headache Society (IHS) (17).
There is continuing debate over end-points traditionally employed in acute migraine trials, and whether these reflect patients’ views or values. In particular it is uncertain whether or not headache response (see below), a measure of efficacy currently required by regulators, adequately captures the multiple dimensions of efficacy or even the one that is most important. In acknowledgement of this debate, and to support research into other end-points, this study adopted a range of secondary measures. In reporting these, the authors recognize the importance of showing that differences between treatments are not only expressed in the primary end-point but also reflected consistently across the range of efficacy variables.
Methods
Patients
Male or female patients aged 18–65 years were included, meeting IHS criteria for migraine with or without aura (18) and with attacks at least once every 6 weeks. All gave informed consent in line with recommendations of IHS (19) prior to a physical examination, 12-lead ECG and (in women of child-bearing potential) a urine pregnancy test.
The following were the principal grounds for exclusion: migraine that had been consistently resistant to all treatments; basilar migraine; hemiplegic migraine; frequent nonmigrainous headaches; any clinically significant medical illness or laboratory abnormalities, especially those indicative of coronary artery disease, heart failure or uncontrolled hypertension; other contraindications to treatment with eletriptan or zolmitriptan including use of potent CYP3A4 inhibitors concomitantly or of MAO inhibitors within 2 weeks of entry; severe reduction in gastrointestinal absorption; misuse of alcohol or other substances including analgesics, ergotamine or triptans; pregnancy or breast-feeding. Women who might become pregnant were required to use effective contraception.
Study design
The study was a randomised, double-blind, placebo-controlled parallel-groups comparison between treatments in outpatients, conducted in 144 centres in 19 countries in Europe. The protocol was consistent with the Declaration of Helsinki and approved by Institutional Review Boards or Research Ethics Committees for every site.
After recruitment, patients received study medication to treat, within 4 h of its onset but after resolution of any aura, one attack of migraine with headache of moderate or severe intensity and not improving at the time of treatment. Study medication was not to be used in an attack already treated with any analgesic or anti-emetic or within 48 h of taking any triptan, ergotamine or dihydroergotamine.
Baseline assessments and later outcomes, including adverse events, were recorded in diaries. All patients were asked to arrange, within 48 h of use of study medication, their return for review within 14 days. Any who did not treat an attack within 12 weeks after randomisation, for whatever reason, were withdrawn from the study.
Treatments
Entered patients were randomly assigned, in a 3 : 3 : 3 : 1 ratio, to eletriptan 80 mg, eletriptan 40 mg, zolmitriptan 2.5 mg or placebo. Randomisation was in blocks of 10, but investigators were not made aware of this. Each centre was given a computer-generated list and allocated prenumbered treatments to consecutive patients by next-number on this list.
Patients who failed to respond to their study medication by 2 h were permitted to take rescue medication of their choice, but not any triptan or ergot. Patients who did respond but later experienced recurrence (see below) were allowed a second dose of the same study medication no earlier than 4 h after the first dose. Rescue medication was similarly permitted 2 h after this second dose if needed. Use of rescue medication and/or a second dose of study medication was noted in the diary. No efficacy data were requested after a second dose, but adverse events continued to be recorded.
Masking
A double-dummy design was used. Eletriptan tablets were matched with placebo tablets; thin gelatin capsules containing zolmitriptan were matched with identical capsules containing inactive substance.
Zolmitriptan blinded in this manner met standard criteria for full bioequivalence. These require the 90% confidence intervals (CI) about the ratios of the geometric mean pharmacokinetic parameters AUC and Cmax of new (encapsulated) vs. reference (unencapsulated) formulations to fall within the range 0.80–1.25. A bioequivalence study (n = 34; data on file, Pfizer Inc, New York, USA) previously conducted to support the current trial found the AUC ratio to be 0.97 (90% CI: 0.93–1.01) and the Cmax ratio to be 1.02 (90% CI: 0.94–1.10). Time-to-maximal concentration (tmax) was 1.3 h for the encapsulated formulation and 1.5 h for the unencapsulated.
Evaluation
Patients recorded migraine-related symptoms in their diaries immediately before treatment (baseline) and at 30 min and 1, 1.5, 2, 4 and 24 h afterwards. The primary efficacy end-point was headache response within 2 h of taking the first dose of study medication. Headache response was defined as improvement in headache intensity from a baseline of severe or moderate intensity (3 or 2 on a 4-point scale) to mild or no pain (1 or 0).
Secondary efficacy variables were the following:
headache-response rates at 30 min, 1 and 1.5 h;
pain-free rates at 30 min, 1, 1.5 and 2 h;
absence of associated symptoms of nausea, photophobia and phonophobia at 30 min, 1, 1.5 and 2 h;
functional recovery at 1 and 2 h on a 4-point functional impairment scale (3, bed rest required; 2, severe impairment in work, study or housekeeping activities, but not requiring bed rest; 1, some but not severe impairment in these activities; 0, normal level of functioning even if headache is still present) in the subset of patients recording baseline impairment of 3 or 2;
headache-recurrence rate, where recurrence was defined as return of moderate or severe headache within 24 h after response within 2 h;
use of rescue medication;
sustained headache response, defined as headache response within 2 h with no recurrence or use of rescue medication within 24 h;
sustained pain-free, defined as pain-free within 2 h with no recurrence or use of rescue medication within 24 h;
patient's global evaluation of study medication at 24 h on a 7-point Likert scale ranging from 0 (extremely poor) through 3 (neutral mid-point) to 6 (excellent) in response to the question: ‘Overall, how would you rate the study medication you received to treat the migraine attack?’;
acceptability of study medication, defined by the patient's answer (‘yes’ or ‘no’) to the question: ‘Given the choice between this and any other previous medication you have used to treat a migraine attack, would you take this again?’
Adverse events were recorded by patients in free text in their diaries as they occurred, together with quantitative evaluations of severity and duration. Attributions of seriousness and causation were made at follow-up by investigators applying standard procedures.
Statistical analyses
The primary efficacy measure was the percentage of patients per treatment group with headache response at 2 h after the first dose of study medication. The primary efficacy comparison was between eletriptan 80 mg and zolmitriptan 2.5 mg. The study was powered at 90% to detect as significant (P < 0.05 two-tailed) an 11-percentage-point difference in response rates between eletriptan 80 mg and zolmitriptan 2.5 mg, but at only 52% for the secondary comparison between eletriptan 40 mg and zolmitriptan. Assuming 2-h headache response rates of 70% for eletriptan 80 mg, 59% for zolmitriptan and 40% for placebo, 1584 patients should be randomised.
All efficacy analyses were performed on the intention-to-treat (ITT) population, defined as all subjects who took study medication and had a valid baseline and at least one post-treatment evaluation. Baseline characteristics of the treatment groups within the ITT population were compared descriptively for homogeneity. The primary analysis was conducted using a categorical linear model based on the SAS procedure CATMOD, which included terms for treatment and baseline headache intensity. In case of statistically significant between-groups differences at baseline, adjustments were made. Secondary end-points were analysed similarly.
All statistical tests were reported as showing significance by convention at the 5% level (2-sided unless otherwise specified). No adjustments were made for multiple comparisons. Pair-wise comparisons were not done unless the overall comparison of treatment groups was significant.
Results
Study dates
Patient recruitment began on 10 June 1999 and ended on 2 October 2000. The last patient visit was on 16 January 2001; data collection ended on 8 March 2001.
Baseline characteristics of patients
A total of 1587 patients were randomised and 1337 (84.2%) received treatment (Fig. 1). Of these, 25 were not available for efficacy analysis in the ITT population (n = 1312; 82.7%), mostly because of lack of a protocol-defined baseline assessment. The demographic and baseline clinical characteristics of the treated patients in each treatment group (Table 1) were similar, and typical of patients entering short-term clinical trials in migraine.
Demographic and baseline clinical characteristics of treatment groups
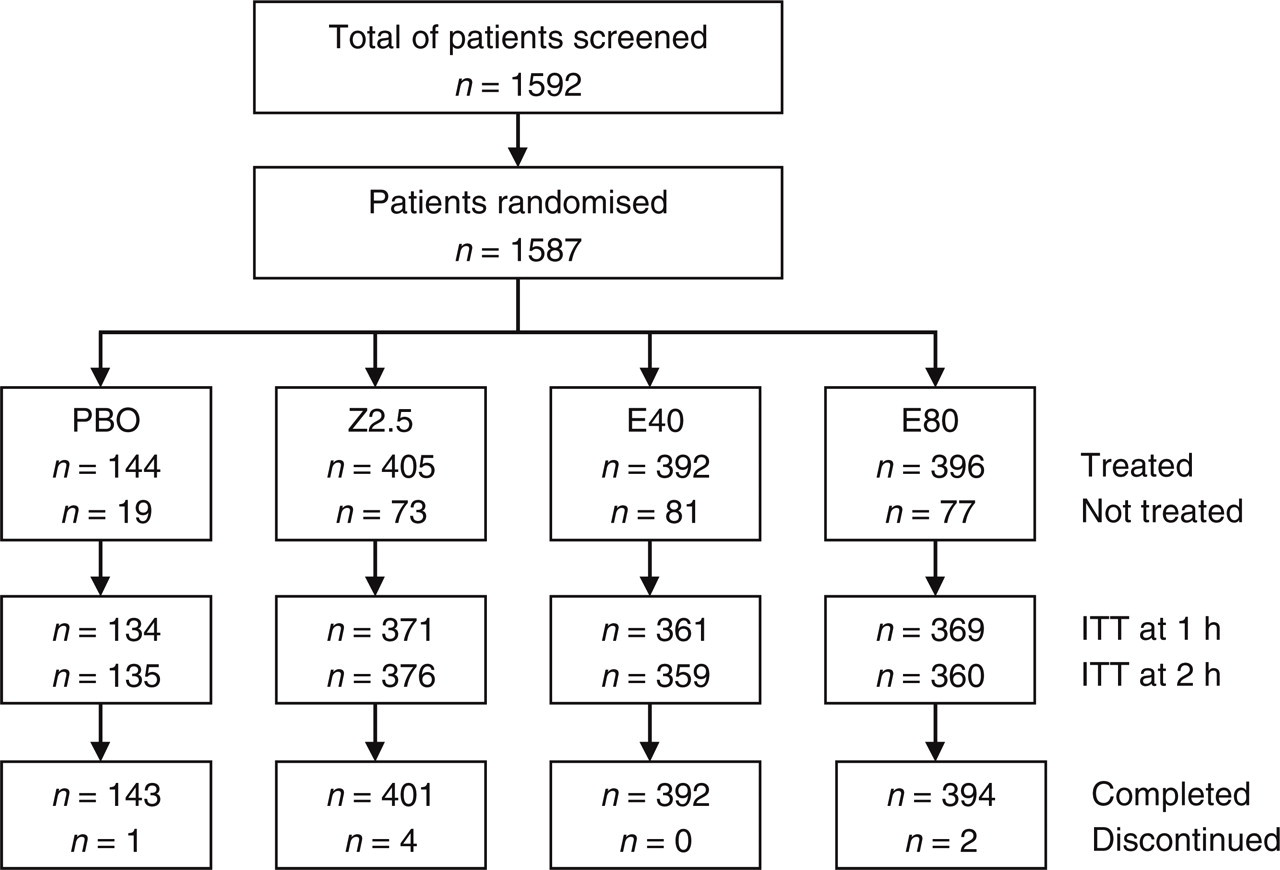
Disposition of patients. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; ITT, entering the intention-to-treat analysis.
Headache
On the primary efficacy end-point of headache response at 2 h (Fig. 2), eletriptan 80 mg (265/360, 74%) was significantly superior to zolmitriptan (224/376, 60%; P < 0.0001) and placebo (30/135, 22%; P < 0.0001). This finding was reflected in 1-h response rates: eletriptan 80 mg (149/369, 40%) was superior to zolmitriptan (93/371, 25%; P < 0.0001) and placebo (7/134, 5%, P < 0.0001). Separation was in fact evident at 30 min (eletriptan 80 mg: 44/378, 12%; zolmitriptan: 27/382, 7%, P < 0.05; placebo: 7/134, 5%, P < 0.05). Eletriptan 40 mg was superior to placebo (P < 0.0001) at 2 h (229/359, 64%) and 1 h (101/361, 28%) but not significantly better than zolmitriptan at any time point.
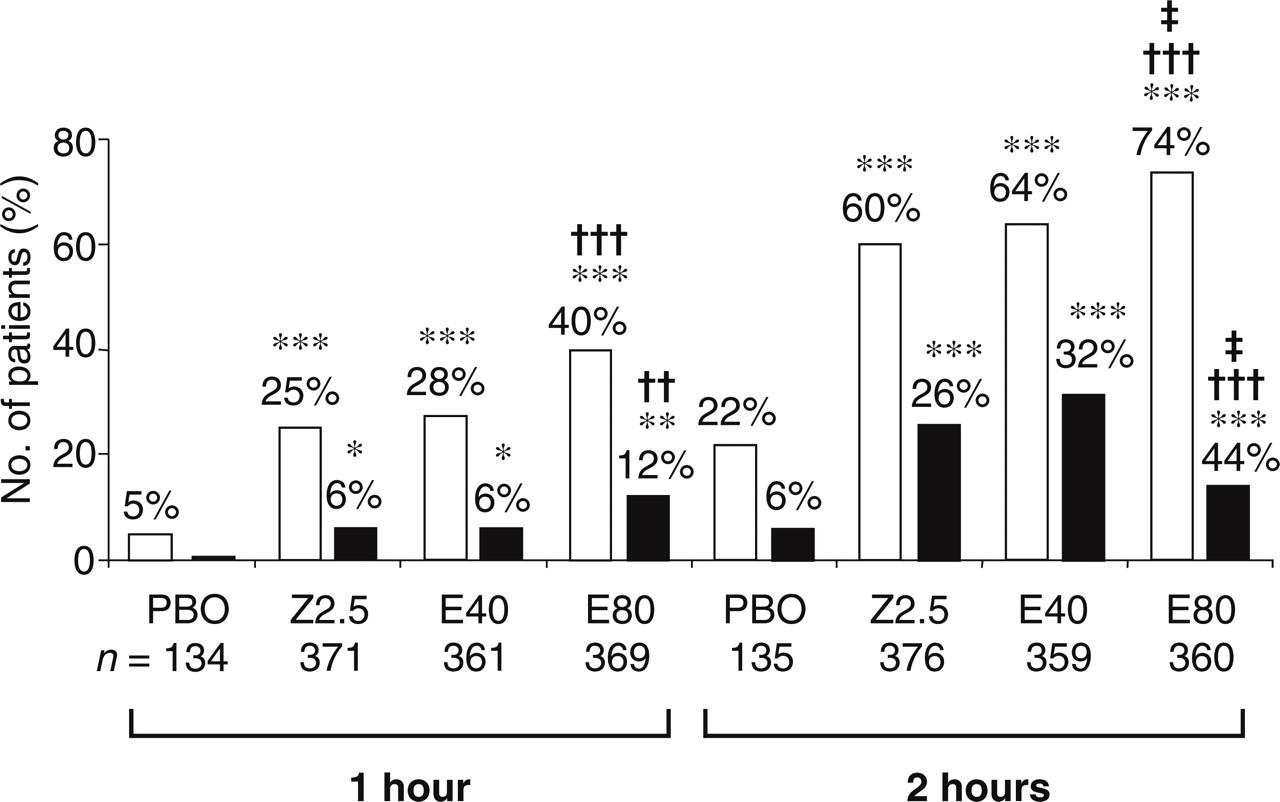
Headache-response and pain-free rates at 1 and 2 h post-treatment by treatment group. □ Headache response; ▪ pain free. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗P < 0.05; ∗∗P < 0.01; ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, ††P < 0.01; †††P < 0.0001; comparisons between eletriptan 80 mg and eletriptan 40 mg, ‡P < 0.01.
Pain-free rates also reflected the primary analysis (Fig. 2). Eletriptan 80 mg was superior at both 2 h (157/360, 44%) and 1 h (44/369, 12%) to zolmitriptan (99/376, 26%: P < 0.0001; 21/371, 6%: P < 0.01) and placebo (8/135, 6%: P < 0.0001; 1/134, < 1.0%: P < 0.01). Eletriptan 40 mg was significantly better than placebo at 2 h (115/359, 32%: P < 0.0001) and 1 h (21/361, 6%: P < 0.05) but not zolmitriptan.
Eletriptan 80 mg was significantly better (P < 0.01) than eletriptan 40 mg in headache response and pain-free rates at 2 h.
Associated symptoms
These analyses were conducted in all patients (Fig. 3) since baseline frequencies of these symptoms were broadly similar between groups (Table 1). This approach took account of emergent symptoms (especially nausea) not present at baseline that might not be reported as adverse events. All active treatments were significantly better than placebo (P < 0.001) at reducing all symptoms within 2 h. Numerically, more patients treated with eletriptan 80 mg than with zolmitriptan were free of nausea (257/357, 72% vs. 242/376, 64%: P < 0.05), photophobia (254/357, 71% vs. 240/375, 64%: P= 0.058) and phonophobia (260/354, 73% vs. 253/372, 68%: P = 0.064). Eletriptan 40 mg was better than zolmitriptan in reducing nausea (259/358, 72%: P < 0.05) but not significantly different from zolmitriptan in reducing photophobia and phonophobia.

Absence of associated symptoms at 2 h post-treatment by treatment group. □ PBO;  Z2.5;
Z2.5;  E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗∗P < 0.001; ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05.
E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗∗P < 0.001; ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05.
Functional impairment
In the subsets with severe or moderate functional impairment at baseline (3 or 2 on the scale 0–3), all active treatments were better than placebo (P < 0.0001) at bringing improvement (Fig. 4). Patients on eletriptan 80 mg (response rates: 194/285, 68% at 2 h; 100/296, 34% at 1 h) did better than those on zolmitriptan (171/303, 56% at 2 h: P < 0.05; 73/303, 24% at 1 h: P < 0.05). Eletriptan 40 mg (181/296, 61%; 73/300, 24%) was not significantly different from zolmitriptan on this measure.

Functional response at 1 and 2 h post-treatment by treatment group in the subset of patients with severe or moderate functional impairment (3 or 2 on a scale of 0–3) at baseline. □ PBO;  Z2.5;
Z2.5;  E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05.
E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05.
Use of rescue medication
Significantly fewer patients used rescue medication after eletriptan 80 mg (53/390, 14%) than after zolmitriptan (101/395, 26%: P < 0.0001) or placebo (81/140, 58%: P < 0.0001) (Fig. 5). This was true of those taking eletriptan 40 mg also (76/387, 20%: P < 0.05 vs. zolmitriptan; P < 0.0001 vs. placebo).

Headache-recurrence rate and use of rescue medication within 24 h post-treatment by treatment group. □ PBO;  Z2.5;
Z2.5;  E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗P < 0.05; ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05; †††P < 0.0001.
E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗P < 0.05; ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05; †††P < 0.0001.
Headache recurrence
In the subsets of patients achieving headache response by 2 h, headache-recurrence rates were numerically lower on eletriptan 80 mg (84/253, 33%: P = 0.271) and significantly lower on eletriptan 40 mg (65/225, 29%: P < 0.05) than on zolmitriptan (83/218, 38%) (Fig. 5). Both doses of eletriptan had significantly lower recurrence rates than placebo (16/31, 52%: P < 0.05).
Sustained headache response and pain-free
More patients achieved headache response by 2 h and survived to 24 h without recurrence or use of rescue medication (sustained headache response) on eletriptan 80 mg (160/338, 47%: P < 0.001) and 40 mg (151/345, 44%: P < 0.01) than on zolmitriptan (125/362, 35%) (Fig. 6). Eletriptan 80 mg (P < 0.0001) and 40 mg (P < 0.0001), as well as zolmitriptan (P < 0.0001), were all significantly better than placebo (14/131, 11%).

Sustained-response and sustained-pain-free rates at 24 h post-treatment by treatment group. □ PBO;  Z2.5;
Z2.5;  E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗P < 0.05; ∗∗P < 0.01; ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05; ††P < 0.01.
E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗P < 0.05; ∗∗P < 0.01; ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05; ††P < 0.01.
Sustained-pain-free rate was higher for eletriptan 80 mg (100/343, 29%) than for zolmitriptan (61/367, 17%: P < 0.001) (Fig. 6). Eletriptan 80 mg (P < 0.0001) and 40 mg (75/349, 22%: P < 0.0001), as well as zolmitriptan (P < 0.01), were better than placebo (6/134, 5%).
Tolerability and safety
All adverse events reported within 7 days of treatment were recorded regardless of causation. Both eletriptan and zolmitriptan were well tolerated and most adverse events were mild and transient. The totals of patients reporting at least one all-causality adverse event of any sort were 168/396 (42%) on eletriptan 80 mg, 117/392 (30%) on eletriptan 40 mg, 137/405 (34%) on zolmitriptan 2.5 mg and 57/144 (40%) on placebo (Table 2). Only four classes of adverse events not attributable to migraine occurred with an incidence of ≥3% on any active treatment (Table 2). Each of these showed a dose–response relationship with eletriptan and they were generally more frequent after eletriptan than after zolmitriptan and after all active treatments than after placebo.
Treatment-emergent adverse events (all-causality) with incidence ≥3% in any active-treatment group in the population (n = 1337) taking treatment

Numbers are % of patients reporting at least one event coded to that term. Symptoms attributable to migraine (i.e. nausea, photophobia, phonophobia) are not included since they are factored into 2-h efficacy data (Fig. 3).
There were no serious adverse events on either drug and no safety concerns arose in this trial.
Patients’ global ratings
Patients’ ratings of treatment acceptability (‘would use again’) (Fig. 7) showed preferences for eletriptan 80 mg (232/381, 61%: P < 0.05) and 40 mg (244/379, 64%: P < 0.01) over zolmitriptan (205/389, 53%). All active treatments were rated significantly better than placebo (26/137, 19%: P < 0.0001).

Patients’ global assessments at 24 h post-treatment by treatment group: global evaluation of treatment as ‘excellent’ or ‘good’; treatment acceptability (‘would use again’) against previously used treatments. □ PBO;  Z2.5;
Z2.5;  E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05; ††P < 0.01.
E40; ▪ E80. PBO, placebo; Z2.5, zolmitriptan 2.5 mg; E40, eletriptan 40 mg; E80, eletriptan 80 mg; comparisons with placebo, ∗∗∗P < 0.0001; comparisons between eletriptan and zolmitriptan, †P < 0.05; ††P < 0.01.
On the 7-point global rating of study medication (Fig. 7), analysis was of the percentage of patients in each group recording either ‘excellent’ or ‘good’. Eletriptan 80 mg (254/387, 66%) and 40 mg (243/380, 64%) were both rated more highly than zolmitriptan (214/389, 55%: P < 0.01). All active treatments scored significantly better than placebo (24/139, 17%: P < 0.0001).
Discussion
The principal outcome of this large double-blind, placebo-controlled, head-to-head comparison between eletriptan and zolmitriptan is that eletriptan 80 mg is statistically and clinically significantly better on the primary efficacy end-point – headache response at 2 h – than zolmitriptan 2.5 mg. Treatment with eletriptan 80 mg conferred a 10–15% efficacy advantage over zolmitriptan between one and two hours after treatment. Previously published studies comparing other triptans head-to-head with zolmitriptan 2.5 mg have found no significant differences in headache response between zolmitriptan and rizatriptan (20) or sumatriptan (16).
At the beginning, emphasis was laid on the importance of showing that differences apparent in the primary efficacy end-point were reflected across a range of variables expressing other dimensions of efficacy. The superiority of eletriptan 80 mg over zolmitriptan 2.5 mg has been demonstrated in headache-response rates at 1 h and at 30 min, pain-free rates at 2 h and 1 h, relief of nausea by 2 h, functional recovery at 2 h and 1 h, use of rescue medication, sustained headache-response and pain-free rates, treatment acceptability and global evaluation. The trial was therefore highly internally consistent.
The greater efficacy of eletriptan 80 mg against nausea as well as pain deserves comment. This advantage over zolmitriptan was shared by eletriptan 40 mg (Fig. 3). Next to headache, nausea is generally considered the most disabling symptom of migraine. Lipton et al. (21) analysed the relative efficacies of several triptans in relieving nausea, finding that only zolmitriptan, and not sumatriptan or naratriptan, achieved equivalent relief to rizatriptan. They speculated that nausea relief might, at least in part, be correlated with lipophilicity and the ability to reach central nervous system targets. Whether or not this hypothesis is correct, the superior effect of eletriptan against nausea in the current study is consistent with it, since eletriptan has the highest lipophilicity among triptans (1).
Comparisons between eletriptan 40 mg and zolmitriptan were secondary purposes of this study, and findings should be interpreted cautiously. It is fair to say that eletriptan 40 mg was at least as efficacious as zolmitriptan 2.5 mg. It performed better than zolmitriptan on some measures of efficacy including relief of nausea and, perhaps importantly, both global ratings of treatment effect (Fig. 7). It appears that the early onset advantage over zolmitriptan of eletriptan 80 mg was not so evident with eletriptan 40 mg (which had only a 4–6%-point advantage over zolmitriptan in 2-h headache-response, pain-free and functional-response rates), but eletriptan 40 mg was associated with a lower headache-recurrence rate than zolmitriptan. Therefore, sustained headache-response rate, a 24-h measure of efficacy, showed a significant (P < 0.05) difference between eletriptan 40 mg and zolmitriptan 2.5 mg. These results suggest that increasing the dose of eletriptan from 40 mg to 80 mg in unselected patients shifts the response curve to the left, resulting in higher early response rates.
All three treatments were well tolerated, in that adverse events were mostly mild or moderate and none were serious. Asthenia, dizziness, chest symptoms and somnolence all featured, as they commonly do in triptan trials. All were most frequent on eletriptan 80 mg, and three were reported more often on eletriptan 40 mg than on zolmitriptan, suggesting that the incidence of these class side-effects is to some extent a penalty of efficacy. Patients presumably make judgements based on the balance between these opposing factors, with overall satisfaction perhaps more strongly correlated with efficacy than with tolerability (22, 23). We see this expressed in global ratings which were higher for eletriptan than for zolmitriptan in this study (Fig. 7).
Safety is not a major issue with triptans, as is demonstrated by the accumulating evidence from many trials as well as postmarketing experience. Given this fact, an approach that may place differences between triptans in adverse-event rates into a clinically relevant context is numbers-needed-to-harm (NNH) analysis. For example, asthenia had its highest incidence (8.3%) with eletriptan 80 mg. Compared with the rate for zolmitriptan (2.5%), this gives rise to an NNH of 17; in other words, a physician must treat 17 patients with eletriptan 80 mg rather than zolmitriptan 2.5 mg before observing one additional patient who reports asthenia. For the other adverse events, NNHs are in the range 32–56. These calculations may provide one explanation for why the apparent differences in tolerability have little impact on global ratings of study medication.
Two issues in this study relate to its external validity. The first is encapsulation of the comparator drug, which is a routine practice in randomised clinical trials. Regulators require that studies of a new drug are conducted using formulations that will be marketed, whilst competitor companies are resistant to making available placebos to match their own drugs. Evidence of bioequivalence of encapsulated zolmitriptan to the marketed formulation was adduced, relying upon standard pharmacokinetic methodology. In the present study we find evidence of pharmacodynamic bioequivalence also. Firstly, zolmitriptan in this study showed significantly greater efficacy than placebo across all of the outcome measures. Secondly, headache-response rates at 2 h (the primary end-point), both absolute (60%) and placebo-subtracted (38%), were similar to those obtained in past studies with unencapsulated zolmitriptan (2, 14). Pain-free and sustained-pain-free rates in the current study accord (within the 95% confidence intervals) with results reported in a large recent meta-analysis (2).
A second issue, and an important validity check, is the placebo-response rate. Because of the ‘subjective nature of migraine and the high placebo effect’, the IHS clinical trials guidelines (17) require that a ‘placebo control should be included in order to test the reactivity of the patient sample.’ A variety of factors that may influence placebo response include illness severity, past experience of treatments, and unequal randomisation in which the probability of receiving active drug is high enough to influence patient-response through enhanced expectation of benefit (24). Other variables that contribute to placebo response, however, remain mysterious and unidentified (25). Nonetheless, the observed placebo-response rate in a given trial is the most practical benchmark for the validity of the treated population sample, and suspicion is raised when it is very high. Reported rates are as high as 44% in acute migraine trials (2). The influence on efficacy evaluations of patients highly responsive to placebo is uncertain. We are comfortable with the relatively low placebo-response rate (22%) on the primary end-point in this study.
Whilst pain and the associated symptoms of migraine are notoriously subjective, it may be argued that need for bedrest behaviourally expresses severity and therefore has some external validity. In the current study 29% of patients recorded need for bed rest at baseline, which may be compared to a mean of 17% in rizatriptan studies in which placebo-response rate was persistently high (26). The likely inverse correlation between baseline functional impairment and responsiveness to placebo deserves further investigation.
Meanwhile meta-analyses seeking to ‘compare’ different treatments, unprotected by randomisation, cannot control for such cross-study differences in patient samples no matter how comprehensive and carefully presented they may be. Head-to-head comparisons are necessary (1). In this head-to-head trial we found eletriptan 80 mg to be superior to zolmitriptan 2.5 mg in the acute treatment of migraine, and this was demonstrated consistently across a range of measures including the primary end-point. On secondary protocol-specified analyses we found eletriptan 40 mg to be as efficacious as zolmitriptan 2.5 mg, but with better acceptability to patients.
Footnotes
Acknowledgements
Financial support for this study was provided by Pfizer Inc. We express our appreciation to the members of the study group:
Austria: U Baumhackle, D Klingeler, G Ladurner, E Ott, J Rainer, E Rumpl, PG Wessely; Belgium: J Caekebeke, M DeTourtchaninoff, LF Herroelen, J Jacquy, P Louis, G Monseu, J Schoenen, MEH Van Zandijke; Croatia: T Babic, V Demarin; Czech Republic: P Docekal, I Rektor, M Vesela, G Waberzinek; Denmark: F Bach, E Hansen, R Kirkeby, TB Kristensen, M Rishoj, D Rost, P Staehr, A Ziebel; France: A Auteret, G Chazot, G Geraud, P Henry, MM Lanteri-Minet, B Laurent, C Lucas, H Massiou, B Minout, A Pradalier, E Roullet, JM Senard, T Soisson, D Valadec, JM Visy, JE Werner; Germany: A Beckmann-Reinholdt, K Bornhovd-Braeuer, H-C Diener, S Evers, R Frieling, H Göbel, W Gudden, R Haberl, B Kukowski, W Molt, C Panhans, V Pfaffenrath, JM Ribbat, U Schax, R Schellenberg, G Schumann, B Steinberg, S Strache, W Meyer; Greece: A Kazls, K Karageorgiou, S Peioglou-Harmoussis, T Thomaides; Hungary: J Czopf, G Gacs, A Valikovics, L Vecsey; Italy: M Boccuni, G Bono, M Bussi, G Bussone, A Carolei, G D'Andrea, R DiPerri, V Gallai, F Lanaia, GC Manzoni, F Perini, L Pinessi, FM Puca, G Relja, G Sandrini, M Silvestrini, ES Sternieri, G Zanchin, Netherlands: A Keyser, HJ Kok, PHM Pop, MG Smits, TJ Tacke, PMM Van Erven, HBM Van Lieshout; Norway: D Bansevicius, KM Hellum, R Kloster; Poland: H Kwiecinski, W Nyka, A Prusinski, Z Stelmasiak, A Stepien; Portugal: LAS Cunha, P Esperancia, F Ferreira, JM Pereira Monteiro; Slovakia: I Gogolak, P Kukumberg, P Turcani; Slovenia: T Pogacnik, E Tetickovic; Spain: CR Arnall, M Asensio Asensio, F Morales Asin, M Aguilar Barbera, D Jimenez Hernandez, JJ Zarranz Imirizaldu, C Hernandez Lahoz, RG Maldonado, R Leira Muino, M Gracia Naya, JJ Vilchez Padilla, JA Heras Perez, J Sancho Rieger, S Gimenez Roldan, J Prat Roto, RA Serrano, E Diez Tejedor, F Titus; Switzerland: C Meyer, B Nater; UK: A Dowson, PJ Fell, PJ Goadsby, P Husselbee, N Legg, EA MacGregor, PJ Mrazzi, TJ Steiner, FG Wright.
