Abstract
The risk of cluster headache (CH) is associated with the G-allele of the G1246A polymorphism in the hypocretin receptor 2 (HCRTR2) gene. First-line medication is effective in only about 70-80% of CH patients. We hypothesized that the HCRTR2 G1246A polymorphism is also of pharmacogenetic relevance in CH and may affect treatment response. We performed a prospective cohort study among 184 unrelated White CH patients. While the HCRTR2 1246G allele was significantly associated with CH in this group, treatment outcomes with triptans, oxygen, verapamil and corticosteroids remained unaffected. Our results do not support a role of the HCRTR2 G1246A polymorphism in drug responses in CH.
Introduction
Cluster headache (CH) is a rare idiopathic headache disorder with a distinct clinical presentation including a striking rhythmicity of attacks and autonomic symptoms (1). First-line treatment options of CH include triptans and oxygen for acute CH attacks and verapamil and corticosteroids for CH prevention (1). However, both treatment of CH attacks and preventive therapy of CH episodes are effective in only about 70–80% of patients (1–8). Currently, the lack of effect in individual patients is unclear, suggesting pharmacogenetic influences.
Functional neuroimaging studies suggest that the postero-lateral hypothalamus is the prime trigger for CH attacks (9). The hypothalamus is the only origin of hypocretin (orexin) neurons projecting into virtually all brain regions (10, 11). The hypocretin system is involved in the regulation of many vegetative functions including control of rhythmicity, feeding behaviour and hormone secretion, suggesting a link between the hypocretin system and CH (12). Indeed, two independent studies have associated the gene for the hypocretin receptor type 2 (HCRTR2) with CH (13, 14), while a genome-wide scan and HCRTR2 candidate gene analysis found no association of the HCRTR2 gene with CH (15). Furthermore, accumulating evidence suggests that the hypocretin system is involved in nociception and pain modulation (16, 17). Given the close relation of the hypocretin system to pain modulation, we aimed to evaluate whether the HCRTR2 G1246A polymorphism, in addition to its association with CH, affects treatment efficacy in CH patients.
Subjects and methods
Patients
The cohort for this study has been described previously (14). In brief, we prospectively recruited 257 unrelated White CH patients between 2002 and 2004. Seventy-three patients were excluded for reasons of incomplete data on treatment response, associated features or genotyping failures, leaving 184 patients for analysis. Of these, 44 (23.9%) were recruited from the headache clinic of the University Hospital in Essen, Germany and 140 (76.1%) from self-help groups from all over Germany. One hundred and forty-three (77.7%) were men and 41 (22.3%) women. CH diagnoses were verified according to the 1988 International Headache Society (IHS) criteria (18) and re-evaluated using the revised IHS criteria from 2004 (19). Mean age at first diagnosis was 44.8 years (
Special emphasis was paid to medication: we asked which first-line medication for CH attacks was used and whether it was effective at least three times. In accordance with established standards, we considered the acute therapy effective if the medication reduced CH pain by at least 50% within 15 min of a subcutaneous application or within 30 min of any other application form compared with untreated attacks (2, 6, 8). Preventive medication was considered effective if the CH attack frequency was reduced by at least 50% within 2 weeks (20). Written consent was obtained from all participants and the local ethics committee of the University of Essen approved this study.
DNA genotyping
Genomic DNA was extracted from whole blood or buccal swabs using the QiaAmp Mini DNA kit (Qiagen, Hilden, Germany). The HCRTR2 G1246A polymorphism (dbSNP: rs2653349) was analysed by restriction fragment analysis. The genomic region harbouring rs2653349 was polymerase chain reaction (PCR)-amplified with primers 5′- CCTTTCAGATCCCTGGAACA-3′ and 5′- ATTGGTAGATAGCAAATT-GCAAATACCAAGATCACAA-3′ under standard PCR conditions. The resulting amplicons were digested with the restriction enzyme BseJI (MBI Fermentas, St Leon Rot, Germany) in the provided reaction buffer at 65 °C and visualized after electrophoresis on 2.5% agarose gels. For the G-allele we observed a non-restricted fragment of 204 bp, for the A-allele two fragments of 34 and 170 bp.
Statistics
Consistency with Hardy–Weinberg equilibrium was determined by comparison of predicted and observed genotype distributions with χ2 tests. Logistic regression was used to compare responder and non-responder of the various therapy regimes. We calculated crude and multivariable-adjusted odds ratios (ORs) and the corresponding 95% confidence intervals (CIs). We a priori considered age at enrolment (continuous), gender (male vs. female), CH form (episodic vs. chronic) and smoking habits (current vs. never/past) as covariates for the adjusted model. We considered a two-tailed P-value of <0.01 to be statistically significant, as proposed standard for genetic association studies (21). All analyses were performed using the SAS system (version 8.2; SAS Institute, Cary, NC, USA).
Results
Table 1 summarizes major characteristics of CH patients stratified by HCRTR2 genotype. The observed genotype distribution was not significantly different from a theoretical Hardy–Weinberg equilibrium (P = 0.37). Carriers of the AA genotype were older than carriers of the GA genotype, who were older than carriers of the GG genotype. However, the differences were not statistically significant and are most likely to be coincidental because of the small number of GA and AA genotype carriers.
Baseline characteristics of cluster headache (CH) patients according to HCRTR2 genotype status
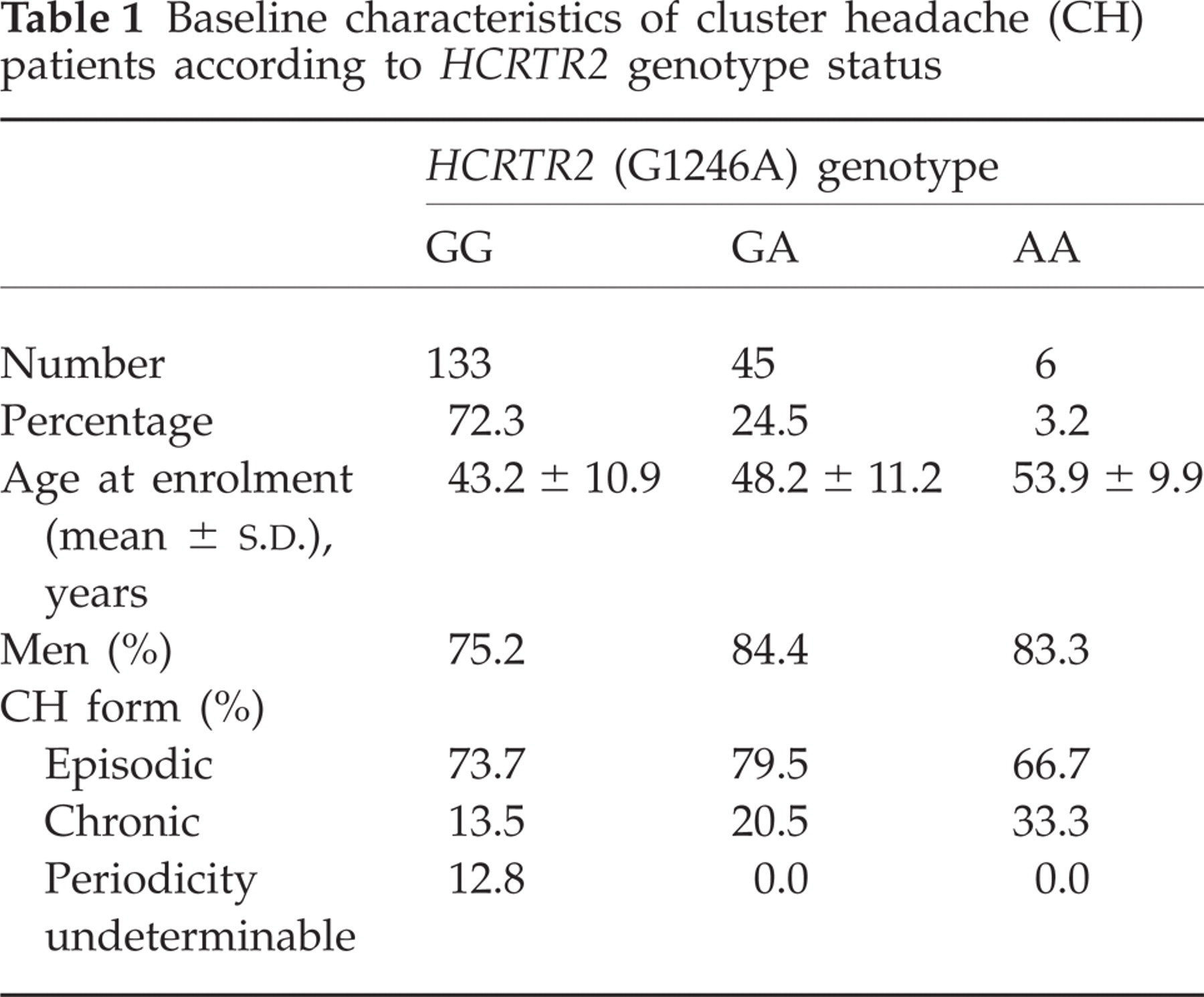
Our analyses focused on drug responses to triptans, oxygen, verapamil and corticosteroids. Other drugs (including lidocaine, non-steroidal anti-inflammatory drugs, opioids, ergots, lithium, valproate) were used by only a small fraction of patients, mostly with uncertain effects. For treatment of acute attacks, 149 CH patients (81.0%) reported the use of triptans and 132 of oxygen (71.7%). For prophylaxis, 134 CH patients (72.8%) used verapamil and 110 (59.8%) used corticosteroids.
Treatment response was reported by 104 triptan users (69.8%), 101 oxygen users (76.5%), 85 verapamil users (63.4%) and 83 corticosteroid users (75.5%). The HCRTR2 G1246A genotype distribution did not differ between responders and non-responders for any of these drugs (triptans χ2 = 0.05, 2 d.f., P = 0.97; oxygen χ2 = 3.64, 2 d.f., P = 0.16; verapamil χ2 = 2.89, 2 d.f., P = 0.24; corticosteroids χ2 = 4.43, 2 d.f., P = 0.11). Table 2 summarizes these data.
Utilization and treatment response rates of acute and prophylactic cluster headache (CH) medication according to HCRTR2 genotype status

Percentage of all CH patients analysed (n = 184).
Percentage of patients reporting the use of the respective treatment.
Percentage of responders/non-responders.
Table 3 presents the results of the comparison of treatment response between carriers of the 1246A allele and homozygous carriers of the wild-type G-allele. There was no difference in response to any of the investigated medication between heterozygous and homozygous carriers of the A-allele or a combination of both and wild-type carriers. After adjusting for age, gender, CH type and smoking status, the results did not change.
Cluster headache (CH) treatment responses and HCRTR2 genotypes
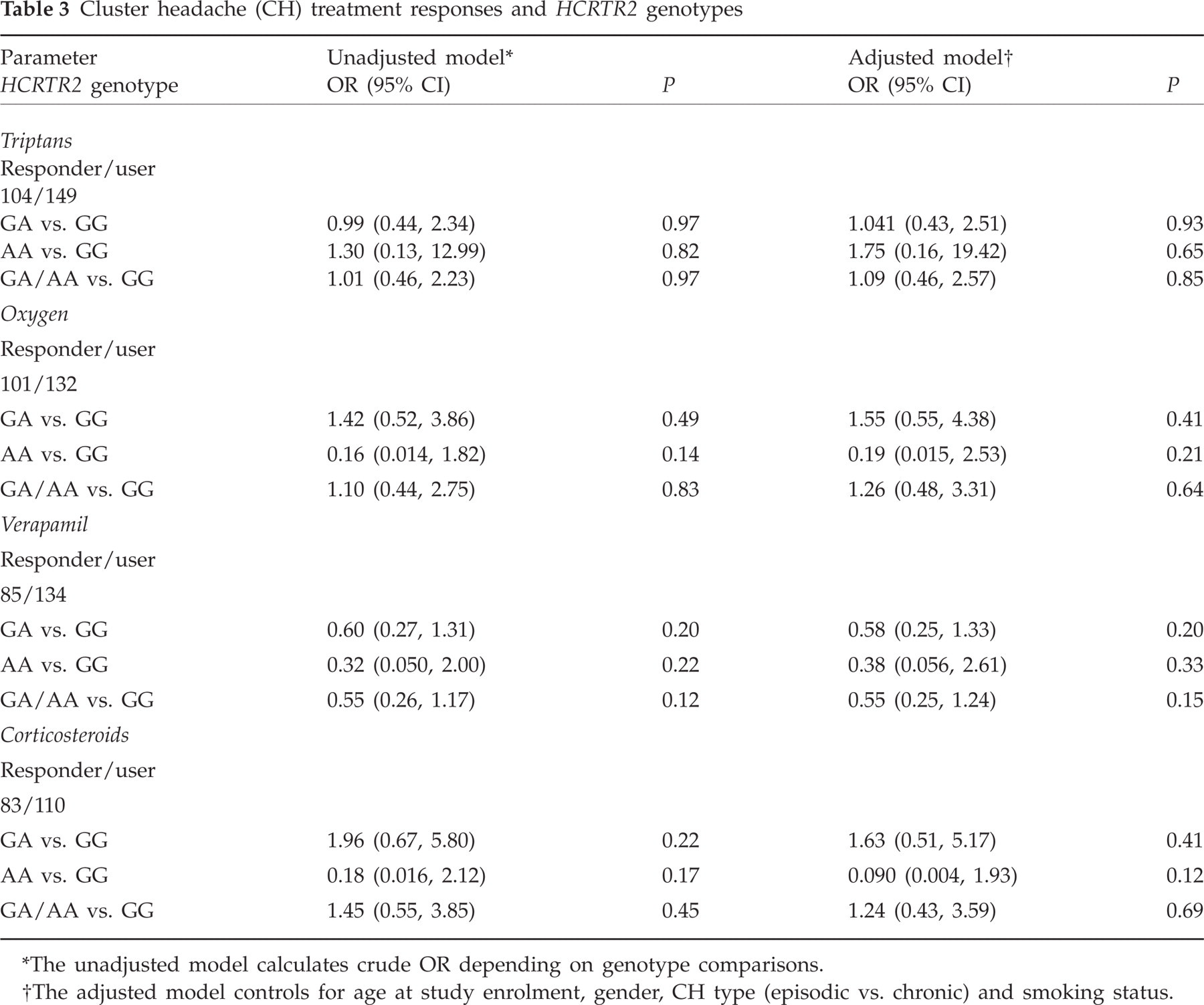
The unadjusted model calculates crude OR depending on genotype comparisons.
The adjusted model controls for age at study enrolment, gender, CH type (episodic vs. chronic) and smoking status.
Discussion
In this large sample of 184 CH patients, we observed no association between the HCRTR2 G1246A polymorphism and responses to first-line CH medications. The genotype distribution did not differ between responders and non-responders to triptans, oxygen, verapamil and corticosteroids.
A major problem of CH treatment in clinical practice is the limited efficacy of first-line medication, leaving 20–30% of patients with inadequate pain relief. The reason for this lack of effect is currently unclear; potential reasons include, among others, an incomplete treatment response because of pharmacogenetic factors, which could arise from genetic differences in drug metabolism, drug transport, nociception or pain modulation. A detailed understanding of CH pharmacogenetics could ultimately result in improved algorithms for differential treatment and dose adjustments in CH. However, the goals of highly individualized treatment regimes for CH patients are not easily met with our limited knowledge of the molecular or neuroanatomical structures, which are essential to convey acute and sustained pain relief in CH patients.
The hypocretin system has been associated with CH (13, 14) and, furthermore, shown to be involved in pain modulation. Available evidence suggests that microinjection of hypocretin 1 into the posterior hypothalamus diminishes nociceptive activity in trigeminal nuclei, while hypocretin 2 injection leads to an increased nociceptive response (22). In addition, dural vasodilation generated by electrical stimulation of dural afferents is inhibited by hypocretin 1 (23). Hypocretin 1 binds with equal affinities to the hypocrectin receptor isoforms 1 and 2, while hypocretin 2 has a considerably higher affinity for the receptor isoform 2 (12). The HCRTR2 G1246A polymorphism translates into an isoleucine for valine substitution at position 308. Whether such a substitution leads to an altered affinity for the two hypocretin isoforms, however, requires further experimental analysis. A refined genetic analysis of the HCRTR2 locus is hampered by its huge size, comprising more than 100 kbp. Based on our results, it is tempting to speculate that alterations in the hypocretin system attributable to the HCRTR2 G1246A polymorphism may predominantly affect trigger mechanisms and rhythmicity control in CH, but are less likely to influence pain modulation. However, in that case an effect on the efficacy of prophylactic medication might also be expected. Thus, future studies should focus on an advanced understanding of the neurobiology and genetics of the hypocretin system.
The strengths of our study include the large sample size, IHS-confirmed CH diagnoses by an expert neurologist and the standardized assessment of data, making admixture of other headache entities unlikely. Limitations of our study include potential misclassification of self-reported CH-associated features and treatment responses. However, responder rates in our cohort of CH patients were in accordance with previous reports (2, 6–8). Furthermore, because of the uniform and excruciating pain occurring periodically, patients are highly motivated to find effective treatment options to treat and prevent subsequent CH attacks. Despite this and perhaps other limitations, we believe that our data do not support an involvement of the HCRTR2 G1246A polymorphism in treatment response of CH.
Acknowledgements
The authors thank the Cluster-Kopfschmerz Selbsthilfe Gruppen Deutschland (CSG) for their support and all patients who participated in this study.
