Abstract
Psychologically based interventions such as relaxation training, biofeedback and cognitive-behavioural therapy are increasingly discussed as options for the treatment of migraine and tension-type headache in children and adolescents. In order to determine the state of evidence regarding the efficacy of these treatments, a meta-analysis of randomized controlled studies was conducted. In a comprehensive literature search including data from 1966 to 2004, 23 studies were found meeting the inclusion criteria. Due to the application of the random effects model, generalization of the results is possible. Specific statistical procedures were used to account for a possible publication bias. Significantly more patients improved to a clinically relevant extent (headache reduction ≥50%) in treatment conditions compared with waiting list conditions (high effect sizes). Long-term stability was also confirmed. The analysed treatments lead to improvement (up to 1 year) in headache status in children and adolescents with primary headache. However, more well-designed studies are needed to support and consolidate the conclusions of this meta-analysis and to compare the effects of psychological treatment with those of prophylactic medical interventions (in migraine), to examine potential differences between treatments, to identify moderators of efficacy and to determine effects of treatment on other health- related variables such as quality of life.
Keywords
Introduction
Headache in childhood and adolescence is an almost ubiquitous phenomenon. According to a recent epidemiological study in Germany of more than 5400 children and adolescents, 65% of children at the age of 14 years have experienced headache and 10.5% regularly suffer from headache at least once per week (1). Several other studies have yielded similar results (2–5). Sillanpää and Anttila (6) reported a significant increase in the prevalence of paediatric headache in Finland over two decades. The negative consequences of headache in childhood and adolescence are often underestimated. Long-term consequences can include impairment of social interaction and deterioration of functioning in school, including increased absence from school, a greater amount of school problems, more time needed to finish homework and more self-reported exhaustion after school (7–10). Anxiety and depression can also sometimes be heightened, which could be interpreted as a consequence of suffering from recurrent pain (11). Thus, headache can substantially reduce a child's quality of life (12).
These findings underline the need for effective treatments of paediatric and juvenile headaches. The two main approaches to treating headache are pharmacological and psychological therapy. In light of the drawbacks of frequent medication (13) and the lack of knowledge regarding its effects on children (14), psychological treatments have gained increasing attention. Relaxation techniques (including progressive muscle relaxation, autogenic training and self hypnosis), biofeedback (EMG feedback, hand temperature feedback, vasomotor feedback, neuro-feedback) and cognitive-behavioural interventions are the most frequently examined interventions. Two recent meta-analyses (15, 16) (see also (17)) on the treatment of migraine and tension-type headache have corroborated the efficacy of these psychological interventions for adults.
Although a number of published reviews have documented the effectiveness of psychological treatment for paediatric and juvenile headache (e.g. (18–20)), only two meta-analyses have been conducted so far (21, 22). The analysis of Hermann et al. (21) does not include more recent studies and can be criticized for lacking transparency regarding methodology. Moreover, some of the sophisticated methods allowing for the correction of biases in the estimation of effect sizes were not available at that time, which may have led to unrealistically high effect sizes in that review. The authors limited their analysis to paediatric migraine by excluding treatment studies of tension-type headache.
The more recent study by Eccleston et al. (22) used advanced methods for meta-analysis but included treatment studies on different types of paediatric pain (e.g. recurrent abdominal pain). Furthermore, only a responder analysis was carried out and the outcome regarding the extent of symptom reduction in specific headache variables (e.g. intensity, frequency, duration) was left unexplored.
The purpose of the current study is to describe the up-to-date state of evidence in the treatment of paediatric headaches using only randomized controlled trials (RCTs) and including studies conducted and published up to 2004. Furthermore, methodologically advanced strategies for meta-analysis were employed. Thus, estimation of effect sizes took into account all possible sources of information (from means and SDs to significance levels). Also, the statistically advanced random effects model was used, which allows generalization of results. The trim-and-fill method used in the analysis offers a mode for adjustment of effect sizes for publication bias.
Methods
Inclusion criteria
To be included, studies had to fulfil the following criteria regarding design, objective, and presentation of the study, participants, type of intervention and type of outcome measure.
The study had to be a RCT (allowing for randomization after matching procedures regarding specific subject characteristics, e.g. symptom severity). Trials had to incorporate psychological treatments aimed at preventing the occurrence of migraine or tension-type headache episodes and/or at reducing their intensity and duration. The control condition was to include waiting list or therapy placebo (active control group). Therapy placebo was defined as realizing unspecific ‘treatment’ factors common to all psychological therapies. Studies with a comparative design were excluded.
All subjects (including those in the control groups) had to suffer from at least two headache attacks per month before starting treatment.
Studies had to be available in English or German.
Study participants had to be children or adolescents in their majority, i.e. the average age of the sample had to be <18 years.
Studies had to examine one or more of the following types of treatment: relaxation training, biofeedback, (cognitive-) behavioural therapy, or combinations of these interventions.
The number of subjects in each treatment and/or control group had to be n ≥ 4 [based on bias estimation, see (23)].
Sufficient data had to be available to calculate effect sizes regarding headache frequency, duration, intensity and/or medication. Effect size calculation was based on means and SDs, t- or F-values, change scores, or other statistical scores (frequencies or significance levels).
Medication (number of days analgesics or antimigraine medication were taken) was also explored. Analysis was restricted to headache variables and medication since no other outcome measures were assessed in a sufficient number of studies to qualify for meta-analysis.
Coding schemes comprising methodological and clinical aspects of the original studies (Table 1) were conducted by the first and second author. The coding form had an overall interrater reliability of κ= 0.73, with a minimum of 0.70 regardingvariables which were used for effect size calculation and description of the studies.
Studies included in the meta-analysis


PMR, Progressive muscle relaxation; BFB, biofeedback; CBT, cognitive-behavioural therapy; AC, active control group; WLC, waiting list control group.
Search strategy
Studies were located through a systematic search of the following databases: Medline, PsycInfo, Psyndex, and the Cochrane Library. Searches were carried out from the first available year to July 2004 using combinations of the keywords ‘headache’, ‘migraine’, ‘tension headache’, ‘(psycho)therapy’, ‘treatment’, ‘biofeedback’, ‘relaxation’, ‘cognitive’, ‘behavioural’, ‘paediatric’, ‘juvenile’, ‘children’ and ‘adolescent’. Additionally, a literature search was done in secondary sources (reviews and previous meta-analyses) and on the internet. Effort was made to locate unpublished work by contacting researchers in order to reduce the file-drawer problem. Of more than 30 authors contacted, only two answered.
On the basis of the evaluated abstracts 43 outcome studies on the treatment of paediatric headache employing a control group were identified as potential candidates for the meta-analysis. Twenty studies were excluded for the following reasons: six studies did not include sufficient information for the calculation of effect sizes; four included only one treatment group and no control group; five used control or treatment conditions which did not fulfil the inclusion criteria of at least two headache attacks per month (criterion 2); in two studies assignment of subjects to conditions was not randomized; one study reported on treatments with fewer than four subjects; one included patients with different pain syndromes; and one study recapitulated data of a study already included in our analysis. The 23 original studies (Table 1) which finally entered the meta-analysis comprise a sample of 935 children and adolescents in total (see Table 2 for study characteristics).
Characteristics of studies included in the meta-analysis

Study subjects with migraine, tension-type headache and/or both and mixed headache (no diagnoses of migraine or tension-type headache, but recurrent headache).
Some studies investigated more than one intervention. In those cases, each of the interventions have been included separately.
Statistical analysis
Computation of effect sizes was accomplished using Hedges' g (24, 25). The only difference of g from Cohen's d is the use of the sample standard deviation (26). If possible, the calculation of Hedges' g was based on means and SDs. However, some studies (n = 6) indicated results only by description of probability levels. For these studies effect sizes were inferred from the reported significance levels (non-significant results were set to g = 0). Odds ratios were computed for dichotomous data and subsequently transformed into Hedges' g (27).
A correction formula to account for small sample bias was also applied (24). Effect sizes were calculated for the following outcome categories: headache symptom variables (frequency, intensity and duration of headache or a comprehensive headache index), medication, and clinical significant change (reduction of headache symptoms ≥50% based on the headache index or any of the above-mentioned variables). If more than one measure was used in one outcome category, a mean g was calculated by averaging the corresponding effect sizes.
Before computing the average effect sizes, the scores of each single study were weighted by the reciprocal of the variance components, and a random effects analysis was carried out.
The random effects model (REM) is preferable to the fixed effect model (FEM) because interpretations of results are not limited to the actually analysed studies, but can be generalized to the universe of potential studies on treatment efficacy in the analysed domain of research: psychologically based interventions like relaxation, biofeedback and (cognitive-) behavioural treatments in comparable children and adolescents with recurrent headache. In contrast to the often used FEM, a variance component is included (the random effects variance) to account for the variance resulting from drawing studies from the population of all possible studies in the given domain of research. For a more comprehensive description regarding the advantages of the REM see Mitte (28).
The analyses examined two different aspects of outcome: between-group effects (comparing treatment and control groups) and within-group effects (comparing different assessment periods within treatment and control groups). No analyses could be conducted for between-group effects at follow-up and within-group effects for wait list control at pre-follow-up and post-follow-up comparisons, since they were treated after the waiting period and no follow-up measures were available.
As a consequence of the standardization of effect size polarity, a positive score indicates a better outcome in the treatment condition and an improvement in pre-post-, pre-follow-up and post-follow-up comparisons.
The trim-and-fill method (29) was applied to account for a possible publication bias. This analysis checked the robustness of the results by taking into account that insignificant effects are less likely to be published (30). In systematic reviews and meta-analyses such studies are thus less likely to be identified. The trim-and-fill method offers an estimation of missing studies and their effect sizes and consequently an estimation of ‘true’ treatment effects.
The computations were accomplished with the statistical software Study Input 2.08 (31).
Results
Descriptive characteristics of the studies
Most studies were conducted in North America (n = 12), the remainder in Europe (n = 10) and Australia (n = 1) (Table 2). Eighteen studies (78%) used randomization for the assignment of subjects to the treatment conditions without a matching procedure. The most frequently examined treatment was relaxation training (n = 16) with four studies adding behavioural components to it (pain or stress management techniques, Tables 1 and 2). Ten trials analyse (cognitive-) behavioural therapy and seven biofeedback. Fifteen studies evaluated individual training formats, four examined group trainings and four both types. Sixteen studies had implemented a waiting list control group (continuously monitoring their headaches by means of a diary).
The selection criteria for treatment inclusion in the original studies varied (e.g. ≥2 migraine attacks per month; ≥1 attack per week). In 10 studies only children with migraine, in one study only subjects with tension-type headache were treated, 12 studies included subjects with tension-type headache, migraine or both diagnoses. Twenty-one trials comprised a follow-up, which varied in length from 4 to 12 months. The average age of the participants was 12 years (data available from 19 studies), 64% were female. On average 11% of the sample dropped out (data available from 20 studies), which can be considered to be within the normal range or even below (28). Headache symptoms and medication (n = 6) were always assessed by diaries. Two trials did not allow medication to be taken by the participants, three gave no information about it. In nine trials medication was allowed, but not assessed. In three studies allowing medication, no data were reported to calculate effect sizes.
When conducting meta-analyses many decisions on procedures have to be made. It is therefore recommended to examine whether alternative decisions would have been likely to influence the results. This was done by two sensitivity analyses. Five of the 23 primary studies reported on non-clinical, school-based interventions and were included in the original analysis. The first sensitivity analysis excluded these studies and effect sizes were compared with that of the comprehensive analysis. Also the consequence of the use of REM was examined by concurrently calculating the fixed effect model. The assumed advantage of REM is attained at the cost of a lower statistical power in case of a small study sample (23). The Q-test for homogeneity of effect sizes was conducted with P ≤ 0.10 as a level of significance to compensate for the low power of the test (32). When the Q-test is non-significant, REM and FEM yield comparable results for mean effect size and random-effects variance is not significantly different from zero.
Between-group effects (post-treatment measures)
Only 10 of the 23 selected original studies allowed for a between-group comparison (treatment vs. control) regarding headache symptoms in the post-therapy period (Table 3, Fig. 1). Children and adolescents from 16 treatment/control groups were included (n = 407). The mean effect size regarding all headache variables is small (g = 0.35). Table 4 gives detailed information on the specific headache variables: frequency, duration and intensity. These analyses show that only intensity of headache is significantly affected by treatment (Table 4). Excluding the non-clinical, school-based interventions, comparable effects are found [n = 8; REM: g = 0.36, 95% confidence interval (CI) 0.03, 0.69, τ2 = 0; FEM: g = 0.36, 95% CI 0.10, 0.63]. Medication intake (Table 3) is not significantly modified by treatment (CI including zero). The analysis of clinically significant change is based on the largest sample (16 studies, 22 groups, 587 subjects) and displays a large effect size (g = 0.87) with a small positive, but non-significant, random effects variance (Fig. 2). Comparable results can be demonstrated when excluding the five non-clinical school-based interventions (n = 11, REM: g = 0.80, 95% CI 0.50, 1.08, τ2 = 0; FEM: g = 0.80, 95% CI 0.54, 1.05).

Study effect sizes (between groups) and confidence intervals in the outcome category ‘headache variables’ (post-treatment, n = 10).

Study effect sizes (between groups) and confidence intervals in the outcome category ‘clinically significant change’ (post-treatment, n = 16).
Effect sizes for outcome categories: headache variables, medication, and clinically significant change

FEM, Fixed effects model; g, effect size/Hedges' g; 95% CI, 95% confidence interval; REM, random effects model; τ2, random effects variance; Q, χ2 (all Q-tests were non-significant); N, number of effect sizes.
Effect sizes for headache variables frequency, duration and intensity
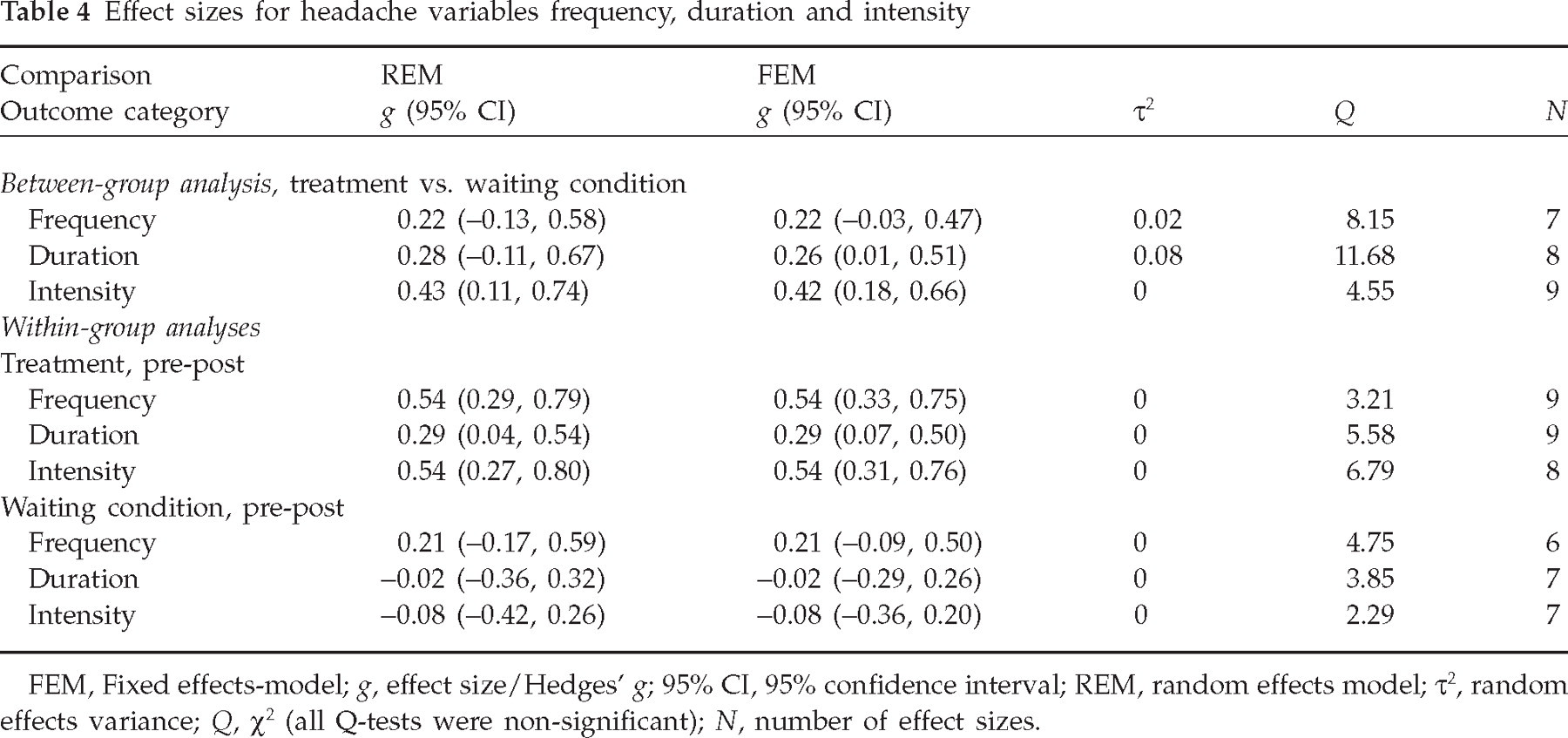
FEM, Fixed effects-model; g, effect size/Hedges' g; 95% CI, 95% confidence interval; REM, random effects model; τ2, random effects variance; Q, χ2 (all Q-tests were non-significant); N, number of effect sizes.
Additionally, binomial effect size displays (BESD) for clinically significant change and headache variables were calculated, after converting Hedges' g to r (68). The BESD represents the difference in outcome rates between treatment and control group (33). Success rates of 70% for the treatment condition and 30% for the control group at post treatment were found for clinically significant change. For headache variables a success rate of 60% for psychological treatment was found, whereas the control condition showed a rate of only 40%.
Only four trials included an active control group design, with only two of them using comparable outcome variables, so that no meta-analysis could be conducted. The comparison of intervention and active control groups at post therapy displays the following effect sizes: headache variables (two studies): g 1 =−0.06; σ2 = 0.11/g 2 = 0.10, σ2 = 0.15; clinical significance (two studies): g 1 = 1.33, σ2 = 0.20/g 2 = 0.66, σ2 = 0.20. Thus no differences between treatment and active control were found regarding headache symptom variables, medium to large effect sizes were found, however, regarding the number of responders (clinically significant change).
Within-group effects
Regarding the difference between outcome variables assessed before and directly after therapy (pre-post), 12 studies with 23 groups including 322 children and adolescents entered the analyses (Fig. 3). Both REM and FEM show moderate mean effects regarding headache variables (g = 0.55) in the treatment conditions. Excluding the non-clinical, school-based interventions, similar effects are found (n = 11, REM: g = 0.55, 95% CI 0.31, 0.80, τ2 = 0; FEM: g = 0.55, 95% CI 0.34, 0.77).

Pre-post study (within groups) effect sizes and confidence intervals for headache variables (n = 12).
Frequency and intensity of headache (Table 4) display moderate mean effects and duration a small effect. No improvement is seen in the waiting list control groups (n = 8, g = 0.10, see Table 3). Regarding medication, only four studies with six groups could be analysed, demonstrating a moderate average effect for FEM in the treatment condition. However, in REM the effect size is not significant (Table 3). The waiting list condition does not achieve a significant pre-post change in medication (n = 4, g = 0; see Table 3). No information was available for medication in the active control conditions. Two of these studies, however, presented information on headache variables with moderate to large effect sizes (g 1 = 0.80; σ2 = 0.09/g 2 = 0.49, σ2 = 0.18).
Long-term efficacy was determined by analysing pre-follow-up differences. Eight studies included follow-up data on headache variables for the treatment groups (three studies with a 12-month follow-up, three studies with a 6-month follow-up, one study each with 4-month and 8-month follow-up). A small positive random effects variance did not reach significance (Table 3). Large mean effect sizes were calculated for the pre-follow-up changes in REM (g = 1.0) and FEM (g = 0.97). Only two studies provided information necessary for pre-follow-up comparison for medication (g 1 = 0.36; σ2 = 0.21/g 2 = 0.47, σ2 = 0.19); no REM or FEM could be computed.
Improvement after post-therapy assessment
Examination of a possible gain in symptom reduction after the post-therapy assessment period was attempted by computation of post-follow-up effect sizes for the treatment conditions. Two analyses were conducted. Follow-up periods, 1–6 months and 6–12 months, were analysed separately regarding headache variables and clinically significant change. If a study included more than one follow-up period an average effect size was calculated (Table 3). Only two studies provide data on medication in the follow-up period, so no REM or FEM could be calculated.
From 20 studies presenting long-term results, only 16 provide information for these analyses. The positive mean effect sizes indicate small improvements from post-therapy to the 1–6-month follow-up assessments regarding headache variables (g = 0.36, n = 6) and clinically significant change (g = 0.27, n = 11), but the latter fails to reach significance. For 6–12-month follow-up assessments, improvement in headache symptoms also descriptively, but not significantly increases, indicated by a small effect size and a CI including zero (g = 0.26, n = 4). For clinically significant change no REM and FEM could be calculated at the 6–12-month follow-up, since only one original study (34) provided information (g = 0.2, σ2 = 0.16).
Differential analyses (types of intervention, type of headache)
To examine possible differential effects regarding types of intervention, separate between-group analyses were calculated. REM and FEM regarding headache variables could be calculated only for relaxation treatments and revealed a small, but non-significant mean effect (n = 4, REM: g = 0.20, 95% CI 0.42, 0.77, τ2 = 0; FEM: g = 0.20, 95% CI −0.20, 0.54). Relaxation and biofeedback treatments both display large effect sizes when clinically significant change is used as outcome criterion (relaxation treatments, n = 7; REM: g = 0.80, 95% CI 0.23, 1.35, τ2 = 0.14, NS; FEM: g = 0.80, 95% CI 0.45, 1.11; biofeedback, n = 4; REM: g = 0.90, 95% CI 0.10, 1.68, τ2 = 0; FEM: g = 0.90, 95% CI 0.40, 1.38). A within-group comparison of headache variables was possible only regarding relaxation treatments. A small and non-significant mean effect size were found (n = 5; REM: g = 0.28, 95% CI −0.15, 0.72, τ2 = 0; FEM: g = 0.28, 95% CI −0.02, 0.59).
Only four studies allow direct comparison of different treatment approaches. Three studies compared relaxation combined with biofeedback vs. relaxation training alone (headache variables: g 1 = 0, σ2 = 0.21; g 2 = 0.5, σ2 = 0.10; g 3 = 0.25, σ2 = 0.20; clinically significant change: g 1 = 0.20, σ2 = 0.20; g 2 = 0.34, σ2 = 0.11; g 3 = 0, σ2 = 0.35). Only one study compared biofeedback with cognitive-behavioural treatment vs. relaxation with cognitive-behavioural treatment (headache variables: g = 0.6, σ2 = 0.14; clinically significant change: g = 0.25, σ2 = 0.19). No REMs or FEMs could be calculated.
In a further analysis, REM and FEM were separately computed for patients suffering from migraine (between-group comparison). A large effect size is displayed in the outcome variable clinically significant change (n = 5, REM: g = 1.22, 95% CI 0.61, 1.84, τ2 = 0; FEM: g = 1.22, 95% CI 0.78, 1.65). No REM and FEM were computed for headache variables and medication, and only two studies provided this information. Headache variables: g 1 = 0.62, σ2 = 0.22; g 2 = 0.12, σ2 = 0.11; medication: g 1 = 0.15, σ2 = 0.20; g 2 = 0.21, σ2 = 0.11.
The within-group analysis for migraine patients also results in a significant but moderate effect size regarding headache variables (n = 4; REM: g = 0.68, 95% CI 0.10, 1.30, τ2 = 0; FEM: g = 0.68, 95% CI 0.30, 1.06). No REM or FEM could be computed for medication, since only one study presented relevant data (g = 0.21, σ2 = 0.20). Only one study was dedicated to the treatment of tension headache, thus no REM or FEM could be determined.
Publication bias
To check for publication bias, the robustness of the results was evaluated (30). After trim-and-fill analyses (between-group comparison) had been conducted, estimated effect sizes were only marginally smaller than those calculated on the basis of the original data (headache variables, REM: g = 0.23, 95% CI −0.03, 0.50, τ2 = 0; FEM: g = 0.23, 95% CI 0, 0.45, χ2 = 7.89; clinical significance, REM: g = 0.76, 95% CI 0.50, 1.02, τ2 = 0.04; FEM: g = 0.76, 95% CI 0.55, 0.96, χ2 = 19.95).
When the trim-and-fill analysis was applied to the within-group effect scores (pre-post), the corrected means were reduced from moderate to small effect sizes (headache variables, REM: g = 0.40, 95% CI 0.17, 0.61, τ2 = 0.04; FEM: g = 0.40, 95% CI 0.20, 0.60, χ2 = 5.61).
Discussion
The analysis presented here provides an up-to-date estimate of the evidence regarding psychologically based interventions for children and adolescents with recurrent headache based on a total of 23 studies. The results have specific validity for the treatment of headaches, both tension-type and migraine, since studies including the treatment of other pain syndromes were excluded. For the outcome criterion ‘clinically significant change’, large effects sizes are observed regarding between-group comparisons at post-treatment. Thus, it can be concluded that taking the percentage of responders into account, psychological treatment demonstrates greater efficacy when compared with a control. The parametric outcome variables used (headache intensity, duration, frequency, headache activity index), however, yield markedly smaller effect sizes in the between-group analysis.
The within-group effect sizes reflecting the change in headache in the patients over different assessment periods point to a significant, but only moderate, treatment effect. They are, however, distinctly larger in the treatment than in the control groups. The differences between effect sizes regarding headache symptom variables and the criterion clinically significant change are in need of interpretation. Treatment obviously increases the number of responders considerably (headache reduction ≥50%), whereas the waiting list self-monitoring procedure in the control groups stimulates small but no marked improvements in the participants. Thus a subject in the control condition may reduce their headache frequency by 20%, but will not reach the criterion of clinically significant change. It can be argued that the considerable amount of self-monitoring induced by diary keeping may have led to changes in symptom-provoking behaviour (35) resulting in some decrease of headache.
Within-groups effect sizes document a trend towards increasing improvement at follow-up. It is thus safe to say that improvement experienced after therapy is not lost, but at least maintained up to 12 months after the end of treatment. Since the outcome criteria clinically significant change and headache variables could not be determined in a between-group analysis at follow-up, a further corroboration of this finding is not possible. It can be assumed that the practising of coping strategies aimed at the prevention of headache attacks acquired during therapy and applied in daily life leads to a stable decrease of headache.
Separate analyses of intensity, frequency and duration of headache reveal differences regarding sensitivity to change. Between-group analyses demonstrate a significant effect of treatment only for the intensity of headache. Within-group analyses indicate significant improvement also for frequency and duration of headache. The assumption of a significant decrease in medication parallel to symptom decrease could not be statistically supported. However, only six studies contributed to this finding, making it less robust. Furthermore, the studies used different, and sometimes not clearly defined, definitions of medication. Further studies examining the influence of psychological treatment on medication intake are therefore needed.
Because of the relatively small number of RCTs found, the range of differential analyses was limited. Separate analyses for children with migraine (n = 9) could be performed and supported the general conclusions regarding treatment efficacy. Tension-type headache, however, could not be analysed separately. Separate analyses for younger and older participants were also not feasible. Neither could comparative analyses of different treatment approaches be conducted.
Since REM rather then FEM (sensitivity analysis shows small differences to the REM) was chosen for the analysis and interpretation of data, the conclusions derived from the analyses can be assumed to be very robust with a strong potential for generalization. However, it has to be acknowledged that our results may be not generalizable to the treatment of children and adolescents from countries not represented by a trial in our study sample, such as South America, Asia or Africa.
The existence of moderators of efficacy, e.g. conducting treatment in a clinical or non-clinical setting, is not supported by the data (non-significant Q-tests), thus giving another reason for generalizing the findings.
The trim-and-fill analysis correcting for bias due to unpublished studies supports the assumption that the results reflect the ‘true’ state of evidence, although corrected pre-post effect sizes regarding headache symptoms are somewhat lower than the original mean effect sizes.
Despite the relatively large number of initially identified original studies, only a small number of trials remained in the analysis. This reflects a scarcity of methodologically adequate data evaluation. Personally contacting the authors did not lead to a significant enrichment of available data.
Only a few of the original studies examined the efficacy of psychological treatment in comparison to an active control group which controls for non-specific effects of giving attention to the child's problem and allowing for positive social interaction. Therefore evidence for the efficacy of psychological treatment of paediatric headache so far is based only on the ‘soft’ criterion of being more effective than waiting for future treatment and monitoring one's headache in a diary.
Our meta-analytic approach did not include outcome measures such as subjective disability, anxiety, quality of life, depression, coping, self-efficacy or physiological measures, which are known to be associated with recurrent headache (36–43). Only nine studies assessed one or the other of these variables, so that a meta-analytic approach was not possible. It seems worthwhile to examine whether psychological treatment can positively affect these concomitant variables. A few studies gave the first evidence of the beneficial influence of headache treatment on stress coping, somatic complaints and self confidence (44–46). The findings of our study imply that future research should widen its focus to include further outcome parameters and correlates of headache improvement due to psychological treatment. In outcome studies of adult treatment of pain a broad range of variables, such as those mentioned above, are commonly assessed (47), which is also recommended for treatment of paediatric headache.
Some methodological weaknesses of the studies should be mentioned. There is a large variance in the age of the treated participants within the samples, even within one study (see Table 2). Patients have different or even unspecified diagnoses; the severity of headache also varies as well as allowing medication during treatment. However, if these differences are assumed to affect treatment outcome differentially, they should have led to a significant heterogeneity in the results of the analyses, which was not confirmed.
A major characteristic of the original studies (and thereby a possible limitation of the conclusions derived from this meta-analysis) is that only completer analyses were conducted, which may have lead to an overestimation of efficacy. Comparing our results with those of Hermann et al. (21), however, the difference in the general level of effects sizes is striking, with the average effect score being much lower in the current study. Although the origin of this discrepancy cannot be explained completely, some differences in methodology may have contributed: Hermann et al. (21) did not limit their analyses to RCTs, which means that they included methodologically weaker studies. However, the most important reason may be the use of rather liberal strategies for meta-analysis, whereas the present analysis utilized very conservative methods (e.g. small sample size correction, REM).
Our results, however, compare very well with the findings of Eccleston et al. (22). The authors used a responder criterion comparable to the one applied in our analysis. Their findings point to a high efficacy (number needed to treat = 2.3, clinically significant change), as does our mean effect size of g = 0.87 in clinically significant change at post-therapy assessment. The additionally calculated binomial effect size display depicts with a 70% success rate in the treatment groups (compared with 30% in the control groups) the satisfying level of efficacy and its clinical relevance.
Our general conclusion derived from the meta-analysis is that efficacy of psychological treatment in paediatric headache patients is corroborated on evidence level 1a (several consistent RCTs support efficacy (48)). Treatment success can at least be maintained up to 1 year but shows a trend even towards a further increase.
Various research questions still remain to be examined by studies with methodologically adequate designs. Can differences in improvement be expected between children with different diagnoses? Is there an interaction between diagnosis and treatment type? The mechanisms of change should also be explored by assessing further process and outcome parameters such as behavioural and cognitive coping, changes in dysfunctional cognition, self-efficacy and health-related attitudes and behaviour. Possible physiological mediators should also be explored. Furthermore, disability and emotional distress, main outcome variables in adult pain treatment, need to be included in future research. Disability should be assessed by adequate instruments for the behavioural (e.g. PedMIDAS, see (9)) and emotional levels (anxiety, depression). Quality of life could be the most comprehensive and meaningful index of improvement.
Also, comparative efficacy of pharmacological treatments should be explored, especially for migraine, where treatment recommendations for the use of preventive drug treatment exist. The combined effectiveness of psychological and medical interventions deserves examination.
Thus, future researchers can be reassured by the high probability of treatment success in applying psychological interventions, but they will have to contribute to the elucidation of mechanisms, differential effects and moderators of efficacy. They are advised to establish active control groups or use a comparative treatment design and to incorporate intent-to-treat analyses. A detailed overview of guidelines for the design of such clinically based investigations is given by Penzien et al. (49), who describe important areas of critical needs and priorities for headache research which are in concordance with the conclusions drawn here. Further data are needed to establish psychological intervention as a valid treatment option in clinical settings.
