Abstract
Sensitivity to sound and vertigo are often components of migraine. Recent studies suggest that plasma extravasation from intradural blood vessels may contribute to migraine pain. This study documented plasma extravasation in the mouse inner ear after intravenous administration of serotonin (5-HT). Horseradish peroxidase (HRP) was injected intravenously to trace protein extravasation in mice, followed 15 min later by intravenous 5-HT or saline. Forty-five minutes later, mice were euthanized. HRP extravasation was visualized immunohistochemically and quantified densitometrically. Baseline and evoked extravasation in stria vascularis and tectorial membrane were indistinguishable from skin, dura mater and tympanic membrane. Brain parenchyma, Scarpa's ganglion, basal spiral ganglion and modiolus, and the central vestibular nerve segment showed no significant 5-HT-induced extravasation. In contrast, 5-HT produced extravasation in the apical spiral ganglion, modiolus, and intralabyrinthine superior and inferior vestibular nerve. Thus, inner ear plasma extravasation is a potential mechanism for migraine-associated vertigo and sound sensitivity.
Introduction
Neurogenic vasodilation and extravasation from meningeal vessels are recognized as basic mechanisms in migraine (1, 2). These effects are mediated by trigeminovascular reflexes and release of neuropeptides from trigeminal afferent fibres, which contribute to a sterile inflammatory response in dura mater and perivascular tissues. This concept was established by Moskowitz and coworkers' (3–5) demonstration of plasma extravasation in dura mater (but not brain parenchyma) following chemical, electrical or immunological stimulation. Subsequent studies have indicated that similar extravasation may occur in tissues surrounding the spiral modiolar artery and its radiating arterioles (6–8).
Epidemiological studies have revealed that the prevalence of migraine is 15–18% in women and 5–8% in men (9, 10). Migraine accompanies several transient neurological deficits, typically sensitive to light and sound. There has also been a growing recognition of vestibular dysfunction associated with migraine (11). This association can be explained partly by a basilar artery migraine mechanism, based upon the finding that capsaicin and electrical stimulation of the trigeminal nerve can produce plasma extravasation from the basilar artery and anterior inferior cerebellar artery (7). This study uses a mouse neurogenic inflammation model to evaluate the extent of plasma protein extravasation in different tissues of the mouse inner ear.
Materials and methods
Injection procedure and tissue preparation
Experiments were performed in 15 adult mice (25–35 g) with normal Preyer's reflex. Animals were anaesthetized with an intraperitoneal injection of sodium pentobarbital (40 mg/kg). Fifty thousand units of a single basic isozyme of horseradish peroxidase (HRP, EIA grade, 521 Worthington units/mg; Worthington Biochemical Corp., Lakewood, NJ, USA) was diluted in 1 ml of 0.9% saline solution and 10 ml/kg was delivered via a tail vein, followed 15 min later by a tail vein injection of either 10−7
Visualization of HRP
Following incubation in formalin vapour for 20 min, sections were treated with 3% H2O2 solution for 10 min. Specimens were rinsed in three changes of distilled water. Following rinsing with phosphate-buffered saline (PBS), non-specific binding was blocked by incubation in PBS containing 2% bovine serum albumin and 0.05% Triton X-100. Sections were then incubated with biotin-SP-conjugated goat anti-HRP (Jackson Immunoresearch Labs Inc., West Grove, PA, USA), diluted at 1 : 200 in the blocking solution overnight at room temperature. After the slides were rinsed in PBS, the tissue was treated with Vectastain® ABC reagent (Vector Labs Inc., Burlingame, CA, USA) according to standard methods. The location of immunoreactive HRP was then visualized with a diaminobenzidine chromogen.
Regions of interest
Measurements were made in six different groups of structures and each structure was measured separately.
Barrier-property reference structures. Skin, dura mater and tympanic membrane were used as references for structures with relatively high levels of protein extravasation. The brain parenchyma was a reference for blood–brain barrier properties.
Membranous parts of the cochlea included the basilar membrane, Reissner's membrane, tectorial membrane and membranous interscalar septum.
Membranous part of the vestibular system.
Lateral wall of the cochlea. Sampled structures included the stria vascularis, spiral ligament and spiral limbus.
Neural structures in the cochlea included the spiral ganglion and nerve fibres in spiral lamina and modiolus. The apico-middle turn and basal turn were measured separately.
Neural structures in the vestibular system included Scarpa's ganglion and nerve fibres at the inside-labyrinthine part and outside-internal auditory canal part. The superior and inferior vestibular nerves and ganglia were evaluated separately.
Densitometry of HRP reaction product
Each region of interest was identified using an Olympus BH-2 microscope with a DplanApo® UV 10× objective which was coupled to a Cohu CCD video camera with a Diagnostic Instruments 0.6× objective. The density of each area was measured on a 486-based computer equipped with a Matrox IP-8/AT (Matrox Electronics Systems, Dorval, QC, Canada) video processing board running Optimus® software (Ver. 4.02; Bioscan, Edmonds, WA, USA). The density was measured after selecting the region of interest using a ‘mouse’. The range of measurements was 0 (brightest) to 256 (darkest). The system was calibrated with the same control slide at the beginning of each measurement session.
Hair cell regions are not included in the density measurements to avoid bias due to edge artefacts. Areas contaminated with melanin, such as vestibular dark cell regions, are excluded from densitometry measurements.
Statistical analysis
Analysis of variance (repeated measures on structures, between-groups measures on treatment), followed by Bonferroni-corrected t-tests, was used to test for differences in HRP reaction density across structures and treatment groups. Individual comparisons are described in the text. These tests were performed with
Results
Basic properties of the mouse model
The penetration of HRP into the brain, skin, dura mater and tympanic membrane (Fig. 1) of control (HRP alone) and 5-HT (after HRP)-treated animals illustrates several basic properties of the mouse model. First, there was no evidence of HRP immunoreactivity in mice that did not receive HRP injections. Second, extravasation of HRP occurred in the skin, dura mater and tympanic membrane of both control (Fig. 1a–c) and serotonin-injected (Fig. 1d–f) mice, but it was excluded by the intact blood–brain barrier. In the control animals, there was no evidence of HRP extravasation in the brain (Fig. 1b), but HRP immunoreactivity was prominent in the other tissues (Fig. 1a,c). Densitometric measurements indicated that skin (P < 0.005, Bonferroni-corrected t-test) and tympanic membrane (P < 0.05, Bonferroni-corrected t-test), but not dura mater, showed a significantly denser HRP reaction than the brain in control mice (Fig. 2a). In the 5-HT-treated mice, the skin, tympanic membrane and dura mater also showed a significantly denser HRP reaction than the brain (P < 0.001, Bonferroni-corrected t-test, Fig. 2a). Third, 5-HT administration did not significantly affect extravasation of HRP into the brain (NS, Bonferroni-corrected t-test). Fourth, the 5-HT-treated mice showed an apparent augmentation of HRP immunoreactivity in histological sections of the skin (Fig. 1d), dura and pia mater (Fig. 1e) and tympanic membrane (Fig. 1f), without any change in the appearance of reactivity in the brain parenchyma (Fig. 1d). The distribution of the reaction was similar to the control animals in the skin and the dura mater. In the tympanic membrane, however, HRP distribution changed from dense staining of internal and external surfaces in the control group (Fig. 1c) to a homogeneous distribution after 5-HT administration. Analysis of variance (

Horseradish peroxidase immunoreactivity of reference structures. The dura showed denser staining compared with the brain parenchyma and the difference between two structures is greater in 5-HT-injected animals (e) than in controls (b). The skin (d) and tympanic membrane (f) of the 5-HT group also showed denser staining than control skin (a) and the tympanic membrane (c). Bar: 50 µm.

Comparison of the optical density of the reference structures between groups. (a) In control mice, the optical density of the skin and tympanic membrane was significantly higher than that of the brain. In 5-HT-treated mice, the optical density of the dura, skin and tympanic membrane was significantly higher than that of the brain parenchyma. Horseradish peroxidase immunoreactivity was augmented significantly by 5-HT treatment in the dura (P < 0.05) and the tympanic membrane (P < 0.05), without any change in the brain parenchyma. The skin showed borderline significance by 5-HT treatment (P = 0.09). (b) The correlations of optical densities were highly significant between the skin and dura mater (r = 0.80), the skin and tympanic membrane (r = 0.86) and dura mater and tympanic membrane (r = 0.95), but there was no significant correlation between the brain and other tissues.
HRP extravasation in the inner ear and the eighth nerve
The stria vascularis showed extravasation of HRP comparable to that in the tympanic membrane and dura mater after 5-HT administration. In histological sections, the stria vascularis of the control mice showed a relatively uniform light reaction, with occasional granular staining in cells in the intermediate layer; a lower intensity of staining was observed in the underlying spiral ligament (Fig. 3b). After 5-HT administration, there was a uniformly high intensity of HRP immunoreactivity throughout the marginal cells and intermediate layers of the stria vascularis (Fig. 3e), demarcated starkly by the lack of immunoreactivity in the basal layer and the underlying connective tissue membrane (the classical light microscopic ‘basement membrane’). Densitometric data confirmed that the HRP reaction density in the 5-HT group was significantly elevated from the control group (Fig. 5a, P < 0.01, Bonferroni- and Sidak-corrected t-tests). There was also a dense HRP reaction on the surface of the tectorial membrane in the 5-HT-treated mice (Figs 3d and 5a), which was suggestive of binding of extravasated protein from the endolymph.

Horseradish peroxidase (HRP) immunoreactivity in the cochlear structure between the control (a–c) and 5-HT-treated group (d–f). Tectorial membrane and stria vascularis show higher immunoreactivity in the 5-HT-treated group. Nerve fibres and spiral ganglion cells show increased cellular uptake of HRP and staining in the interstitial space (f). Bar: 50 µm.
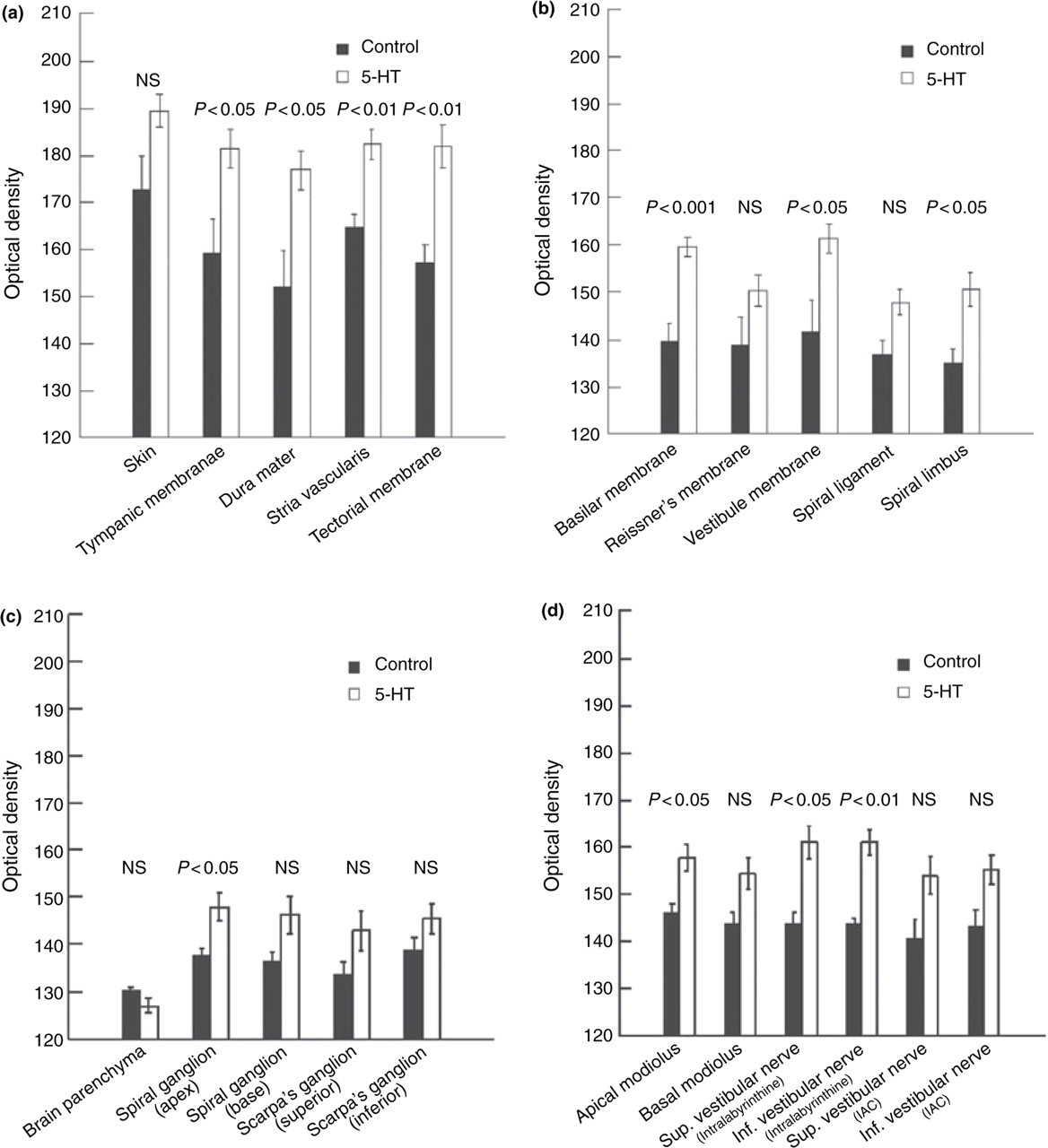
Comparison of the optical density between groups. (a) Optical densities of stria vascularis and tectorial membrane were comparable to those of the tympanic membrane and dura mater in control mice and were significantly elevated in the 5-HT group. (b) Other membranous structures in the inner ear showed intermediate horseradish peroxidase (HRP) immunoreactivity between dura mater and brain in control mice. In the 5-HT group, HRP extravasation was significantly elevated in the basilar membrane, vestibular membrane and spiral limbus (P < 0.05). (c) Spiral ganglion and Scarpa's ganglion showed higher HRP immunoreactivity than brain parenchyma in control mice. The increased immunoreactivity in the 5-HT group was significant in the apical part of the spiral ganglion. (d) Optical density increased significantly around nerve fibres in the apical modiolar and intralabyrinthine segment of the superior and inferior vestibular nerve.
The spiral ligament, basilar membrane and spiral limbus are components of a single structural complex that forms the floor of the scala media. Although the basilar membrane is largely avascular, a spiral capillary vessel lies on its tympanic surface near its junction with the spiral limbus (12). In control mice (Fig. 5b), the light HRP immunoreactivity in these regions was intermediate between the density in brain and that in the dura matter, but did not differ significantly from either of these structures (Bonferroni- and Sidak-corrected t-tests). After 5-HT administration, however, there were modest but significant homogeneous increases in the density of HRP immunoreaction in the spiral limbus and basilar membrane (Figs 3e and 5b), but no significant difference in the spiral ligament (Bonferroni- and Sidak-corrected t-tests). The HRP reaction density in these structures after 5-HT treatment was similar to the baseline extravasation of HRP in the control tympanic membrane, stria vascularis and dura mater (Fig. 5a,b).
The spiral ganglion and Scarpa's ganglion showed little extravasation in control animals (Figs 3c and 4a–c). After 5-HT treatment, there was evidence of both cellular uptake of HRP and diffuse staining in the interstitial space (Figs 3f and 4d–f). The densitometric measurements of ganglia from control mice were higher than brain parenchyma (Fig. 5c). After 5-HT administration, HRP immunoreactivity increased significantly in the apical part of the spiral ganglion (Fig. 5c). The interstitial space surrounding nerve fibres showed a density similar to the basilar membrane, Reissner's membrane and membranous labyrinth of the vestibular system. The density of the HRP reaction increased significantly around nerve fibres in the apical modiolus and intralabyrinthine segments of the superior and inferior vestibular nerves (Fig. 5d).
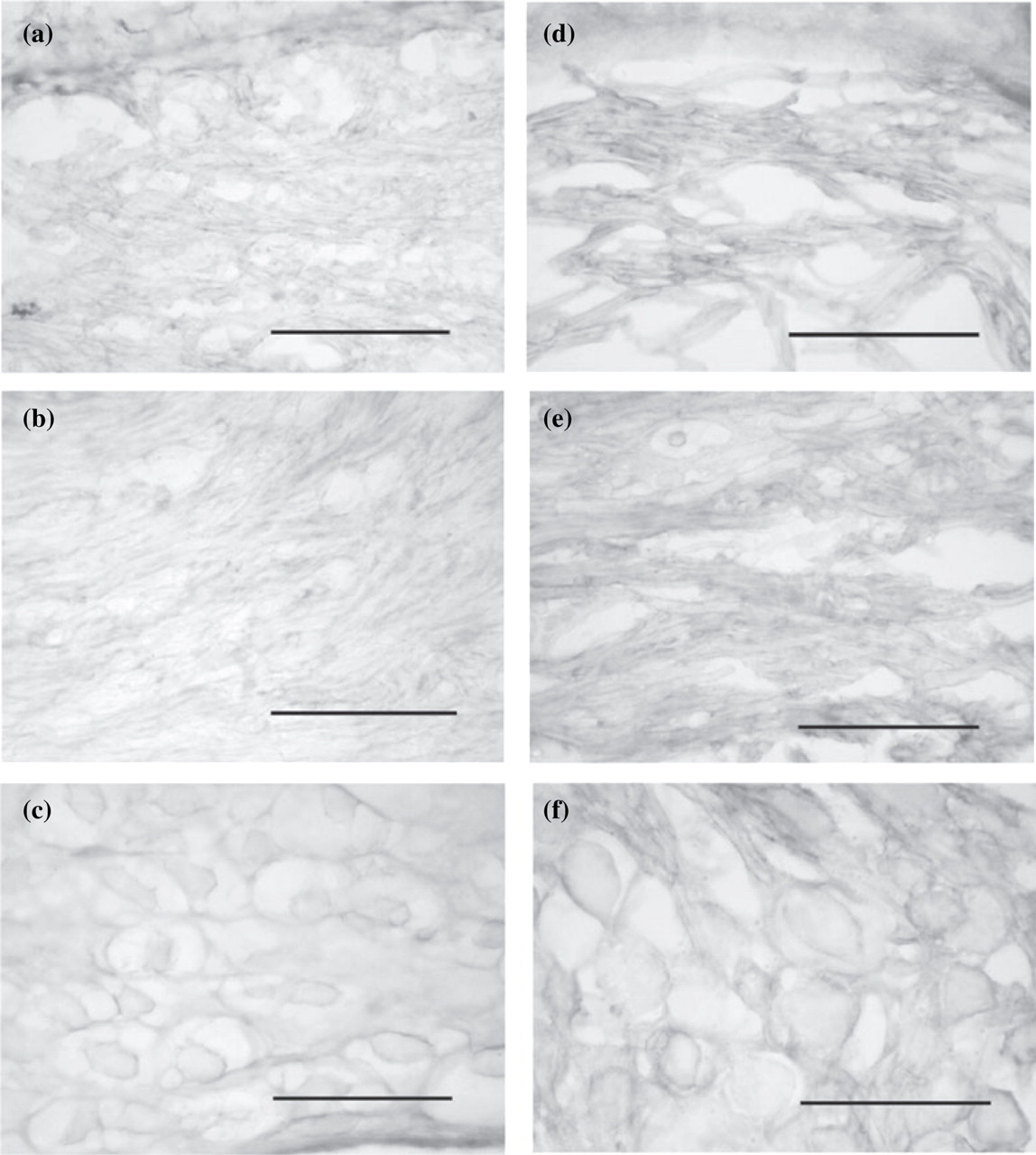
Horseradish peroxidase immunoreactivity in the vestibular nerve. These photomicrographs illustrate nerve fibres in the bony labyrinth (a,d) and internal acoustic canal (b,e) and Scarpa's ganglion (c,f) of control (a–c) and 5-HT-injected animals (d–f). Bar: 50 µm.
Discussion
Plasma extravasation from dural and cerebral blood vessels is believed to produce a local sterile inflammatory response that contributes to pain in migraine. Moskowitz and coworkers (3, 5) provided the initial demonstration that electrical stimulation of the trigeminal nerve and chemical stimulation of blood vessels (by substances such as capsaicin, histamine and 5-HT) produces both dural and corneal extravasation of circulating tracer substances. Vass et al. (7, 8) have extended these findings to the basilar artery, anterior inferior cerebellar artery and spiral modiolar artery, showing local extravasation along the blood vessels after either electrical stimulation of the trigeminal nerve or capsaicin application to the round window membrane. Our data extend the findings of extravasation into specific tissues of the inner ear.
This study identified three different patterns of HRP extravasation in different inner ear tissues after an acute intravenous infusion of 5-HT. A high degree of baseline and 5-HT-evoked extravasation (similar to skin, dura mater and the tympanic membrane) occurred in the stria vascularis and the tectorial membrane. The remaining structures in the inner ear had baseline extravasation levels that were considerable lower than in dura mater or skin, but slightly higher than in brain parenchyma. Scarpa's ganglion, the basal portion of the spiral ganglion and modiolus and the vestibular nerve segment within the internal acoustic canal (IAC) were similar to brain parenchyma in the sense that they showed no significant 5-HT-induced extravasation of the tracer. In contrast, structures such as the apical spiral ganglion and modiolus, and intralabyrinthine segments of the superior and inferior nerve showed elevated extravasation after 5-HT infusion.
The inner ear structures that show evidence of uptake of extravasated HRP vary in both their degree of vascularization and trigeminal innervation. For example, the stria vascularis is a highly vascular structure, receiving its blood supply from radiating arterioles from the spiral modiolar artery. Vass et al. (6) have demonstrated that trigeminal sensory fibres terminate densely throughout the depth of stria vascularis. Given evidence for perivascular extravasation of vessels receiving trigeminal sensory innervation from other studies (3, 7, 8), it seems likely that there is direct extravasation into the stria vascularis. The same considerations are true for the extravasation in the basilar membrane, vestibular membranous labyrinth, spiral limbus, apical spiral ganglion and modiolus, and intralabyrinthine segments of the superior and inferior nerve. On the other hand, the tectorial membrane is avascular and lacks innervation. Hence, it is important to consider other routes for tracer uptake in this structure. Since the tectorial membrane is bathed in endolymph, it seems likely that the tracer uptake originates from extravasated protein that leaks across the stria vascularis and dark cells into the endolymph.
One of the most remarkable findings of this study is that the stria vascularis shows both baseline and 5-HT-evoked extravasation that is indistinguishable from properties of dura, skin and the tympanic membrane. Early protein tracer studies of the blood–labyrinth barrier used intravenous injections of HRP (mixtures of acidic and basic isozymes) to demonstrate endocytotic uptake by vascular endothelial cells and intermediate cells of the stria vascularis (13–17). This ‘extravasated’ HRP was also present in the intrastrial space, but there was a discrepancy as to whether the HRP entered the endolymphatic fluid compartments (13) or was confined within the intrastrial fluid by tight junctions between marginal cells (16). Since these studies were performed under baseline conditions, they imply that there is a baseline transendothelial pathway for transport of proteins from blood to the intrastrial space. Thalmann and Thalmann have suggested that the baseline protein concentrations in the endolymph are produced by a selective blood–perilymph barrier and poorly selective but tight perilymph–endolymph barrier, with the perilymph serving as a buffer compartment (18) for protein transfer (19). However, they noted that plasma proteins such as apolipoproteins D and J are enriched in the endolymph, which implies that a selective blood–endolymph transport system may also be operative (19). The fact that basal and 5-HT-evoked extravasation in the stria vascularis paralleled the pattern in skin and dura mater in this model of neurogenic migraine (3, 5) raises the possibility that neurogenic migraine is accompanied by protein extravasation into the intrastrial compartment, with subsequent partitioning into endolymph and perilymph.
The stria vascularis plays an important role in the generation of the endocochlear potential and maintaining the K+ concentration in the endolymph. Since K+ provides the major charge carrier for the hair cell transduction mechanism, a robust K+ cycling system is essential for maintenance of hearing and balance function. Wangeman has recently reviewed the cellular physiological bases for K+ homeostasis in the inner ear in terms of three compartments of the stria vascularis, the fibrocyte-basal cell-intermediate cell compartment, the intrastrial fluid compartment and the marginal cell compartment (20). Potassium is transported across the gap junctions between fibrocytes, basal cells and intermediate cells, which form a barrier between the perilymph and intrastrial fluid compartment. Potassium is transferred from intermediate cells to the intrastrial fluid via the opening of the inward rectifier K+ channel, subfamily J, member 10 (KCNJ10), generating the endocochlear potential. Potassium in the intrastrial fluid then enters marginal cells through actions of Na+/K+-ATPase and a Na+/2Cl–/K+ cotransporter. Potassium exits the marginal cells though KCNQ1/KCNE1 K+ channels, with the endolymph–intrastrial barrier maintained by tight junctions near the luminal surface of marginal cells. Local plasma extravasation into the intrastrial space has the potential to create an osmotic load that may interfere with the K+ recycling mechanism, resulting in abnormal auditory transduction.
The trigeminal innervation of the stria vascularis, dark cells, spiral modiolar artery and radiating arterioles has been proposed as a mechanism for vestibular and auditory symptoms in migraine (6–8). Our results provide direct evidence of extravasation at sites that have the potential to generate these symptoms (Fig. 6). Trigeminovascular reflex activation (21) can produce vasodilation of intracranial, dural and inner ear arteries, which activates trigeminal sensory afferents. The activated trigeminal afferents release neuropeptides (e.g. calcitonin gene-related peptide, substance P and neurokinin A) that produce local extravasation. The extravasation results in addition trigeminal sensory activation, which contributes to persistent activation of the trigeminovascular reflex and the migraine pain. Our data suggest further that extravasation in the tympanic membrane may produce a perception of otalgia. In inner ear, the extravasation and vasodilation may produce neurogenic sterile inflammation in the ganglia and the intralabyrinthine portions of the eighth cranial nerve. Extravasation into the stria vascularis may also alter K+ cycling, with an impact on hair cell transduction.
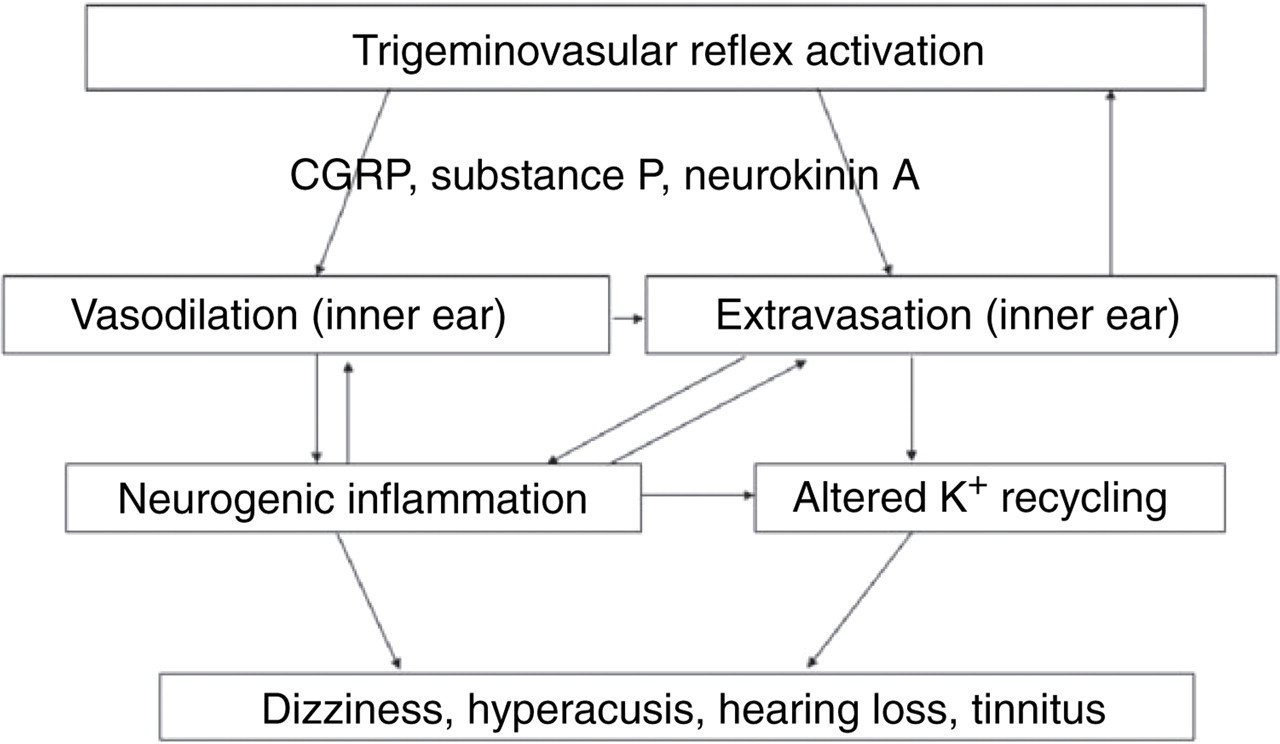
Possible mechanism of the inner ear migraine model. Trigeminovascular reflex activation can produce vasodilation of intracranial, dural and inner ear arteries, which activates trigeminal sensory afferents. The activated trigeminal afferents release neuropeptides [e.g. calcitonin gene-related peptide (CGRP), substance P and neurokinin A] that produce vasodilation and local extravasation in the inner ear as well as in the meninges. Plasma extravasation induces local aseptic inflammation, especially in the stria vascularis, which may influence K+ recycling in the inner ear.
It is interesting to note that the vestibular ganglion and spiral ganglion cells express many of the same neuropeptides as the trigeminal afferents on arteries (11). Therefore, spillover of neuropeptides from the eighth nerve afferents has the potential to affect vascular permeability. Conversely, substance P spillover from trigeminal vascular afferents can potentially affect spiral ganglion cell activity (22). Finally, released neuropeptides in the inner ear space may also stimulate mast cells in the endolymphatic sac (23, 24), which may exacerbate inflammation.
In conclusion, our results have demonstrated that intravenous serotonin administration evokes extensive plasma extravasation in the inner ear and around the eighth nerve as well as in the dura mater. These findings are consistent with the concept that neurogenic plasma extravasation in the inner ear may contribute to the association of dizziness, tinnitus and sound sensitivity in migraine patients.
