Abstract
Animal models for migraine have provided substantial advances on the mechanisms and mediators underlying migraine attacks. The neurogenic inflammation model has helped understanding the perivascular mechanisms underlying the pathophysiology of migraine attacks, the receptors involved and the effect of specific antimigraine drugs. The model based on probing the neuronal effects of nitroglycerin – an organic nitrate known to induce spontaneous-like migraine attacks in predisposed subjects – in the rat has provided interesting insights into the neuroanatomic circuits and neuropharmacological mechanisms involved in the initiation and repetition of migraine attacks.
Keywords
Introduction
The utility of electroencephalography (EEG) in the diagnosis of patients with headache has been controversial. Although early EEG studies of migraine emphasized the frequent abnormal recordings, contemporary reviewers have criticized most studies for various methodological omissions and flaws (1–4). The EEG is not useful in the routine evaluation of patients with headache but it may be useful in those headache patients who have unusual symptoms that suggest a possible seizure disorder (5, 6).
Electroencephalography in migraine
A few controlled and blinded studies have shown slight excess of various (interictal) EEG rhythms in migraine (with and without aura) patients (7–10). Focal slow activity has been reported in 0–15% and spike activity in 0.2–9% of patients, generally not different from control group prevalences (7,9–16). However, spikes may be more common in migraine sufferers than in headache-free control subjects (17). Larger population-based blinded studies are needed to resolve this question.
During visual aura, either slow waves, depression of background activity amplitude or normal EEG have been reported (18–21). Definitely abnormal EEG with unilateral or bilateral delta activity is often recorded during attacks of hemiplegic migraine (22–24), and during attacks of basilar migraine with disturbed consciousness (25–29). A recent retrospective study report that 80% of children with migraine with aura have abnormal EEG during headache (30). EEG in tension-type headache is generally considered to be normal.
Photic EEG driving responses
Golla and Winther (31) described persistence of photic driving to 20 Hz flashes or above (the H-response) in 108 of 113 patients with ‘idiopathic’ and post-traumatic migraine-like headache or epilepsy, while 42 of 50 headache-free subjects (15 with epilepsy) lacked a response above 14 Hz. The H-response is accordingly not specific to migraine and it occurs frequently in the post-concussion syndrome and in healthy subjects when EEGs are evaluated blindly (10,12,32,33). An epileptiform photoparoxysmal response is not a feature of migraine, however (34).
In a critical literature review, van Dijk et al. (35) reported highly variable sensitivity (25–100%) and specificity (14–100%) estimates of photic driving in migraine. The authors correctly emphasized several methodological problems including choice of variables, complexity of analysis methods, variable frequency analysis packages, lack of luminosity control, and lack of information about the time relationship to preceding (and following) headache attacks (35). More recent authors report lack of alpha-blocking during photic stimulation (36), increased driving power in adults and children with migraine with and without aura (37), and a trend towards a greater driving response in migraine with aura compared with migraine without aura (38). Chorlton and Kane (39) recently reported high H-response sensitivity and specificity in an open study of 33 migraine patients (with and without aura) and 40 healthy controls.
De Tomasso et al. (40) recently published a blinded study of a large sample of migraine without aura patients compared with tension-type headache patients and controls. Photic driving power, averaged across the 15–30 Hz stimulus range, was higher in migraine as well as in tension-type headache compared with controls. Interestingly, photic driving power seemed to decrease (towards ‘normal’ values) during the headache phase (41). The photic driving response is depressed during emotional stress (42), and these responses accordingly should also be investigated in other chronic pain syndromes and disorders.
Electroencephalography frequency analysis including topographic brain mapping
Quantitative frequency analysis of EEG (QEEG), with or without topographic mapping is a research method to be used in conjunction with visual EEG interpretation performed by a skilled EEG physician (43). Several rather small QEEG studies of headache patients have used a variety of methods (Table 1). The results are not quite consistent, but a pattern of increased alpha-rhythm variability (and/or asymmetry) in the headache-free phase emerged in early studies (44–49). In migraine without aura attacks, a slowing of posterior mean frequency without obvious change in absolute power was recently reported (41). In children during visual aura, alpha power depression followed by bifrontal and parietooccipital increased delta power has been found (50).
Quantitative unblinded EEG studies in headache
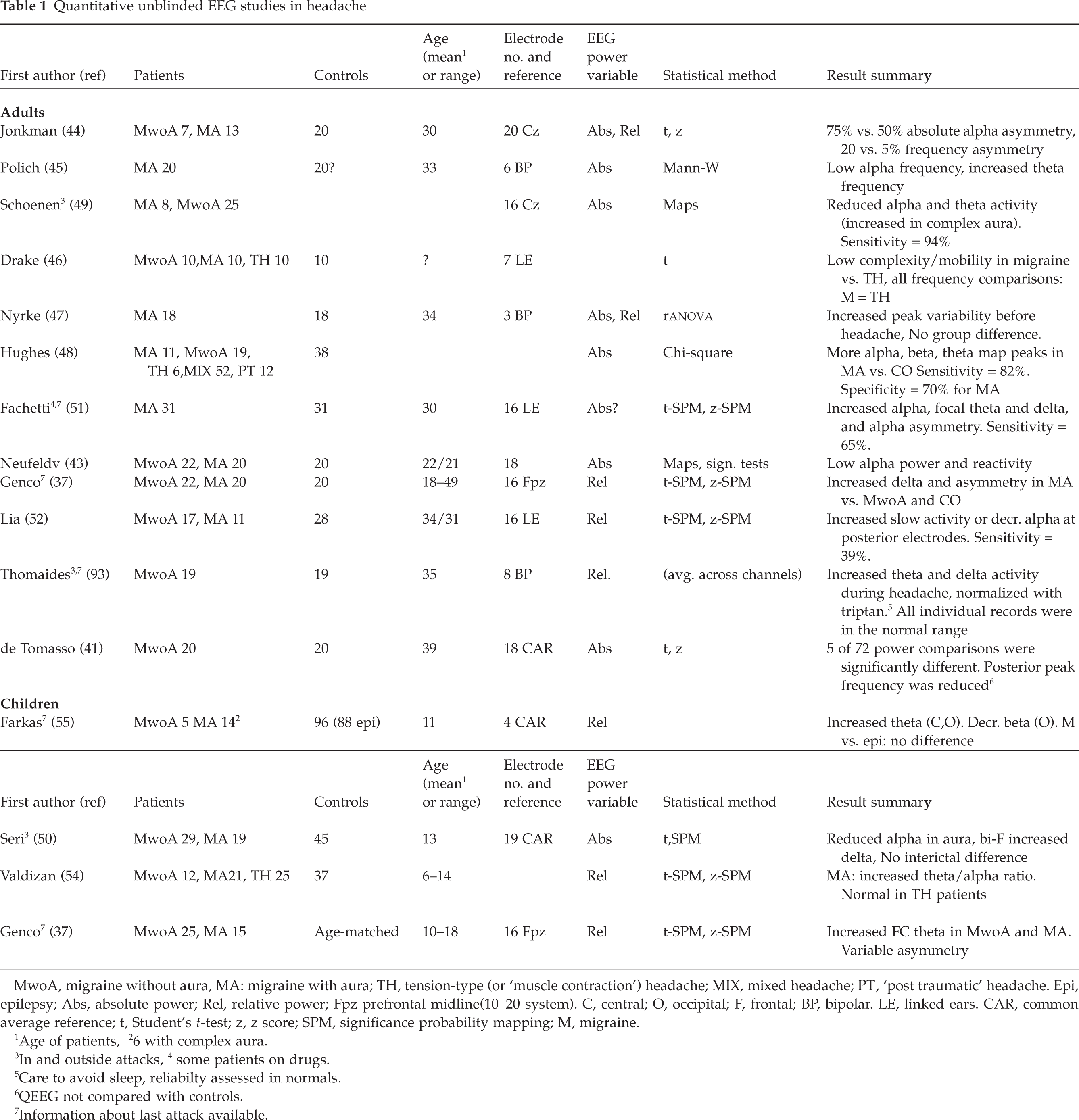
MwoA, migraine without aura, MA: migraine with aura; TH, tension-type (or ‘muscle contraction’) headache; MIX, mixed headache; PT, ‘post traumatic’ headache. Epi, epilepsy; Abs, absolute power; Rel, relative power; Fpz prefrontal midline(10–20 system). C, central; O, occipital; F, frontal; BP, bipolar. LE, linked ears. CAR, common average reference; t, Student's
1Age of patients,
26 with complex aura.
3In and outside attacks,
4some patients on drugs.
5Care to avoid sleep, reliabilty assessed in normals.
6QEEG not compared with controls.
7Information about last attack available.
Recent interictal studies have disclosed varying abnormalities. Fachetti et al. (51) found increased absolute alpha power asymmetry in adult migraine with aura, but this was not confirmed by another group (52), possibly because the latter authors used relative (not absolute) power asymmetry variables. Abnormal QEEGs in a larger proportion of migraineurs, e.g. increased focal slowing in 65% of 31 patients, has also been reported (51). Lia et al. (52) found abnormal relative spectral values in 11 of 28 migraine patients, i.e. mainly increased slow activity or decreased alpha activity in the posterior leads. Neufeld et al. (53), on the other hand, found lower occipital alpha power in migraine (with and without aura) compared with healthy controls. In children suffering from migraine with aura, increased theta–alpha ratio (54), increased theta power (37, 55), and normal bandpowers (50) have been found in the interictal period.
Future studies need to select robust reliable QEEG variables. The need for data reduction must be balanced toward unwanted loss of spatial and temporal information. These tests are still not useful in diagnosis and management of headache patients, but they may continue to provide important pathophysiological information. However, QEEG is probably not so useful in the exploration of the migraine–epilepsy relationship.
Epilepsy and migraine
Two common (independent) disorders will coexist in many patients by chance (56), but a significant association between migraine and epilepsy may exist (57, 58). This topic was recently reviewed by Bazil (59), Welch & Lewis (60), and Andermann & Zifkin (61). The classification published by Welch & Lewis (60) may serve as a useful introduction to the field although the proposed categories are not mutually exclusive (Table 2). The connection between the two disorders has received renewed attention with the discovery of a defect in the P/Q-type voltage-gated calcium channel gene on chromosome 19 in familial hemiplegic migraine. A possible association between absence epilepsy and a defect in another subunit of the same channel is under study (62). It has been shown that (at least) one antiepileptic drug (valproate) is effective in migraine prophylaxis (63).
Suggested categorization of the relationship between migraine and epilepsy (modified after Welch and Lewis 1997)

Among patients with epilepsy, the migraine prevalence was reported to be 14% (64), i.e. similar to the population prevalence (65). Symptoms of migraine and epilepsy coexist in some mitochondrial encephalopathies (61). Migraine-like headache may be an ictal or postictal epileptic manifestation (66, 67), but postictal headache is seldom migraine-like (68). Epileptic seizures may rarely be triggered by migraine attacks (57,64,69,70).
A link between partial childhood epilepsy and migraine may seem to exist based on some observational studies (71, 72) although one well-designed population-based epidemiological study has not shown this association (73). Migraine-like symptoms (visual disturbance, headache and vomiting) occur in many patients with childhood epilepsy with occipital paroxysms (CEOP) (74). Patients with CEOP have occasionally been misdiagnosed as basilar migraine (75) and basilar migraine may possibly coexist with CEOP in some subjects (61,76,77). The nature of visual hallucinations seems to differ in migraine (black and white linear patterns) vs. epilepsy (multicoloured circular or spherical patterns) (78).
Interictal EEG in CEOP patients typically shows occipital spike wave activity that often blocks on eye opening (74, 79). This EEG pattern is also seen in benign nocturnal childhood epilepsy and other syndromes however (80). Ictal vomiting (81) and reflex activation by photostimulation (75) may also occur in CEOP.
Patients with basilar migraine (25, 82) and disturbed cognition or consciousness may have markedly abnormal EEG during attacks (26, 27). Frontally dominant rhythmic delta activity (28, 29) and generalized delta activity (29) has been observed. The significance of ‘excess beta activity’ (83) is still uncertain, because the physiological beta rhythm also increases during light sleep and performance of mental tasks. In general, excess beta activity has not been convincingly linked to any disease or syndrome (84).
Clinical symptoms may sometimes be misleading. Prolonged loss of consciousness was for instance found in 15% of CEOP children in one study (85). On the other hand, migraine was found to be mistaken for epilepsy in 1.4% of referrals to an epilepsy centre (86). Video-EEG may occasionally assist in diagnosing ‘psychogenic basilar migraine’ (87). Non-specific unilateral or bilateral slowing is also found in the syndrome of ‘pseudomigraine with temporary neurological symptoms and lymphocytic pleocytosis’ (88). Young adult patients with assumed ‘migraine-triggered epilepsy’, normal EEGs, and inconsistent response to AED treatment have been described (89).
Kramer et al. (90) performed EEGs in 257 of 312 children with chronic headache; epileptiform activity was found in 31 (12%). The yield was higher in the subgroup with ‘very brief headache’, i.e. 27%. A clinical diagnosis of previously unrecognized epilepsy was not made in any subject, although antiepileptic medication was tried in a few subjects with variable results. Headache attacks disappeared without treatment in seven of the 31 children.
Concluding remarks
When EEG is used to explore migraine pathophysiology, it is not easy to compare the results from different studies because different methodological protocols have been applied. In order to avoid epoch selection bias, we recommend blinding EEG and QEEG studies whenever possible. Interpretations of the EEG results are also affected by the lack of precise knowledge of the physiological nature of various EEG rhythm generators (91). The study of sensory and cognitive evoked responses has apparently contributed more to the understanding of headache pathogenesis than EEG studies during the past decade. Headache patient groups seem to have increased EEG responses to photic stimulation but a useful biological marker for migraine has not been found. It should be informative to study if EEG variability and rhythmicity changes across the headache phase (47).
A clinical EEG is clearly indicated in patients with acute headache attacks when either epilepsy, basilar migraine, migraine with prolonged aura or alternating hemiplegia (92) is suspected. Unequivocal epileptiform abnormalities usually suggest a diagnosis of epilepsy, and in children with occipital spike-wave activity (with or without blocking on eye opening) the probable diagnosis is childhood epilepsy with occipital paroxysms. The final diagnosis of either an epilepsy syndrome or migraine must be mainly based on a clinical judgement.
