Abstract
This open study evaluates the effectiveness and safety of topiramate for the prophylaxis of chronic tension-type headache. Fifty-one patients were enrolled, of whom 46 completed 24 weeks of treatment with topiramate. Daily dosing was titrated from 25 mg to 100 mg by treatment week 4. The primary efficacy parameter was headache frequency at weeks 13-24 compared with baseline. Headache frequency declined from 23.50 ± 5.32 days (baseline, mean, SD) to 12.58 ± 6.28 days at weeks 13-24 (P < 0.0001), with frequency of severe headaches dropping from 8.18 to 3.14 days (P < 0.0001). The average headache intensity dropped from 6.13 to 2.07 on the visual analogue scale (P < 0.0001). These parameters were not significantly reduced at weeks 5-12. A 50% reduction in headache frequency was achieved in 73% of patients at weeks 13-24. Also improved were mood, sleep, quality of life (all parameters, P < 0.0001) as well as the Beck Depression Inventory-II (P < 0.0001). In addition, a highly significant weight loss of 2.14 kg (mean) was observed between baseline (71.64 ± 10.65 kg) and week 24 [69.50 ± 10.04 kg (SD)] (P < 0.0001). There were only few side-effects, none of these rated severe. The results provide preliminary confirmation of the efficacy and tolerability of topiramate in the prophylaxis of chronic tension-type headache.
Keywords
Introduction
Chronic headaches are prevalent in tertiary headache centres and often difficult to treat. Drugs with proven efficacy for the treatment of chronic tension-type headache (CTTH) are antidepressants, anticonvulsants and muscle relaxants. The tricyclic antidepressant amitriptyline has been demonstrated to be most effective in several placebo-controlled studies (1) and the newer noradrenergic and specific serotonergic antidepressant mirtazapine was recently demonstrated also to be effective (2). However, these drugs are only moderately effective, with a headache frequency reduction of approximately 30% compared with placebo and treatment is often hampered by side-effects (1).
The analgesic effect of antidepressants in CTTH is not dependent on their antidepressant effect and can not be explained by inhibition of serotonin reuptake (3). It has been demonstrated that the central nervous system (CNS) is sensitized in patients with CTTH (4) and it is most likely that the analgesic effect of antidepressants (2, 5) and of nitric oxide synthase inhibitors (6, 7) in CTTH is explained by modulation of central pain sensitivity. Drugs with potential blockade of central hyperexcitability and with a favourable side-effect profile would therefore be candidates for the prophylactic treatment of CTTH. The antiepileptic drug topiramate meets these requirements.
Topiramate (TPM) has five potential mechanisms of action (for review, see 8). These include the blockage of sodium channels, enhancement of GABA-a receptors (an inhibitory neurotransmitter), an inhibitory effect on glutamate receptors, inhibition of
In a pilot study, we found TPM to be effective in CTTH with only few adverse events. These (unpublished) data suggest that TPM may be an effective and safe prophylactic agent for the treatment of CTTH. This open-label study was designed as a prospective pilot investigation of the efficacy, tolerability and safety of TPM in the prophylaxis of CTTH.
Methods
This open-label trial with a 4-week baseline (i.e. weeks − 4 through − 1), followed by three treatment periods (treatment period I, weeks 1–4; treatment period II, weeks 5–12; treatment period III, weeks 13–24) was designed in accordance with the Declaration of Helsinki and the Good Clinical Practice guidelines. The study was performed between March 2003 and December 2004. Patients were recruited by referral to our tertiary headache centre (General Hospital, Linz). Informed consent was obtained from all patients. Participants received no monetary compensation for taking part in the study.
Patients meeting initial entry criteria underwent screening evaluations, signed the informed consent and received instructions to keep a daily headache diary for 1 month (baseline). One month after the screening visit, patients returned for their baseline visit and further evaluation. Patients meeting baseline inclusion criteria were instructed to start taking the study medication the next day (day 1). TPM therapy was started at a dose of 25 mg daily and increased by 25 mg weekly up to 100 mg daily divided into two doses. Patients returned at weeks 4, 8, 12, 16, 20 and 24 to review their diaries, assess adverse events and concomitant medication. Laboratory tests for liver and kidney parameters were performed at the first screening visit, week 4 and week 24, with the urine pregnancy test repeated at the end of baseline and at each controlled visit. The Beck Depression Inventory-II was completed at the initial screening and in each treatment period.
The primary efficacy parameter was headache frequency in treatment period III compared with baseline. Secondary efficacy parameters were headache frequency at treatment periods I and II, and severity [changes in 10-point visual analogue scale (VAS) measures], frequency of severe headache (VAS 7–10) and duration (headache hours per day) in treatment period III compared with baseline. Further efficacy parameters were changes in sleep, mood and quality of life, as well as depression. Safety objectives included the identification of adverse events, including their significance and severity, based on patient diaries, interviews and changes in laboratory results.
Outcome measures
Patients recorded peak and average levels of headache severity each day using a 10-point VAS, from 0 = no headache to 10 = most severe, intensely painful. Headache duration was recorded as the number of hours of headache each day. Peak level of functional impairment was recorded on a 4-point scale (0 = able to perform normally, 1 = ability to function mildly impaired, 2 = ability to function moderately impaired, 3 = ability to function severely impaired). Patients also recorded in the diary the dose of TPM for each of two daily doses, as well as the name and dose of rescue medications. For sleep, mood and quality of life VAS were performed. Each line was anchored at 0 (‘extremely bad’) and 10 (‘extremely good’). Changes in psychological status (depression) were evaluated using the Beck Depression Inventory-II.
Inclusion criteria
Patients between the ages of 18 and 60 years with CTTH, diagnosed according to the criteria of the International Headache Society (14) for at least 3 months prior to entry, were included in the study. Patients had to complete a 4-week baseline headache diary. Only those patients whose diaries showed at least 15 days of headache in 4 weeks were permitted to start TPM and continue for the remainder of the study.
Exclusion criteria
Patients with other primary chronic headaches, with concomitant migraine with and/or without aura and those with analgesic overuse (>15 days) were not enrolled. If there were any migrainous features, especially additional or intermittent pulsating headache and/or aggravation during movement, those patients were excluded. Patients with coronary artery disease, renal failure, hepatic failure, epilepsy, any neuropsychological problem or with an unstable social situation that might interfere with their ability to complete the study adequately were also excluded. Further exclusion criteria included previous TPM treatment of epileptic seizures or other indications, and women who were pregnant, breast-feeding or sexually active and of childbearing potential who were not using medically accepted means of contraception. No other prophylactic medical treatments or non-pharmacological procedures were allowed 3 months prior to or during the whole time of the study.
Permissible analgesic/abortive/rescue medications included simple analgesics [i.e. aspirin, paracetamol, mefenamin acid and non-steroidal anti-inflammatory drugs (NSAIDs)].
Statistical analysis
Daily patient diary entries were used to calculate the number of days with tension-type headache (standardized to a 28-day period), means for the average level of headache (VAS 1–10), headache duration (min) and peak level of impairment (0–3), and total use of analgesic/abortive medication for baseline and each of the three treatment periods. Comparisons of all outcome measures were made using the paired t-test. In order to minimize the likelihood of spurious findings of significance, the acceptable probability levels for statistical significance were determined using Bonferroni correction. The number of treatment responders was also identified. A responder was defined as any patient with a reduction in headache frequency of at least 50% when comparing the baseline period with each treatment period. The data are presented as mean ± SD.
Results
Disposition of patients
A total of 62 patients were assessed for eligibility. After conclusion of the baseline records, 51 patients continued to meet inclusion criteria and started treatment with TPM (flow of participants, Fig. 1). The sample consisted of 41 women (80.4%) and 10 men (19.6%), with a mean age of 41.6 years (± 11.20; median 37; range 18–58 years). History of CTTH was 7.2 years (± 10.42). Forty-six (90.2%) patients completed the 24-week treatment period. One patient was lost to follow-up at week 16. Four (7.9%) chose to discontinue treatment due to adverse events rated mild to moderate.

Flow of participants through the study.
Prior to study entry, 21 (41.2%) patients had used tricyclics, 11 (21.6%) had had acupuncture and 27 (53%) a chiropractic procedure.
Safety and tolerability
Reasons for dropout were somnolence in combination with difficulty with memory and concentration (n = 3, 5.8%) and paraesthesia (n = 1, 2.1%). Mild to moderate adverse events possibly related to TPM were reported by six patients (11.7%), namely somnolence (n = 2, 3.9%) and fatigue (n = 4, 10.0%). Mild paraesthesia, predominately in the arms, was described by 19 patients (41.3%) and disappeared within the first 4–6 weeks after treatment began. None of the patients reported possibly related adverse events rated severe. In addition, a highly significant weight loss of 2.14 kg (mean) was observed between baseline (71.64 ± 10.65 kg) and week 24 (69.50 ± 10.04 kg; P < 0.0001). Treatment with TPM showed no significant abnormalities in laboratory parameters including values for kidney and liver at week 4 and week 24.
Headache diary measures
For those who completed the study, frequency of headache and frequency of severe headache are shown in Fig. 2. Headache frequency declined from 23.50 ± 5.32 days at baseline to 12.58 ± 6.28 days in treatment period III (P < 0.0001). Headache frequency declined to 21.70 ± 4.28 days in treatment period I (P > 0.05) and to 20.90 ± 7.12 days in treatment period II (P > 0.05). Frequency of severe headache dropped from 8.18 days (± 1.68) at baseline to 3.14 days (± 1.26) in treatment period III. The average headache intensity dropped from 6.13 (± 1.78) to 2.07 (± 1.22), which was also statistically highly significant (P < 0.0001). Post hoc analysis indicated that significant headache improvement occurred from week 13 through 15 vs. week 1 through 12 (Fig. 3). Duration of headache decreased from 8.0 ± 4.8 h/day at baseline to 3.0 ± 4.1 h/day during treatment period III (P < 0.001).
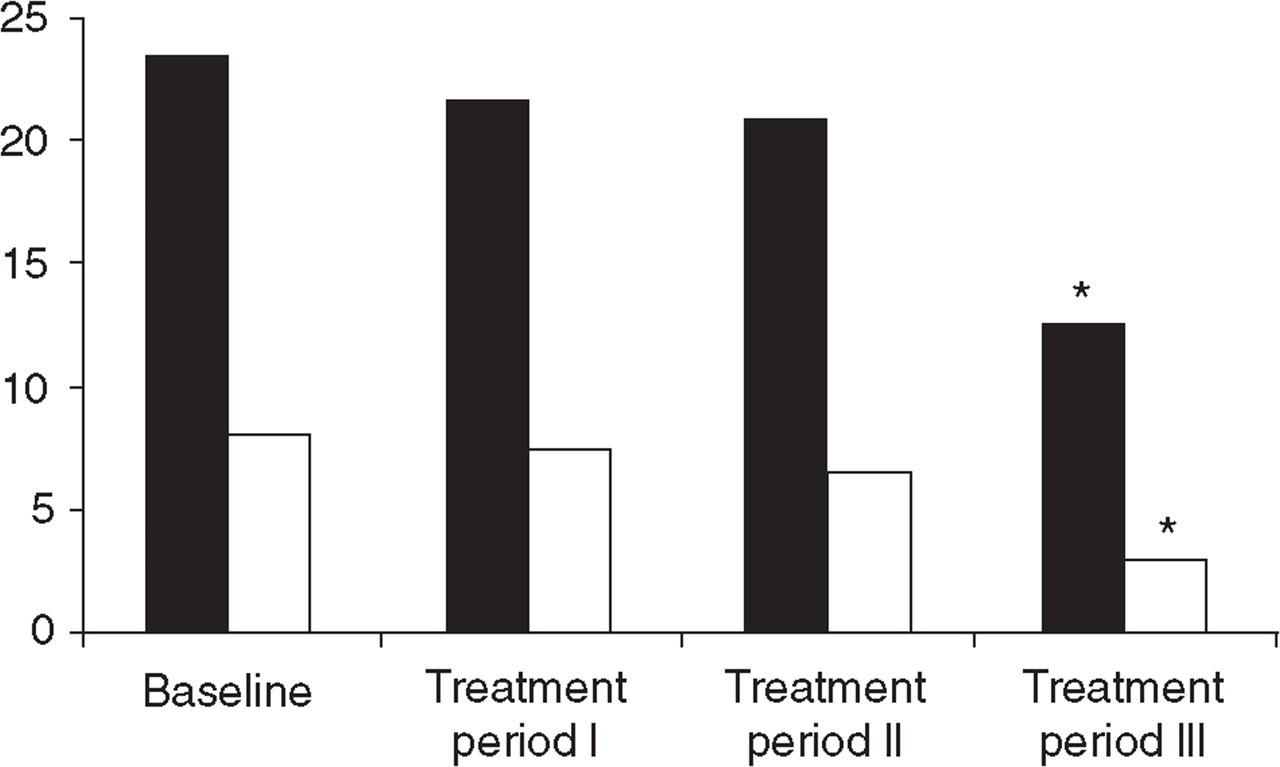
Headache frequency (▪) and frequency of severe headache (□) (days/month). ∗P < 0.0001.
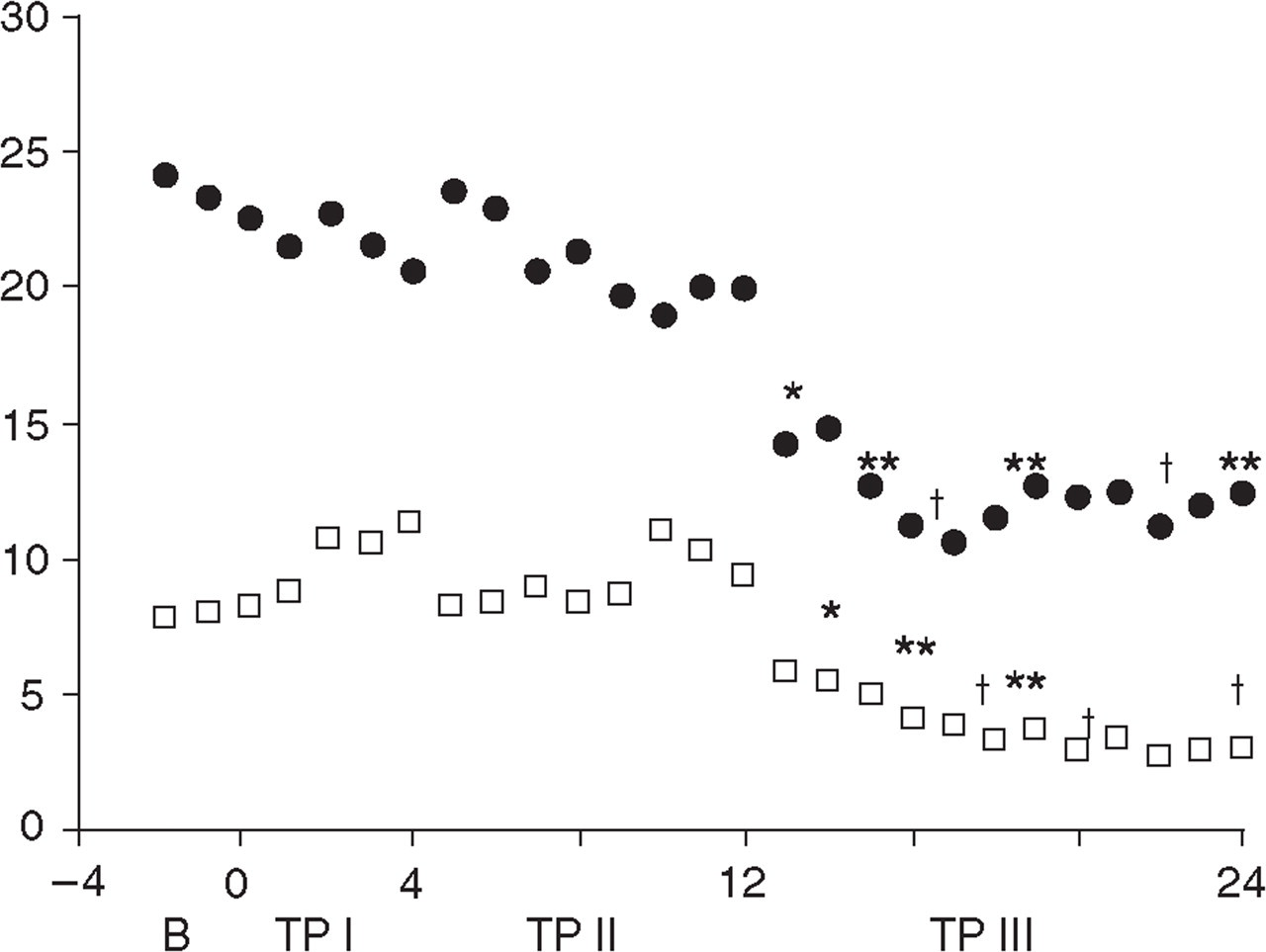
Headache frequency (days/month) and headache duration (h/day) over baseline and all treatment weeks (B, baseline; TP, treatment period). □, Days/month (mean); □, h/day (mean). ∗P < 0.005; ∗∗P < 0.001; †P < 0.0001.
A 50% headache improvement was found in 21% of patients for treatment period II and in 73% of patients for treatment period III (P < 0.0001).
Level of headache-related impairment per day rated on a 4-point scale showed improvement from 2.8 (baseline) to 0.4 (treatment period III), which was statistically also highly significant (P < 0.0001).
Use of abortive/analgesic medication
Use of abortive/analgesic medication showed a statistically significant decline compared with baseline. Used analgesics were paracetamol (n = 31, 67.4%), mefenamin acid (n = 39, 84.8%), aspirin (n = 14; 30.4%) and NSAIDs (n = 17, 37%). Abortive/analgesic use dropped from 2.1 ± 2.6 days per week at baseline to 0.8 ± 1.6 days per week during treatment period III (P < 0.001). There was no statistically significant reduction in the use of abortive/analgesic medication during treatment periods I and II (P > 0.5). There was a slight tendency not to use paracetamol and NSAIDs in treatment period II vs. treatment period I.
Depression, sleep, mood, quality of life (Fig. 4)
A significant reduction was seen in Beck Depression Inventory-II scores, namely from 9.1 ± 6.1 at baseline to 4.1 ± 4.8 at conclusion of the 24 weeks of treatment (P < 0.00073). Also improved were mood, sleep and quality of life (P < 0.0001).
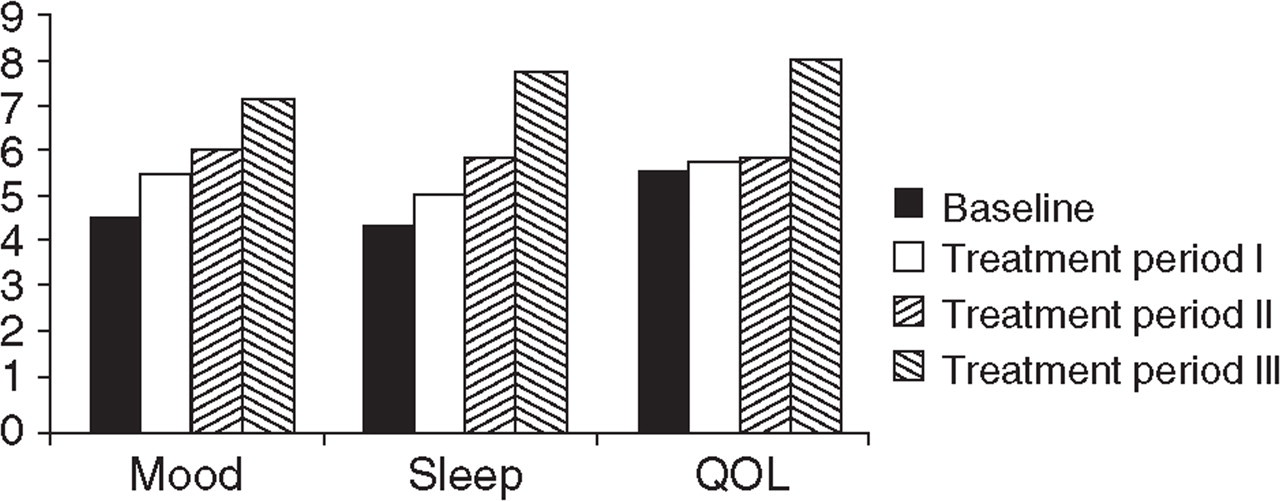
Visual analogue scales (0 = extremely bad, 100 = extremely good, mean rating) for mood, sleep, quality of life (QOL).
Discussion
Our results indicate that TPM is highly effective in preventing CTTH. Although the study was not placebo controlled, efficacy was considerably greater than that usually seen in CTTH trials (2, 15, 16).
Knowledge of the pathophysiology of CTTH is still limited. Peripheral mechanisms (myofascial tenderness) as well as central mechanisms (central sensitization) have been extensively discussed (for review, see 4). Persistent activity in peripheral nociceptors may cause increased responsiveness of second-order neurons and neurotransmitter release from C-fibres may be responsible for altered neuronal function (17). In patients with CTTH the CNS may be sensitized at the supraspinal level (18–20) and at the level of the spinal dorsal horn and trigeminal nucleus (21). A disturbance in pain-modulating transmitters, e.g. deficient descending inhibition, is also likely to be involved in the altered pain perception of patients with CTTH (4).
Thus, CTTH could be considered a disorder characterized by neuronal hyperexcitability. As such, it would not be surprising if anticonvulsants that act by modulating neuronal function and decrease central sensitization by influencing voltage- and receptor-activated ion channels, preventing neurotransmitter and neuropeptide release, were effective in the prophylactic management of CTTH.
In this respect, TPM appears to possess unique ability to decrease excitation and enhance inhibition, which may be mediated through enhancement of GABA-evoked chloride currents (22, 23). However, it is not clear at present whether TPM directly affects neurons within the trigeminovascular complex or is secondary to action at a site distant to the trigeminocervical complex (8). TPM’s broad mechanistic profile could explain why it is efficacious in the prophylactic management of CTTH.
It is unclear why the analgesic effect of TPM was first seen after a treatment period of 3 months. One explanation could be that most of our patients had a history of chronic headaches for more than 7 years. In such individuals, central neuroplastic changes may be firmly established and therefore difficult to change. Increased excitability of supraspinal neurons and decreased inhibition may result in central neuroplastic changes (‘pain memory’). It is noteworthy that in a recent neuroimaging study using voxel-based morphometry, patients with CTTH demonstrated a significant grey matter decrease in regions known to be involved in pain processing (24). These changes were restricted to structures involved in pain processing, namely the antinociceptive system, and could reflect either the cause or the consequence of chronic head pain. Chronic pain is thought to be an expression of neuronal plasticity and as such is generated partly in the periphery and partly within the CNS (25). Abnormal modulation of brain nociceptive systems, at first transient but becoming permanent with continuing illness and predisposing to central sensitization, may explain the shift in the episodic phenotype to chronic headache. Following this line of thought, the areas of altered grey matter in CCTH are thus to be seen as pain-transmitting systems and these data may be interpreted as the consequence of central sensitization (24). This could well explain why TPM was not effective in a time frame usually observed in headache prophylaxis, but took more than 3 months to become effective, possibly by modulating neuronal function (e.g. decreasing excitation and enhancing inhibition) and decreasing central sensitization.
Clinical implications
TPM was associated with significant improvement of all parameters in treatment period III. These results argue for maintaining of scheduled TPM for at least 4 months; 12 weeks are not long enough to judge treatment efficacy. This hampers the clinical use of the drug. It is possible that it is necessary to use initially a higher dose to reverse the central hyperexcitability in these chronic patients and thereby reduce time to efficacy.
Our study demonstrates that the incidence of adverse effects, compared with that in other prophylactic treatment studies, is very low for CTTH. TPM showed a favourable side-effect profile in comparison with that of the commonly used prophylactic agents amitriptyline (26) and mirtazapine (2). None of the adverse effects involved hepatotoxicity or renal toxicity or was of a serious nature. We observed a significant weight loss with TPM, in contrast to the weight gain seen with amitriptyline and mirtazapine. The latter is a concern of many patients and may prompt discontinuation of prophylactic treatment, which was not observed in our study population.
It must be pointed out that the effects seen after 4 months could be due to a non-specific placebo effect or just a ‘time effect’, i.e. the natural course of the headaches in our sample. Due to the high number of responses and, moreover, due to the fact that the response started only after several months of continuous intake of topiramate, we suggest that neither a placebo effect, nor the natural course of the headaches in our patients could explain the whole picture. The results of our study point to a new treatment possibility with a favorable side-effect profile for a common and difficult-to-treat condition, Further investigations of TPM in the prophylactic treatment of CTTH should be conducted as double-blind, placebo-controlled, long-term studies to confirm the results of this first observation. Furthermore, the efficacy of higher doses, e.g. 150 and 200 mg daily, should be investigated.
