Abstract
Blink reflex R2 amplitude was investigated in seven patients with cervicogenic headache (CEH), 12 patients with chronic tension-type headache, 23 patients with migraine (10 with aura) and 17 headache-free controls. Standard electrical stimulation of the supraorbital nerve was applied and the response was recorded from the ipsilateral and the contralateral orbicularis oculi muscles. Low R2 amplitude was found in CEH patients compared with control subjects. Headache is unilateral in CEH and the ipsilateral and contralateral responses after stimulation on the painful side were most depressed. R2 amplitude was not significantly affected in migraine and tension headache patients. The results suggest that lower brainstem excitability is reduced in CEH. A state of hypoactivity may be present in caudal trigeminal nucleus neurons on the symptomatic side.
Introduction
Blink reflex abnormality may be caused by brainstem lesions in various neurological diseases (1–3) and it may reflect possible brainstem dysfunction or excitability changes in headache (4). The second part of the response (R2) is not an exclusively pain-mediated response because the fastest nerve fibres in the reflex are of the A-β type and R2 may persist when pain fibres have been blocked (2). Several lines of evidence still suggest that R2 partly depends on nociceptive activation: R2 can be evoked by laser heat-pain stimuli, is inhibited by distant pain stimulation which activates the diffuse noxious inhibitory control system (5), R2 abnormality is associated with facial pain in lateral medullary infarction (6) and R2 is partly suppressed by the opiate fentanyl, although to a lesser degree than the more pain-specific cornea reflex (7). R2 is probably mediated by medullar wide-dynamic range (WDR) neurons (5) and the nociceptive contribution to R2 has recently been elucidated with a new concentric (nBR) stimulation electrode (8).
Clinical criteria for (unilateral) cervicogenic headache (CEH) were published in 1990 (9) and revised in 1998 (10). Previous physiological studies have found generalized low pressure pain thresholds, most pronounced on the symptomatic side (11). Thermal thresholds were generally increased in cephalic and non-cephalic sites in CEH, suggesting a trigeminal and generalized sensory dysfunction (12). Experimentally induced neck muscle pain can increase trigeminal jaw-opening reflex excitability (13) and induce central sensitization of dural neurons (14). Thus, secondary trigeminal dysfunction may be involved in CEH (15) as well as in tension-type headache (TTH) (16) and migraine (17).
Blink reflex latency has been studied in CEH (4), but R2 amplitude (or area), a useful measure of brainstem excitability, has not been previously studied. The purpose of the present study was to evaluate if R2 amplitude could reveal unilateral or bilateral brainstem excitability changes in CEH, compared with TTH, migraine and headache-free control subjects.
Methods
Headache patients (Table 1) were diagnosed according to the International Headache Society 1988 criteria (18). The usual headache intensity was scored as slight (= 1), moderate (= 2) or severe (= 3). Cervicogenic (unilateral) headache patients were diagnosed according to the clinical 1990 criteria (9) because the study was initiated before 1998. Eleven of the 23 migraine patients had their headaches predominantly on one side and 10 had migraine with aura (MA), four of which had one-sided dominance. The investigator was blinded to the diagnosis. The supraorbital nerve was stimulated with 0.2-ms square wave pulses with irregular 2–5-s interstimulus intervals (ISI) and a typical intensity 10–12 mA, at least three times the sensory detection threshold, and mean intensities did not differ either between groups (
Subjects in the study

Ten responses for four conditions were exported to a Matlab® program, rectified and averaged. Three of 10 CEH patients originally studied (4) were excluded from the analysis because curve data files for one or more recording sites were missing in the database. The steepness of the gradient was calculated as a function of time within a preselected time window. R2 start latency was determined by a software program when the steepness change exceeded a preset cut-off value. Similarly, a R2-stop value was determined when the steepness declination changed from a high negative to a low preset cut-off value. Because the interindividual variability in rectified R2 response pattern was large we chose to determine the cut-off values on a trial and error basis. The computer-generated R2 start and stop values were also checked visually by the clinical neurophysiologist. The integrated R2 amplitude (area in µV × ms under the rectified curve) was calculated as the main response variable.
All CEH patients had strictly unilateral headache and a subset of migraine patients had unilateral headache dominance. Two symptomatic side (P) and two non-symptomatic side (N) responses were analysed for each patient: PS–PR [i.e. pain side stimulated (PS) and pain side recorded (PR)], PS–NR (contralateral response to pain-side stimulation), NS–PR (contralateral response to non-pain side stimulation) and NS–NR (ipsilateral response to non-pain side stimulation). For controls, TTH patients and 12 patients with bilateral migraine either the right or the left side was randomly chosen as the comparison side for P and N responses. Mann–Whitney tests were used to compare R2 area between each patient group and the control group, whereas side differences were evaluated with the Wilcoxon test. Pearson correlation analysis was used to explore the relation between R2 and age, headache duration and intensity. P-values < 0.05 were considered to be significant.
Results
A low R2 area recorded after stimulation of the painful side was found in CEH patients compared with control subjects, both from ipsilateral and contralateral orbicularis oculi (Table 2, Fig. 1). The R2 area was not significantly affected in migraine and TTH patients. No differences were found in migraine without aura (MoA) vs. MA patients and unilateral migraine patients did not differ from patients with bilateral pain.

Box plots with distribution of ipsilateral and contralateral R2 areas in the four groups. ‘Painful’ and ‘non-painful’ sides were selected randomly (right or left) in controls, chronic tension-type headache (CTTH) and migraine patients without side dominance. Left upper: Ipsilateral response to stimulation of the supraorbital nerve on the painful side. Right upper: Contralateral response to stimulation of the supraorbital nerve on the painful side. Left lower: Contralateral response to stimulation of the supraorbital nerve on the non-painful side. Right lower: Ipsilateral response to stimulation of the supraorbital nerve on the non-painful side. The responses recorded after stimulation on the painful side (upper row) are significantly lower in cervicogenic headache (CEH) than controls (Table 2).
Mean R2 area† (SD) in µV × ms
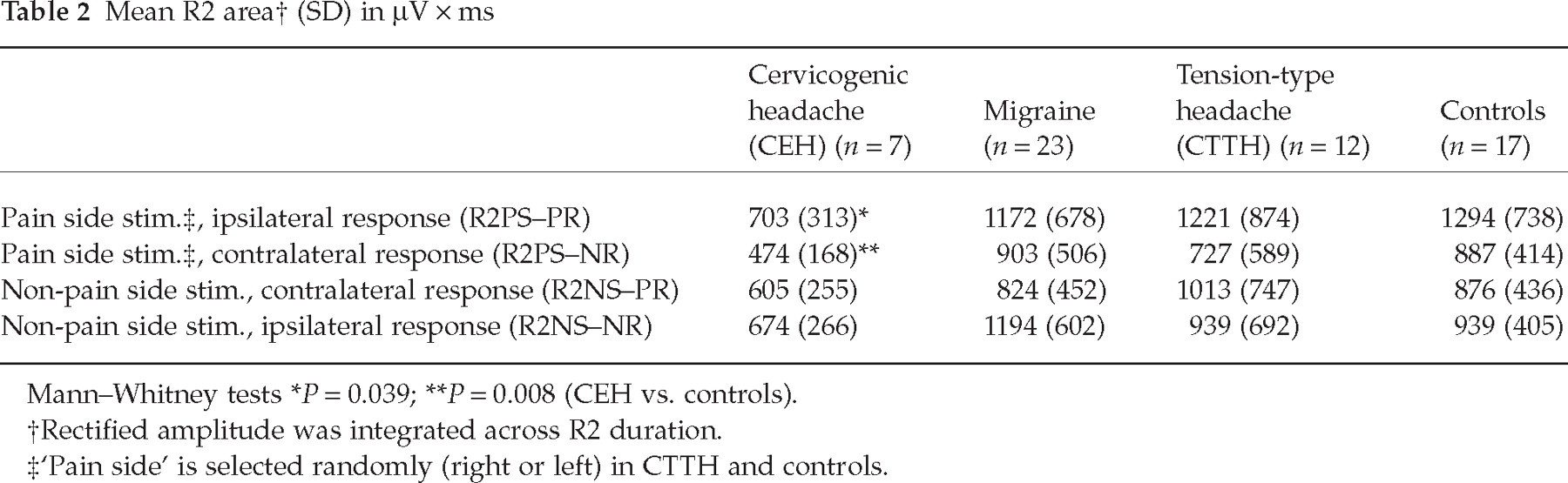
Mann–Whitney tests
P = 0.039;
P = 0.008 (CEH vs. controls).
Rectified amplitude was integrated across R2 duration.
‘Pain side’ is selected randomly (right or left) in CTTH and controls.
R2 duration was abnormal in CEH when the painful side was stimulated; ipsilateral reduced duration and contralateral increased duration were observed (Table 3). R1 amplitude did not differ significantly between groups (Table 3). R2 amplitude did not correlate significantly either with age, headache duration or with usual pain intensity in the control or any patient group.
R2 duration and R1 amplitude: mean (SD)

Non-paramaetric Kruskal–Wallis tests were non-significant for all variables.
Mann–Whitney P < 0.05 (cervicogenic headache vs. controls).
‘Pain side’ was selected randomly in tension-type headache and controls.
In the subgroup > 35 years old, the six CEH patients had a definite trend towards low R2 amplitudes compared with 23 non-CEH subjects mostly on the painful side (ipsilateral R2 mean amplitude was 619 vs. 1121 µV, P < 0.09).
Discussion
Our main finding was that the blink reflex R2 amplitude was significantly lower in CEH patients than in control subjects when the painful side was stimulated. This pattern differed from the bilateral depression which is seen during non-specific cortically mediated R2 inhibition and adds evidence for the notion that CEH is pathophysiologically distinct from both migraine and TTH.
Cervical and trigeminal sensory neurons converge upon secondary sensory neurons in the trigeminal nucleus, probably explaining referred pain in CEH (14, 19). It is not clear why acute cervical pain seems to increase trigeminal excitability (13, 14) while chronic pain patients with CEH have reduced blink reflex excitability. One possibility is that early potentiation may eventually be followed by inhibition (possibly a long-term depression-like mechanism).
Another possibility is that a putative increased excitability in the pain-specific part of R2 is outweighed by inhibition mediated in the mechanosensitive (A-β) part of the reflex. Results from blink reflex studies during migraine attack indicate such dualism. A bilaterally reduced standard R2 area (20) and increased nBR-R2 area (21) have been reported. Whether the inhibition we observed reflects a syndrome-related trait in CEH or a pain intensity-dependent state cannot be answered in the present study, because our patients were studied only once.
R2 is mediated by polysynaptic brainstem pathways within the medial and lateral reticular formation (3, 22). The reflex arc descends at least to the caudal level of the hypoglossus motor nucleus, but it is uncertain if neurons or axons within the caudal spinal trigeminal sensory nucleus contribute to R2 (22). The R2 amplitudes tended to be low also when the non-painful side was stimulated, suggesting that supramedullar bilateral descending inhibition may contribute. We did not observe a definite efferent (lateral tegmental) blink reflex pattern (23). The low R2 duration on the ipsilateral side strengthens the impression of asymmetrical brainstem excitability in CEH. Thus, the observed pattern suggests hypofunction in caudal trigeminal nucleus WDR neurons mostly on the painful side in CEH. Recent results imply that a similar hypofunction (i.e. reduce nBR-R2 area) can be experimentally induced by ipsilateral occipital nerve (C2) anaesthetic block (24).
In a previous study we found a trend towards a short R1 latency on the painful side in CEH (4), while no trend towards low R1 amplitude in CEH is observed in Table 3. Slight hyperactivity in the principal trigeminal sensory nucleus may accordingly coexist with hypoactivity in the caudal trigeminal sensory nucleus. However, a descending mechanism that inhibits motor activity during pain (25) may also contribute to R2 amplitude reduction in CEH patients.
R2 amplitude and area seem to be normal between attacks in MoA (20, 21, 26–28) and we confirmed this result. R2 is easily habituated upon repeated stimulation (2) and reduced R2 habituation was found 1–3 days before a migraine attack (29). The latter result was confirmed for the nociceptive blink reflex method (30).
Normal R2 amplitude and area have been reported in TTH (20, 26) and confirmed in the present study. We found no evidence of trigeminal excitability changes in chronic TTH, in the previous R2-latency study (4) or in an exteroceptive suppression (ES2-duration) study (31), but neither method may be sufficiently sensitive to detect slight excitability changes.
CEH patients were older than the other groups. The correlation between age and R2 amplitude was non-significant, however, and age-matched subgroup analysis showed a clear trend towards low CEH amplitude in spite of low statistical power. It is therefore probable that age does not explain our results.
Our ISI was based on Kimura’s recommendation (3) and it was slightly shorter than the 7 s recommended by Aramideh and Ongerboer de Visser (32). Data from DeMarinis et al. (29) suggest that R2-area evoked with ISI 5 s is approximately 50% of the area with ISI 30 s. Thus, low area in the present study could theoretically also reflect increased habituation in CEH.
A weakness of the present study was the lack of pain and pain threshold measurements during registration. The usual pain level was similar in CEH and chronic TTH, however (Table 1), suggesting that headache intensity probably did not explain the differences between the groups, but we cannot rule out this possibility. Future studies should also focus on the possible effects of habituation and pain during recording.
