Abstract
A sterile inflammation in the cavernous sinus was hypothesized to underlie cluster headache (CH). Neurogenic inflammation is accompanied by the extravasation of plasma proteins in the surrounding tissue. We tested the hypothesis of an inflammatory process in the cavernous sinus in CH patients using 99mTc-human serum albumin (HSA) and single photon emission computed tomography (SPECT). Six patients with episodic CH were enrolled. After baseline imaging, CH attacks were induced by IV injection of nitroglycerin. The patients remained untreated for 20 min. A second SPECT was performed after successful treatment. Region of interest (ROI) analysis was performed on the basis of coregistered MRI/SPECT data. There was no statistical difference between the 99mTc-HSA uptake in the ipsilateral cavernous sinus before and after induction of an acute CH attack. There was no evidence for 99mTc-HSA extravasation in the cavernous sinus during the active episode as compared with the remission phase. Our results do not support the hypothesis of an inflammation in the cavernous sinus.
Keywords
Introduction
Episodic cluster headache is characterized by unilateral headache attacks of excruciating pain intensity. Conjunctival injection, lacrimation, rhinorrhea and Horner's syndrome are typical accompanying symptoms. Cluster headache (CH) attacks occur daily and CH epsiodes may last up to several months (1).
In 1988, MA Moskowitz suggested the cavernous sinus to be the pathophysiological focus of CH (2). All anatomical structures that are supposed to be involved in the pathophysiology of acute CH attacks convene in the cavernous sinus: the ophthalmic division of the trigeminal nerve, parasympathetic nerve fibres running to the supraorbital fissure and sympathetic nerve fibres, branching along the internal carotid artery. The significance of the cavernous sinus for CH pathology was confirmed by clinical observations on patients suffering from symptomatic CH attacks. In numerous reports, symptomatic CH was described to occur in the context of parasellar meningeoma (3), adenoma of the pituitary gland (4) or arteriovenous malformations in the cavernous sinus region (5,6). Moreover, craniometric measures suggest a constitutional narrowness of the cavernous sinus loggia in patients with cluster headache. This anatomic peculiarity could promote the inflammation in this region (7).
The suggestion of a local sterile inflammation in the cavernous sinus of CH patients (2,8,9) was supported by orbital phlebographies performed in CH patients during their active episode. These angiographies showed ipsilateral narrowing of the superior ophthalmic vein and a partial occlusion of the cavernous sinus suggestive of a venous vasculitis (10,11). The inflammation theory was further supported by the clinical observation that corticosteroids are very effective in aborting CH (12,13).
There is no doubt that the unilateral pain syndrome in CH is triggered by ipsilateral activation of the trigeminal nerve system. In animal experiments, electrical stimulation of the trigeminal ganglion results in neurogenic inflammation of the ipsilateral dura mater due to the release of Substance P and calcitionin gene-related peptide (CGRP) from trigeminal terminals (14–16). Substance P mediates an increased vascular permeability at post-capillary venoles via NK1-receptors, thus inducing extravasation of plasma proteins in the surrounding tissue. The protein leakage is semi-quantitatively measured by injecting radiolabelled bovine serum albumin (BSA) into the animal before starting electrical stimulation of the trigeminal ganglion (14).
In an abstract, Goebel and co-workers reported an albumin leakage in the ipsilateral cavernous sinus of CH patients during the active CH episode using radiolabelled human serum albumin (99mTc-HSA) and single photon emission computed tomography (SPECT) (17). As the study remained preliminary, convincing evidence for plasma protein leakage occurring in CH patients is still lacking. Thus, in order to clarify this issue, we decided to test the following hypotheses.
Experimental induction of acute CH results in the extravasation of plasma proteins in the ipsilateral cavernous sinus and is detected by 99mTc-HSA-SPECT.
The 99mTc-HSA uptake in the cavernous sinus is significantly higher during the CH episode as compared with remission.
Methods
Subjects
This study was approved by the Ethical Committee of the University Hospital Charité. All patients participating in our study were under continuous observation at our Tertiary Headache Centre (Charité-Universitätsmedizin Berlin). They were diagnosed according to the criteria of the International Headache Society (18). Written informed consent was obtained from all subjects after the purpose and methods of this study had been fully explained. All subjects underwent a physical and neurological examination. None of the patients had taken prophylactic medication before enrolment in the study. Six patients were examined during the active cluster headache episode. Three patients were treated with verapamil as prophylactic medication after completing the first part of the study. Five of the patients were re-investigated in the remission phase (one was lost to follow-up). The second part of the study was performed at least 4 weeks after the patients had stopped prophylactic medication. Clinical data are given in Table 1.
Clinical data

Six patients were investigated in our study. Patients were asked to assess their headache intensity according to a visual rating scale from 0 (no pain) to 10 (maximum). After the induction of an acute headache attack, two patients were treated with sumatriptan and three patients with oxygen. At the end of part one of the study, three patients (numbers 2, 3 and 4) were treated with prophylactic medication.
Study protocol
Episode
Patients were injected intravenously with 500–600 MBq 99mTc-HSA. 15 min p.i. (= time-point E1, tE1), baseline imaging was performed. To induce cluster headache, patients were injected with 1.2 mg nitroglycerin during online cardiovascular monitoring. In one patient, blood pressure dropped from 130/80 mmHg to 100/60 mmHg after injection of nitroglycerin. In this patient, blood pressure was quickly normalized by fluid intake. Patients remained under clinical observation after the injection of nitroglycerin. The time interval between nitroglycerin injection and the start of a cluster headache varied from 20 to 60 min. Patients remained untreated for 20 min. The pain intensity was assessed using a visual rating scale (0 = no pain, 10 = maximal pain). After this time interval, patients were allowed to abort cluster headache attacks either by inhaling 100% oxygen (patients 2, 4 and 6) or by injecting sumatriptan 6 mg s.c. (patients 1 and 3). As soon as patients were headache free, a second SPECT was performed at tE2 (= time-point E2).
Remission phase
Five patients were re-investigated during the remission phase; one patient was lost to follow-up. Similar to the SPECT series during the cluster headache episode, the first SPECT acquisition was performed 15 min after injection of 500–600 MBq 99mTc-HSA IV at tR1 (= time-point R1). As we could not rule out that the injection of nitroglycerin had an effect on SPECT imaging at time-point tE2, patients also received 1.2 mg nitroglycerin IV in the remission phase. None of the patients developed cluster headache, thus confirming that the patients were in remission. In order to guarantee the comparability of the data, the time interval between tR1 and tR2 (= time-point R2) matched the time interval between tE1 and tE2.
Imaging
MRI
In order to precisely define regions of interest (ROIs) on the SPECT images, detailed anatomical information was obtained by MR-imaging. Using a 1.5 T Siemens Magnetom Vision scanner (Siemens, Erlangen, Germany) equipped with a standard head coil we collected a high-resolution T1-weighted 3D MP-RAGE data set (TR = 9.7 ms, TE = 4 ms, flip angle = 90°, field of view = 256 × 256, voxel size 1.0 × 1.0 × 1.0 mm; 190 slices) in every subject.
Venous MRA consisted of a phase-contrast-sensitive 2D-FLASH sequence in the mid-sagittal plane with a velocity maximum of 10 cm/s (TR 93 ms, TE 11 ms, matrix 256 × 256, FOV 240 mm, slice thickness 45 mm) and a 2D-FLASH time-of-flight sequence of 64 slices of 3 mm thickness in sagittal-oblique orientation, with an angulation of 30° from the sagittal plane (TR 30 ms, TE 9 ms, flip-angle 50°, FOV 250 mm, matrix 224 × 256). Inflowing arterial blood was pre-saturated with an RF pulse at the level of the carotid bifurcation. The time-of-flight slices were reconstructed using a maximum intensity projection.
SPECT
99mTc-HSA (ALBUMOSCINT) was purchased from Bristol Meyers Squibb, Brussels, Belgium. All acquisitions were done for an angular range of 360° with 36 projections per head and a 128 × 128 matrix. The sampling time per projection was 60 s. Images were reconstructed with the iterative ‘ordered subset expectation maximization’ (OSEM) algorithm (19,20) (Software ReSPECT, Scivis Company, Goettingen, Germany). MRI- and SPECT-images were co-registered using MPI tools (Advanced Tomo Vision, Kerpen, Germany). ROIs were drawn on co-registered images. The cerebellum was selected as reference region, and the ipsilateral or contralateral cavernous sinus as target region.
ROI analysis was performed in a blinded fashion. Using the software program MPI tools, the counts/mm3 in the volume of the ipsilateral cavernous sinus, the contralateral cavernous sinus and the cerebellum were assessed. Finally, the counts in the respective ROI were assessed according to the following equation: (target – reference)/reference.
Data analysis
The following parameters were assessed: E1ipsi (counts at tE1 in the ipsilateral cavernous sinus), E1contra (counts at tE1 in the contralateral cavernous sinus), E2ipsi (counts at tE2 in the ipsilateral cavernous sinus) and E2contra (counts at tE2 in the contralateral cavernous sinus). To compare the uptake of 99mTc-HSA in the ipsilateral with the contralateral cavernous sinus at tE1 and tE2, the ratios E1ipsi/E1contra and E2ipsi/E2contra were calculated for each patient, respectively. To test the hypothesis that an extravasation of plasma proteins had occurred in the ipsilateral cavernous sinus after experimental induction of a cluster headache attack, these ratios were compared with each other. As a normality test failed, we performed a Wilcoxon signed rank test (see Table 2).
Results at tE1 (before CH attack) and tE2 (after induction of CH attack)

We compared the uptake of 99mTc-HSA in the ipsilateral and contralateral cavernous sinus at tE1 and tE2. Apart from patient number 6, there was no significant increase of 99mTc-HAS uptake in the ipsilateral cavernous sinus after induction of an acute cluster headache attack (P = 0.21).
To test the hypothesis that cluster headache is characterized by an inflammation of the cavernous sinus during the active episode, which subsides during the remission phase (hypothesis 2), the uptake of 99mTc-HSA in the ipsilateral cavernous sinus during the cluster headache episode was compared with the remission phase. The counts at tE1 and tR1 in the ipsilateral sinus were assessed for each patient (counts at tE1 : E1ipsi, counts at tR1 : R1ipsi, respectively). A paired student's t-test was performed, comparing E1ipsi with R1ipsi (see Table 3).
Comparison of 99mTc-HSA uptake in the active episode and the remission phase

99mTc-HSA uptake in a ROI defined along the anatomic border of the cavernous sinus (ipsilateral cavernous sinus at tE1 and tR1). Patient number 1 was lost to follow-up. No significant decrease of 99mTc-HSA uptake in the remission phase could be observed (P = 0.65).
There have been speculations about nitroglycerin enhancing the venous load in the cavernous sinus due to its vasodilator properties (7). To test whether nitroglycerin had an effect on the venous load of the cavernous sinus, the counts in the ipsilateral and contralateral sinus before (E1ipsi+contra) and after (E2ipsi+contra) the injection of nitroglycerin during the cluster headache episode were compared with the counts in the ipsilateral and contralateral sinus before (R1ipsi+contra) and after (R2ipsi+contra) nitroglycerin injection during the remission phase (see Table 4). Therefore, the ratio E2ipsi+contra/E1ipsi+contra was compared with the ratio R2ipsi+contra/R1ipsi+contra in a paired student's t-test (see Table 4).
Effect of nitroglycerin on 99mTc-HSA uptake

To exclude drug effects, the ratios of counts in the ipsilateral and contralateral sinus after (E2ipsi+contra) and before (E1ipsi+contra) the injection of nitroglycerin during the respective cluster headache episode were compared with the ratios of counts in the ipsilateral and contralateral sinus after (R2ipsi+contra) and before (R1ipsi+contra) nitroglycerin injection during the respective remission phase. Nitroglycerin had no significant effect on our study results (P = 0.57).
Results
Figure 1 shows the intracranial distribution of 99mTc-HSA. Regarding cerebrovascular anatomy, the distribution of the tracer matches best with an accumulation in the venous sinuses (see Figure 2). Figure 3 shows transverse 99mTc-HSA images at the level of the cavernous sinus before (tE1, Figure 3a) and after the induction of an acute cluster headache attack (tE2, Figure 3b). No difference of 99mTc-HSA uptake in the cavernous sinuses is visible. Table 2 illustrates the results of 99mTc-HSA uptake during the cluster headache episode. Comparing the ratios E1ipsi/E1contra of each patient with the respective ratios E2ipsi/E2contra, we found no evidence for an increased 99mTc-HSA uptake in the ipsilateral cavernous sinus after induction of an acute cluster headache attack (P = 0.21). Table 3 shows the results of the ipsilateral cavernous sinus during the active episode (E1ipsi) and during remission (R1ipsi). There was no evidence for an increased E1ipsi as compared with R1ipsi (P = 0.65). Thus, the hypothesis of a protein leakage during the active cluster episode, which subsides in the remission phase, had to be rejected.
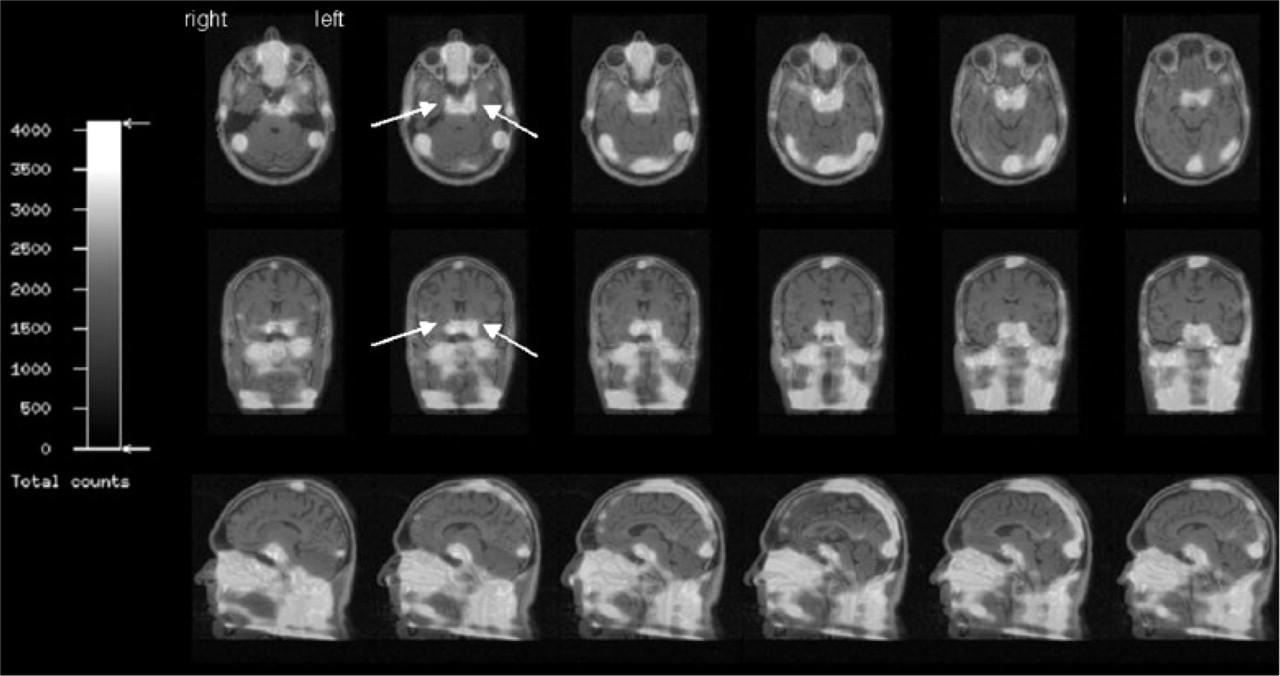
Intracranial distribution of 99mTC-HAS. The figure demonstrates co-registered MRI/SPECT data in transverse (upper row), coronary (middle row) and sagittal (lower row) images of a patient at tE1 (cluster headache episode, before inducing an attack). White arrows illustrate 99mTc-HSA in the bilateral cavernous sinuses. ROI analysis was performed on the basis of co-registered transverse slices and comprised the whole volume of the cavernous sinus. The cerebellum, which showed 99mTc-HSA background activity, served as reference region.

Comparison of MR-venography and 99mTc-HSA-SPECT in a cluster headache patient. (a) Illustrates a transverse 99mTc-HSA image with an inhomogenous uptake of 99mTc-HSA in the transverse sinuses. The 99mTc-HSA uptake is much stronger in the right transverse sinus as compared with the left side. The respective MR-venography (b) illustrates hypoplasia of the left transverse sinus.
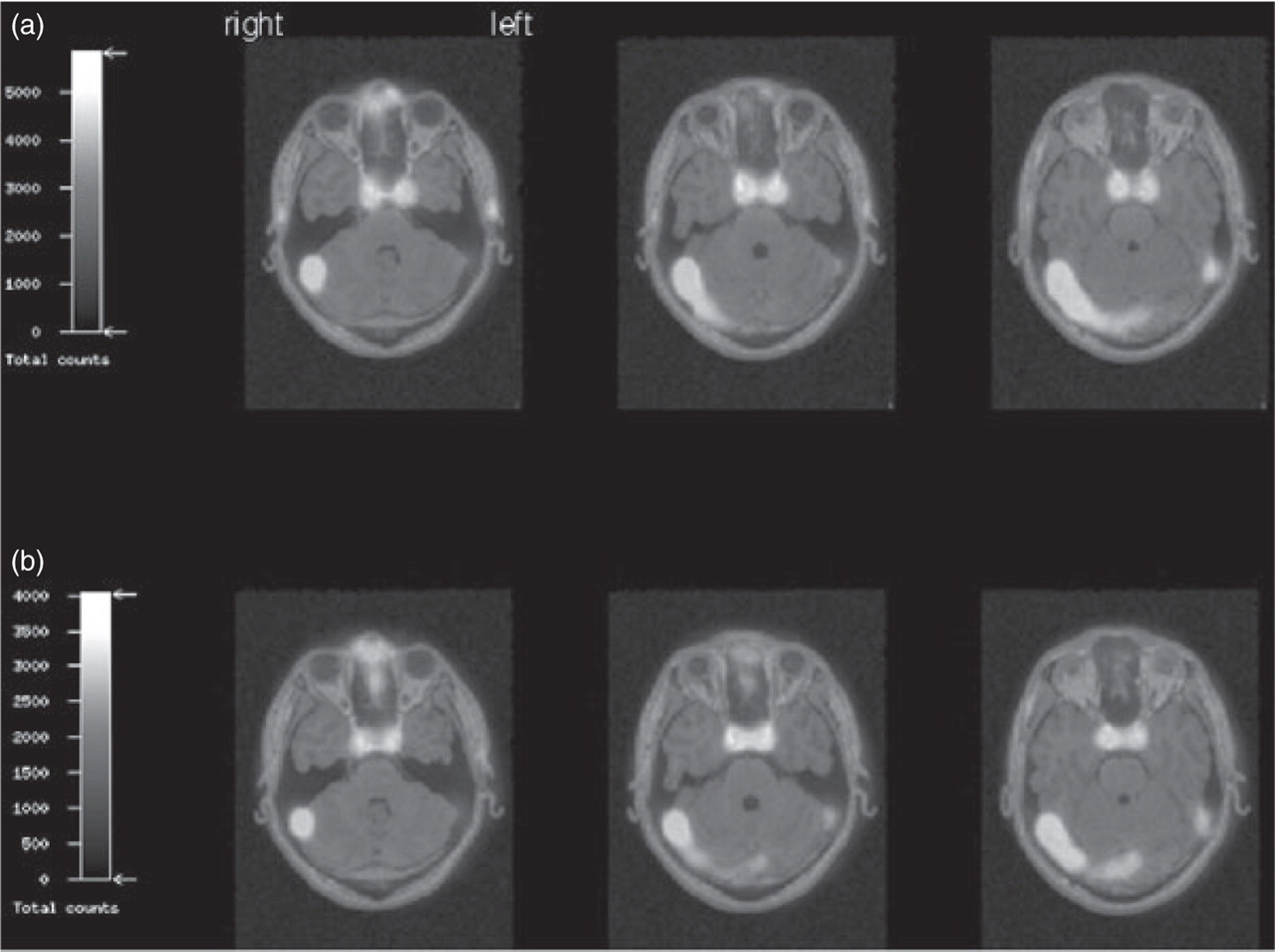
99mTc-HSA-SPECT of a cluster headache patient before and after induction of a CH attack. 99mTc-HSA-SPECT of patient number 4, who developed a severe, left-sided CH attack. ROI analysis did not reveal any significant difference regarding the 99mTc-HSA uptake in the cavernous sinus before (a) (tE1) or after induction (b) (tE2) of the cluster headache attack.
Effect of nitroglycerin on vascular load
To test for confounding effects due to nitroglycerin, we compared the results of 99mTc-HSA uptake before and after nitroglycerin injection during the cluster headache episode with the respective results during remission. Our study results do not confirm a drug effect influencing our study results (P = 0.57, see Table 4).
Discussion
Our study results neither support the hypothesis of an ipsilateral extravasation of 99mTc-HSA in the cavernous sinus during an acute cluster headache attack nor the idea of an inflammatory process during a cluster headache episode that abates in remission.
In the past, there have been considerable attempts to clarify whether neurogenic inflammation is a pathophysiological constituent in trigeminovascular headache disorders such as migraine and cluster headache. The hypothesis of neurogenic inflammation playing a leading role in trigeminovascular headache disorders mainly derives from animal models of trigeminovascular activation. In animal experiments, trigeminal activation results in the release of Substance P and CGRP from trigeminal terminals (15,16,21). To assess whether a similar activation occurs in cluster headache patients, Substance P and CGRP were measured in the ipsilateral jugular vein of CH patients during and after an acute cluster headache attack. These studies revealed that during both spontaneous and provoked cluster headache attacks, CGRP, but not Substance P, is elevated in the ipsilateral jugular venous blood (22). While these studies failed to detect Substance P in the extracranial circulation of CH patients, there has been at least some evidence that Substance P is elevated in the saliva and nasal mucosa of cluster headache patients (23,24).
In 1998, May et al. demonstrated that electrical stimulation of the rat trigeminal ganglion does not only result in dural plasma protein extravasation but also in extravasation of plasma proteins in the retina (25). Like the dura mater and the dural sinuses, the retina is supplied by branches of the ophthalmic division of the trigeminal nerve. Thus, extravasation of plasma proteins in the retina was suggested to serve as an indirect measure for intracranial neurogenic inflammation. In cluster headache patients, May et al. investigated the endothelial permeability in the retina in the acute headache phase using fluoresceine and indocyanine angiography. Contrary to his expectations derived from animal experiments, he could not detect any protein leakage in the retina of patients during their acute headache attacks. Thus, this study failed to confirm the hypothesis of retinal plasma protein extravasation (PPE) occurring in patients suffering from trigeminovascular headaches.
In 2000, Goebel et al. reported an accumulation of 99mTc-HSA in the ipsilateral venous sinuses of CH patients during their active episode, which remitted after prophylactic treatment (17). Our study results are not in accordance with these preliminary findings, which had been published in an abstract. One explanation for the contradictory findings of both studies may be the different mode of data analysis. Goebel et al. did not use SPECT-MRI co-registration for ROI-analysis. However, the reason for the different study results remains a matter of speculation. As the abstract lacks important details regarding methodological issues, we are not able to adequately discuss similarities and differences of both studies.
Methodological considerations
Due to the spatial resolution of SPECT (8–12 mm), the sensitivity of 99mTc-HSA-SPECT to detect plasma protein extravasation is limited. Small amounts of leaked proteins may be missed. In animal experiments of unilateral trigeminal activation, a huge (70–100%) increase of plasma proteins is usually observed in the dura mater of the ipsilateral side (26–28). Regarding the excruciating intensity of CH attacks, one would clearly expect a similar prominent protein leakage in CH patients, which should then be detected by 99mTc-HSA-SPECT.
According to our study protocol, CH attacks were aborted before the second SPECT was performed at time-point tE2. We decided to apply this study protocol for the following reasons. Cluster headache attacks typically develop within a few minutes. From a pathophysiological standpoint, it is very likely that plasma protein extravasation occurs shortly after the beginning of an attack. Thus, a period of 20 min should have been long enough to let plasma protein extravasation evolve. Moreover, it is unlikely that the amount of radiolabelled albumin that had already leaked from the vascular bed into the surrounding tissue during the treatment-free period could have been retroactively influenced or diminished by the cluster headache treatment.
Apart from these pathophysiological considerations, CH therapy was also indispensable for technical reasons. It was unlikely that the patients would have been able to stay motionless during image acquisition for more than 30 min while suffering from a severe cluster headache attack.
Plasma protein extravasation in the dura mater of CH patients
Trigeminal nerve fibres innervate the majority of pain-sensitive structures within the cranium: the dura mater of the anterior and medial fossa, the meningeal vasculature, the intracranial segment of the internal carotid artery and the dural sinuses (29,30,9). Due to the widespread intracranial innervation of the trigeminal nerve, one might propose selecting regions other than the cavernous sinus to study neurogenic inflammation in cluster headache. In animal experiments, plasma protein extravasation induced by trigeminal activation is detected in the ipsilateral dura mater (14). We therefore considered exploring the 99mTc-HSA uptake in the peridural region of CH patients. However, the limited image resolution of SPECT (see above) does not allow a correct attribution of the 99mTc-HSA signal to the epi-/peridural space. Thus, our 99mTc-HSA-SPECT study was not able to clarify this issue.
Involvement of the hypothalamus
During recent years, cluster headache research has changed its pathophysiological focus from the cavernous sinus to the hypothalamus. In 1998, May and co-workers reported an increased regional cerebral blood flow in the hypothalamus of cluster headache patients during provoked cluster headache attacks, indicating that the hypothalamus may be the central generator for CH attacks (31). However, the authors also performed combined 15O-H2O-PET/MRI-studies in order to explore changes in the cavernous sinus during CH attacks. These studies revealed activation in the large intracranial vessels during CH attacks. Corresponding to these findings, MR-angiography revealed a vasodilation of the carotid artery during CH attacks. May and co-workers suggested that the vasodilation in the region of the cavernous sinus does not reflect an inflammatory process, but has to be interpreted in terms of a trigeminoparasympathetic reflex (32,33). The authors concluded that the vascular changes that have been observed in the cavernous sinus of CH patients represent an epiphenomenon, while the source of CH is located in the central nervous system, namely the hypothalamus.
Taken together, clear evidence for an inflammatory process in the cavernous sinus is still lacking. Regarding our own study results, we were not able to demonstrate plasma protein extravasation in CH. Thus, at least, our findings do not support the ‘cavernous sinus theory’ of cluster headache.
Acknowledgements
We thank Andreas Zander and Rüdiger Wenzel for their technical advice.
