Abstract
Little is known about the pathophysiology of cluster headache (CH), one of the most debilitating primary headaches. Interestingly, associations of lung affecting diseases or lifestyle habits such as smoking and sleep apnoea syndrome and CH have been described. Certain genotypes for alpha 1-antitrypsin (α1-AT) are considered risk factors for emphysema. Our aim was to investigate possible associations between common genotypes of the SERPINA1 gene and CH. Our study included 55 CH patients and 55 controls. α1-AT levels in serum and the genotype were analysed. Patients CH characteristics were documented. We could not detect any association between CH and a genotype that does not match the homozygous wild type for α1-AT. Interestingly, there is a significant difference of CH attack frequency in patients who are heterozygous or homozygous M allele carriers. We conclude that the presence of an S or Z allele is associated with higher attack frequency in CH.
Introduction
Cluster headache (CH) as defined by the International Headache Society (IHS) (1) is, in most cases, an episodic (80–90%) and during these episodes a seriously disabling disease. The level of pain described by patients is meant to be one of the most painful conditions one can suffer from. CH is one entity of the trigeminal autonomic cephalalgias; these cephalalgias present with severe and unilateral pain features accompanied by at least one of the following autonomic components ipsilateral to the pain: (i) conjunctival injection/lacrimation; (ii) nasal congestion or rhinorrhoea; (iii) eyelid oedema; (iv) forehead and facial sweating; (v) miosis and/or ptosis; and (vi) restlessness. Beside the acute medication of subcutaneous sumatriptan, the acute treatment recommended is 100% oxygen via a full-face mask with a flow rate of 7–12 l/min over a period of 15–20 min (2). It has been shown that there is an association between CH and central or obstructive sleep apnoea (3–5). Many small studies have shown a positive correlation of CH and tobacco consumption (smoking) (6–11). About 70–80% of CH patients were considered as smokers. The efficacy of oxygen as a therapy as well as the shown associations with lung affecting diseases led us to question if there is any association between CH and alpha 1-antitrypsin (α1-AT) deficiency, a heritable and underdiagnosed condition being found with much higher prevalence in patients suffering from chronic obstructive pulmonary disease (COPD) caused by emphysema. The clinical features can also include acute and/or chronic liver disease as well as, in more rare cases, vasculitis or panniculitis (12–15). α1-AT is produced in the liver and macrophages (16,17). It is a protease inhibitor and helps providing the protease–antiprotease balance, which is crucial especially in the lower parts of the lung where neutrophil elastase is produced by neutrophils (18). Apart from the role as an antiprotease, it is also active as an anti-inflammatory agent through regulating the expression of particular proinflammatory cytokines (19,20), partly by the effect of α1-AT on human neutrophil peptides (21). Impairment in these described mechanisms is considered to be the pathological background for the development of emphysema or is acting as a cofactor for development of COPD in smokers that inherit α1-AT deficiency predisposing allele.
α1-AT is encoded by the SERPINA1 gene in the protease inhibitor (PI) region on chromosome 14q (22,23), and > 100 single nucleotide polymorphisms have been characterized so far. The resulting proteins are classified by their migration speed in gel electrophoresis. Most common forms are F (fast), M (medium), S (slow) and Z (very slow); M represents the wild type. Depending on the type of polymorphism, it is more or less likely to cause emphysema or cirrhosis of the liver (24,25). In cases of α1-AT-related liver disease, intracellular inclusions can be observed, which are due to polymers of α1-AT. According to Mahadeva, the ability to form these polymers is assumed to be Z > S > M.
Due to the increased familial risk for CH (26) and the high prevalence of non-M forms and their suspected role in the lung, an organ whose function seems to affect the characteristics of CH, we hypothesized an interrelation of CH and the genotype for α1-AT.
Methods
All subjects were recruited from the supraregional headache out-patient clinic at the Department of Neurology, University of Münster. Our study population consisted of CH patients and age-matched controls. Patients were diagnosed as episodic CH (diagnosis 3.1.1) or chronic CH (diagnosis 3.1.2) according to the latest edition of the IHS criteria (1). Controls were members of the staff and waiting relatives/friends at the Department of Emergency Medicine of our university hospital. Control subjects with a history of migraine or tension-type headache (frequent episodic or chronic) were excluded. All participants completed questionnaires about onset, frequency and duration of attacks if applicable, comorbidities, etc. The questionnaire was also used to determine exclusion criteria such as diseases of the lung or liver.
After giving informed consent, blood samples for analysis of α1-AT levels as well as characterization of the genotype were drawn. The analysis was performed by the laboratory of the Department of Internal Medicine B, University of Münster. α1-AT concentrations (in mg/dl) were determined by latex-enhanced immunoturbidimetric assays (Roche Diagnostics on a Roche COBAS Integra analyser; Basel, Switzerland) with interassay coefficients of variation of < 5%. Lower limits of detection for the α1-AT assays were 21 mg/dl and reference values were 90–200 mg/dl. All subjects were typed for the M, S and Z.
All subjects were typed for the F, M, S and Z SERPINA1 gene variant by restriction fragment length polymorphism-polymerase chain reaction as described in the literature (27).
Depending on the dataset, we conducted χ 2 tests and Cramer's V statistics, Mann–Whitney or Kruskal–Wallis tests. Significance level was set at P = 0.05.
Results
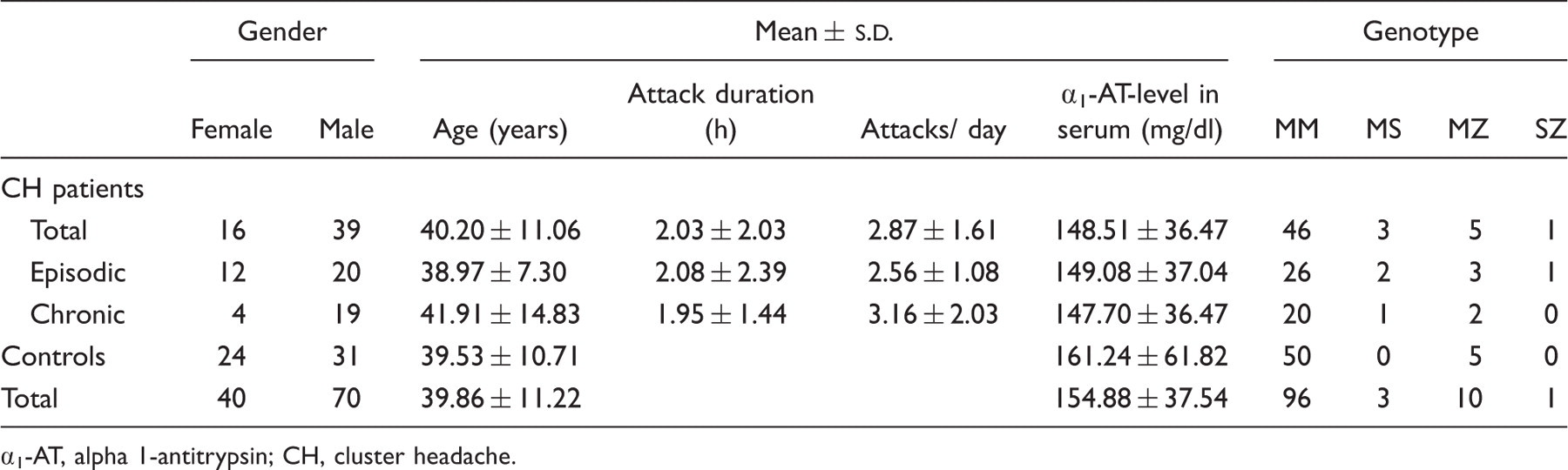
We enrolled 55 CH patients, of whom 32 were suffering from episodic CH and 23 from chronic CH; 55 controls were also included. The groups consisted of 16 women and 39 men in the CH group and 24 women and 31 men in the control group. Two subjects from the control group had serum levels of α1-AT below the protective threshold of 80 mg/dl.
Group characteristics
α1-AT, alpha 1-antitrypsin; CH, cluster headache.
α1-AT levels in serum of CH patients (median = 143.2 mg/dl) and of controls (median = 134.6 mg/dl) did not differ significantly. Furthermore, no significant difference in α1-AT levels in serum between episodic CH patients (median = 142.8 mg/dl) and chronic CH patients (median = 146.0 mg/dl) could be observed.
There was no significant difference between CH patients and controls with respect to the frequency of an MM or not MM genotype (χ 2(1) = 2.63, NS) or between the two groups with respect to the number of M alleles (median for both groups = 2.0). Furthermore, there was no significant difference in the frequency of the MM genotype or in the number of M alleles between the two CH patient groups.
Within the group of CH patients, the number of CH attacks per day in the subgroup of heterozygous M allele carriers (median = 3.0) differed significantly from the subgroup of homozygous M allele carriers (median = 2.0) (P = 0.02).
There were no significant differences in the statistical analysis with respect to sex, and no differences could be detected in the prevalence of any allele between chronic and episodic CH patients, or in the influence on attack frequency between these (data not shown).
Discussion
We have been able to show that there is no association between CH and the prevalence of one of the common genotypes for α1-AT that does not meet the homozygous wild type (MM). Furthermore, we have shown that CH is not associated with α1-AT deficiency. Interestingly, we were able to show that, depending on the presence of the homozygous wild type, there is a difference in the number of CH attacks per day for patients. This means that the non-MM genotype of α1-AT is associated with more frequent CH attacks. The importance of this finding remains unclear, due to the fact that we cannot adopt a known mechanism for this shown association. It is known that α1-AT has anti-inflammatory effects; however, no significant signs of systemic inflammation in CH have been detected (28,29). A role of α1-AT in the model of neurogenic inflammation would be possible, and the presence of a dysfunctional polymer could either trigger attacks or shift the threshold while not causing the disease itself. Reporting these results and hypothesizing about a connection, it has to be considered that recent data support the understanding of CH as a disease originating from central structures and mechanisms in which the hypothalamus is involved (30–32). No differences between carriers of the homozygous M type and heterozygous S or Z carriers could be observed. The ZZ genotype is held responsible for most α1-AT deficiency-related diseases. There is evidence that the MZ genotype predisposes for emphysema and cirrhosis (33,34). It has also been shown that even the heterozygous form can lead to a relevant change of α1-AT. However, a coherence of the Z allele and attack characteristics of CH patients could not be shown in our study. Although the prevalence of the ZZ, SZ and MZ genotypes has been shown to differ between countries and continents, there are large numbers of individuals who have inherited one of these types (35–37). Therefore, our results should be of interest not only for the European CH patients.
The non-MM genotype possibly represents a risk factor for triggering attacks. For further specification, additional studies will be necessary. Therefore, we cannot make a statement about which of the different SERPINA1 genotypes is responsible for the coherence indicated by our study.
In our study, the proportion of episodic vs. chronic CH does not reflect the prevalence ratio found in a general population, in which only about 17% of patients would have been expected to suffer from chronic CH (whereas they represent 42% of the CH patients included in our study) (38). We explain this by the fact that our clinic is a tertiary headache clinic and therefore the percentage of rare forms that are hard to treat is higher. Smoking habits were not evaluated and therefore not taken into consideration in statistical analysis. There has also been a higher number of Z allele carriers than expected in the CH and control groups. Calculated prevalence of PI-MZ in Europe is 1/36, PI-SZ = 1/1051 and PI-MS = 1/16 (35). However prevalences in different regions within continents vary, they are similar between Europe, Australia and the USA (37). Other than the relatively small study population, we cannot provide an additional explanation for the relative overrepresentation of Z allele carriers. Since this study was exploratory, the number of subjects was not based on a power analysis.
One other point of criticism of our study is that we did not have subjects of all possible genotypes. This is explained by the prevalence of carriers of F allele or a SS or ZZ genotype (35). For our study the F form was expected to have no impact because it is meant to have normal functionality (39). However, one must consider that the groups were relatively small, and associations with a relatively low effect size would appear only in much larger study groups. Since we were interested in associations relevant to a large group out of the CH patients, associations of a smaller degree or applicable only to very small subgroups lay outside our focus.
