Abstract
The role of the parasympathetic nervous system in the pathogenesis of migraine is disputed. The headache-eliciting effect of the parasympathetic neurotransmitter, vasoactive intestinal polypeptide (VIP), and its effect on cerebral arteries and brain haemodynamics has not been systematically studied in man. We hypothesized that infusion of VIP might induce headache in healthy subjects and cause changes in cerebral haemodynamics. VIP (8 pmol/kg per min) or placebo (0.9± saline) was infused for 25 min into 12 healthy young volunteers in a crossover, double-blind design. Headache was scored on a verbal rating scale from 0 to 10, regional cerebral blood flow (rCBF) was measured with single-photon emission computed tomography and 133Xe inhalation and mean flow velocity in the middle cerebral artery (VmeanMCA) was measured with transcranial Doppler ultrasonography. The headache was very mild with a maximum score of 2 and described as a pressing or throbbing sensation. Five participants developed headache during VIP and one during placebo. During the infusion, a significant drop in VmeanMCA was seen for VIP compared with placebo (P < 0.001), but the effect quickly waned and no difference was found when comparing the time between 30 and 120 min. In addition, no significant difference in the diameter of the MCA could be found during the infusion. No significant differences in rCBF (P = 0.10) were found between VIP and placebo. A marked dilation of the superficial temporal artery was seen (P = 0.04) after VIP in the first 30 min but no difference was found when comparing the time between 30 and 120 min. We found no difference in mean arterial blood pressure between VIP and placebo days but the heart rate increased significantly on a VIP day compared with a placebo day (AUC0–30min, P < 0.001). Plasma VIP was significantly higher on a VIP day compared with placebo (AUC0–80min, P < 0.001). These results show that VIP causes a decrease in VmeanMCA without affecting rCBF. In spite of a marked vasodilator effect in the extracranial vessels and increased plasma VIP, healthy subjects developed only a very mild headache.
Keywords
Introduction
Animal (1–3) and human (4–6) studies have suggested that activation and sensitization of nociceptors around extra- and intracranial vessels may be a primary source of pain in migraine. The parasympathetic nervous system has for a long time been implicated in the pathogenesis of migraine (7). There has recently been increasing focus on the role of the parasympathetic system in migraine (8–10). It has been suggested that parasympathetic outflow to cephalic vasculature may trigger activation and sensitization of perivascular sensory afferents (10) and thereby migraine pain.
Parasympathetic efferent nerves release various neuropeptides such as pituitary adenylate cyclase activating polypeptide (PACAP) and vasoactive intestinal polypeptide (VIP) to regulate cerebrovascular tone and haemodynamics of the brain (11). Immunohistochemical studies have shown that VIP is located in the cerebral, meningeal and temporal arteries in close relation to the adventitia and adventitia–medial borders of the vessel wall (12–15). Furthermore, in vitro and in vivo studies have demonstrated that VIP acts as a powerful vasodilator in various species (16), including man (16–19). VIP may be increased in the cranial venous blood during attacks in a subgroup of patients with migraine associated with prominent autonomic symptoms (20) and in patients with cluster headache (21). It remains unknown, however, whether VIP may cause headache and its effect on brain haemodynamics has not been systematically studied in humans. We hypothesized that infusion of VIP may induce headache in healthy subjects and that VIP-induced headache may be associated with dilation of intra- and extracerebral blood vessels. To test this hypothesis, we performed a double-blind, placebo-controlled, crossover study in normal human volunteers and studied the effect on headache and haemodynamic variables.
Design and methods
Pilot experiment
Before the main study, we conducted an open-label pilot study in three healthy subjects(two male and one female) to find the optimal dose of VIP with respect to tolerable adverse events and detectable changes in mean blood flow velocity of the middle cerebral artery (VmeanMCA) measured with transcranial Doppler. The subjects received intravenous VIP in stepwise increasing doses of 6, 8 and 10 pmol/kg per min. Each infusion lasted 25 min followed by a 60-min wash-out. The results of the pilot study showed that a dosage of 8 pmol/kg per min was well tolerated and a led to a decrease in VmeanMCA. Therefore, we chose this dose for the main study.
Main experiment
We recruited 14 healthy subjects (seven male, seven female), mean age 24 years (range 22–28 years). Exclusion criteria were: a history of migraine or any other type of headache (except episodic tension-type headache more than once a month); any daily medication apart from oral contraceptives; serious somatic or psychiatric diseases. The study was approved by the Ethics Committee of the County of Copenhagen (KA03099) and was undertaken in accordance with the Helsinki Declaration of 1964, as revised in Edinburgh in 2000. All subjects gave informed consent to participate in the study.
Experimental design
In a double-blind, placebo-controlled, crossover design, the subjects were randomly allocated to receive 8 pmol/kg per min VIP or placebo (isotonic saline) over 25 min on 2 days separated by at least 1 week. All subjects reported to the laboratory at 08.00 h headache free. The intake of coffee, tea, cocoa or other methylxanthine-containing foods or beverages was not allowed for the last 8 h before the start of the study. All procedures were performed in a quiet room at a temperature of 25°C. The subjects were placed in the supine position and a venous catheter (Venflon®) was inserted into the right and left antecubital vein for blood sampling and drug infusion. The participant then rested for 30 min before baseline measurements of blood pressure, heart rate (HR) and ECG were performed and the infusion started, using a time and volume controlled infusion pump (Braun Perfusor, Melsungen, Germany). Headache intensity, VmeanMCA, superficial temporal artery diameter, end-tidal partial pressure of CO2 (PetCO2), adverse events and vital signs were recorded at T−10, T0 and then every 10 min until 120 min after the start of infusion. Cerebral blood flow (CBF) measurements by single photon emission computed tomography (SPECT) were performed at T0, T20 and T60. The subjects were discharged from the hospital after finishing the measurements and asked to complete a headache diary every hour until 10 h after discharge. The diary included headache characteristics and accompanying symptoms according to the International Headache Society (22), any rescue medication taken and adverse events. Subjects were allowed to take rescue medication of their own choice at any time.
Headache intensity
Headache intensity was recorded on a verbal rating scale (VRS) from 0 to 10 [0, no headache; 1, a very mild headache (including a feeling of pressing or throbbing); 5, moderate headache; 10, worst imaginable headache] (23).
Cerebral haemodynamics
Middle cerebral artery blood flow velocity
VmeanMCA was recorded bilaterally by transcranial Doppler (TCD) with hand-held 2-MHz probes (Multidop X; DWL, Sipplingen, Germany). Fixed probes were not used since they may cause discomfort and even headache (24). A time-averaged mean over 4 s or approximately four cardiac cycles was used as final measure for each time point. Identification of the MCA and marking of reproducible fix points were done as previously described (25). All recordings were done by the same skilled technician. PetCO2 was recorded simultaneously with the TCD measurements using an open mask that caused no respiratory resistance (ProPac Encore®; Welch Allyn Protocol, Beaverton, OR, USA). The measurements at T0, T20 and T60 were performed immediately after regional cerebral blood flow (rCBF) measurements to obtain corresponding values. Flow in the territory of the MCA (rCBFMCA) is proportional to the product of VmeanMCA and cross-sectional area of the MCA. Therefore changes in cross-sectional area could be calculated as ΔA = (rCBFMCA(2) · VmeanMCA(2) −1) · (VmeanMCA(1) · rCBFMCA(1) −1) − 1 (26).
Diameter of the superficial temporal artery
Diameter of the frontal branch of the superficial temporal artery (STA) was measured by a high-resolution ultrasonography unit (Dermascan C; Cortex Technology, Hadsund, Denmark: 20 MHz, bandwidth 15 MHz) as previously described (27, 28).
Cerebral blood flow
The examination was performed with the subject in the supine position, in quiet surroundings with eyes closed and ears unplugged. Four markers were drawn on the skin to ensure accurate positioning in each acquisition. PetCO2 was measured during each examination (Datex Normocap 200, Roedovre, Denmark). CBF was measured with 133Xe inhalation (Ceretronix XAS SM 32C, Randers, Denmark) and SPECT, using a brain-dedicated gamma camera (Ceraspect; DSI, Waltham, MA, USA). The system uses a stationary annular NaI crystal and a fast rotating collimator. Flow was calculated in each pixel based on the clearance curve, output was the ki value (29). To obtain CBF values, a partition coefficient (λ) of 0.85 was used. Calculation of flow in the perfusion territories of the major cerebral arteries was performed by fitting standard vascular regions of interest (ROI) on transactional slices of the brain. rCBFMCA was calculated as mean of the right and left side, since there was no statistically significant difference between the sides.
Vital signs
HR and blood pressure were measured every 10 min using an auto-inflatable cuff (ProPac Encore®; Welch Allyn Protocol). ECG (Cardiofax V; Nihon-Kohden, Shinjuku-ku, Tokyo, Japan) was monitored on an LCD screen and recorded on paper every 10 min.
Plasma concentration of VIP
Blood samples for VIP measurement were collected at baseline and 20, 25, 60 and 80 min after start of infusion in ice-chilled tubes containing 50 IU of heparin and 500 kIU aprotinin (Trasylol, Bayer, Germany) per ml blood. Plasma was separated by centrifugation (1500
Data analysis and statistics
We defined an immediate phase as a period from 0 to 30 min (0–30 min) and a postinfusion phase as a period from 30 to 120 min (30–120 min) based on pharmakinetics of VIP. VIP has a short half-life in plasma of only a few minutes (32). The binding of VIP to one of the VIP receptors results in the activation of adenylate cyclase (AC), thereby causing marked increases in cellular cAMP production (33). Even though VIP may be present in plasma for only a short time, it may also have long-term effects after it has been eliminated from plasma. This seems to be the case for the effect of VIP on glycogen metabolism in astrocytes, where the effect is divided in two: an early action (within minutes) caused by cAMP and a delayed phase, caused by gene transcription (34).
The primary end-points were differences in area under the curve (AUC) for headache score (AUCheadache score), VmeanMCA (AUCVMCA) and STA (AUCSTA) between groups in the period 0–30 min. Additional explorative end-points were differences in AUCheadache score, AUCMCA and AUCSTA between groups in the period 30–120 min and differences in peak response between groups at time of maximum plasma concentration of VIP. The secondary end-points were differences in AUCheart rate and AUCMAP between groups in the period 0–30 min. Additional explorative end-points were differences in AUCheart rate and AUCMAP between groups in the period 30–120 min and differences in peak response (HR and MAP) between groups at time of maximum plasma concentration of VIP. Changes in PetCO2 was evaluated over 0–120 min, because VMCA should be corrected with e0.034 for each mmHg change in PetCO2 (35).
All values are presented as mean ± SD, unless otherwise stated. We calculated AUC according to the trapezium rule (36) to obtain a summary measure and to analyse the differences in response (VmeanMCA, headache score, diameter of STA, mean blood pressure, HR, PetCO2, VIPplasma) between VIP and placebo. Analysis was performed with a paired, two-way t-test, except headache scores, where data are presented as medians and quartiles and tested with Wilcoxon signed rank test. Baseline was defined as T−0 before the start of infusion of each dose.
Continuous variables were analysed for changes over time for each dose separately with univariate analysis of variance with the fixed factors volunteer and time. To reduce mass significance the following time points were selected for analysis (T0, T20, T60). If overall differences were found, Dunnett's test was applied to characterize which time points were different from baseline. We tested for period and carry-over effects with the use of independent samples t-test on the difference and the sums, respectively, of the AUC of the first and the second treatment. Headache was tested for period effect and carry-over effect with Mann–Whitney test.
All analyses were performed with SPSS for Windows 12.0 (Chicago, IL, USA). Five percent (P < 0.05) was accepted as the level of significance.
Results
There were no differences at baseline for any variables (Table 1).
Baseline values (±SD) in 12 healthy subjects [cerebral blood flow (CBF) data for 10 subjects) on two trial days: mean velocity of blood flow in the middle cerebral artery (VMCA), global CBF (CBF) and regional CBF in the territory of the middle cerebral artery (rCBFMCA), end-tidal partial pressure of CO2 (PetCO2) measured during transcranial Doppler measurements, mean arterial blood pressure (MAP), heart rate (HR), plasma VIP and diameter of superficial temporal artery (STA)

P-value: paired t-test.
Twelve subjects completed the study on both trial days. One subject was excluded because we could not obtain intravenous access and one subject was excluded on his second trial day because of a vasovagal reaction 5 min after start of VIP infusion. Due to data loss, the CBF data for one subject could not be included and the CBF data for another subject had to be excluded from statistical analysis because of inexplicably high CBF values on the placebo day, probably due to technical problems. Thus, 10 subjects were left for the CBF analysis. Seven blood samples for the analysis of plasma VIP were not done, one sample on a VIP day, six samples on a placebo day, due to either haemolysis or technical problems.
There was no carry-over or period effect for baseline values of headache, MCA, CBF, rCBFMCA, STA, MAP, HR or plasma VIP.
Headache
The AUC0−30min on the VIP day, 0 (0–23.8), was significantly higher than on the placebo day, 0 (0–0) (P = 0.04) (Fig. 1). During the immediate phase (0–30 min), five subjects reported headache on the VIP day and one subject on the placebo day. There was no difference in the AUC30−120min recorded during the postinfusion period between VIP and placebo days (P = 0.72). During the postinfusion period (30–120 min), three subjects reported headache on the VIP day and two on the placebo day.

Individual and median headache scores on a verbal rating scale (VRS). There were significantly higher pain responses after vasoactive intestinal polypeptide (VIP) (▪) compared with placebo (□) (P = 0.04). Thick lines are median pain scores.
We found no difference in the AUCdelayed headache recorded from 120 min to 12 h after infusion between VIP and placebo days (P = 0.75). Delayed headache was reported by six subjects, three on each trial day (Table 2). On the placebo day, the delayed headache lasted 3–6 h and none of the subjects had either a history of frequent headache or family history of headaches. On the VIP day, the delayed headache lasted 1–10 h and none of the subjects had a history of frequent headaches or family history, except subject 2, who reported that her mother had had frequent headaches. One subject reported migraine-like delayed headache on a VIP day (Table 2).
Clinical characteristics in 12 healthy subjects

a, Peak headache during VIP infusion; b, peak headache post-VIP infusion; c, peak headache during placebo infusion; d, peak headache post placebo infusion; Migraine like, migraine-like headache.
Middle cerebral artery and regional cerebral blood flow
During the immediate phase (AUC0−30min), VMCA decreased significantly on the VIP day compared with the placebo day (P < 0.001) (Fig. 2). The peak decrease in VMCA occurred 20 min after the start of VIP infusion. At the end of the infusion (T20), the mean decrease in VMCA was 14.4 ± 4.8% on the VIP day and 2.2 ± 6.0% on the placebo day compared with baseline (Fig. 2). The mean difference in response between VIP and placebo at T20 was −11.3% (95% confidence interval −16, −6.6). There were no changes after VIP for global CBF (AUC0−60min, P = 0.10) or for rCBFMCA (AUC0−60min, P = 0.10) when compared with placebo measured during CBF acquisition (Fig. 3). Analysis of variance (

Individual and mean flow velocities in the middle cerebral arteries (VMCA) assessed by transcranial Doppler ultrasonography. Infusion of vasoactive intestinal polypeptide (VIP) (▪) resulted in significant decrease in VMCA during immediate phase (0–30 min) compared with placebo (□) (P < 0.001). Thick lines show mean values.

Individual and mean global cerebral blood flow (CBF) and regional flow in the territory of the middle cerebral artery (MCA) (rCBFMCA) measured with 133Xenon inhalation and single photon computed tomography in 10 healthy subjects. There were no changes in global CBF or rCBFMCA between vasoactive intestinal polypeptide (VIP) (▪) and placebo (□) days (P > 0.05). Global CBF and rCBFMCA remained unchanged over time on both trial days (P > 0.05). Thick lines show mean values.
Using the corresponding rCBF and VmeanMCA values, the changes in MCA diameter were calculated to be 8.4 ± 6.2% between baseline and T20, 2.2 ± 5.2% between baseline and T60 for VIP, 4.6 ± 9.3% between baseline and T20 and 3.4 ± 4.7% between baseline and T60 for placebo. No significant difference was found between VIP and placebo (T20, P = 0.29, T60, P = 0.62).
During the postinfusion period (AUC30−120min), there was no difference between VIP and placebo days (P = 0.17) and
There were no differences in the PetCO2 recordings during TCD scans between VIP and placebo days, either during the immediate (AUC0−30min, P = 0.82) or postinfusion phases (AUC30−120min, P = 0.63).
Superficial temporal artery
During immediate phase (AUC0−30min), the diameter of STA increased significantly on the VIP day compared with the placebo day (P = 0.04) (Fig. 4) and

Individual and mean diameter of the superficial temporal artery (STA) assessed by a high-resolution ultrasonography. During the immediate phase (0–30 min), the diameter of the STA increased significantly after vasoactive intestinal polypeptide (VIP) (▪) compared with placebo (□) (P = 0.04). Thick lines show mean values.
Mean arterial blood pressure and heart rate
We found no difference in MAP between VIP and placebo days during either the immediate phase (AUC0−30min, P = 0.08) or the postinfusion phase (AUC30−120min, P = 0.65) (Fig. 5).
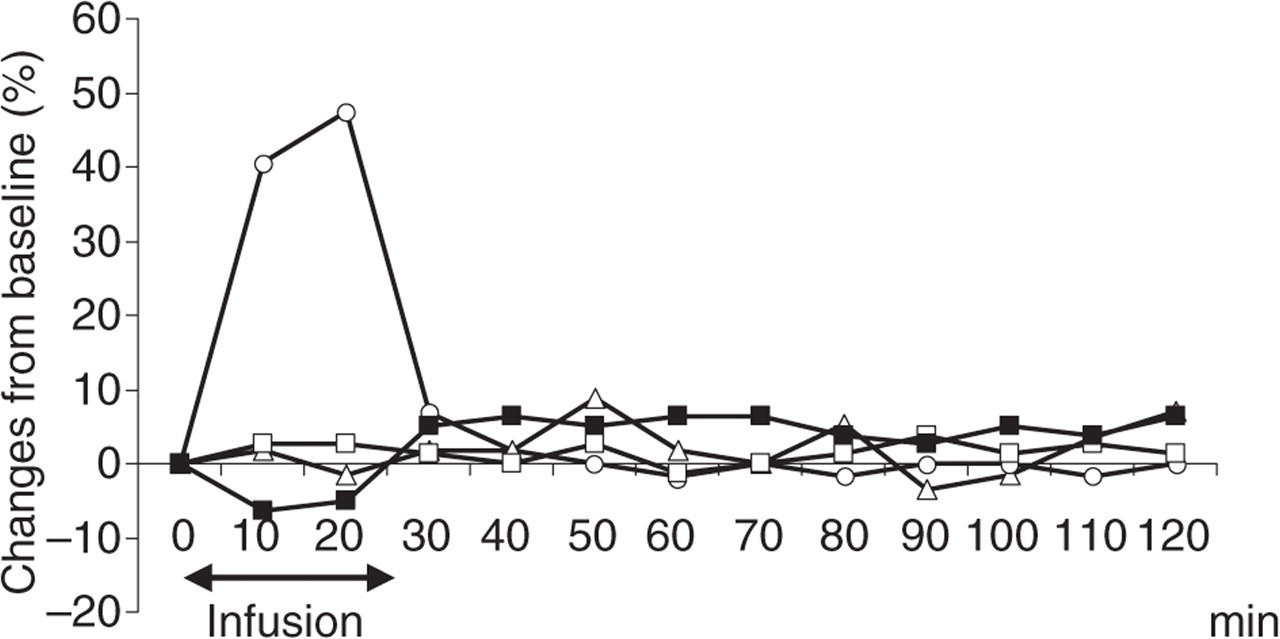
Mean percent change from baseline in mean arterial blood pressure (MAP) and heart rate (HR) during and after infusion of vasoactive intestinal polypeptide (VIP) (▪, MAP; ○, HR) and placebo (□, MAP; ▵, HR). MAP tended to decrease on the VIP day compared with the placebo day, but not statistically significantly (P = 0.08). A post hoc analysis
Plasma VIP
Plasma VIP was significantly higher on VIP days compared with placebo (AUC0−80min, P < 0.001) (Fig. 6).

Individual (black) and mean (red) plasma concentration of vasoactive intestinal polypeptide (VIP) after infusion of VIP (▪) and placebo (□). Infusion of VIP resulted in a significant increase in plasma VIP compared with placebo (P < 0.001). The peak plasma concentration of VIP was measured at the end of the infusion (T25 min) and was (±SD) 111.4 ± 47.8 pmol/l. Thick lines show mean concentrations.
Adverse events
One subject was excluded on his second trial day because of a vasovagal reaction 5 min after the start of VIP infusion. He reported a feeling of dizziness, heat sensation and mild dyspnoea. The blood pressure dropped and the HR rose but without crossing any of the protocol safety limits. ECG and auscultation of heart and lungs were unremarkable. Immediately after cessation of infusion he felt better and 5 min later his blood pressure and HR returned to baseline values. He was observed for an additional 60 min and discharged. There were more reported adverse events such as flushing, palpitation, heat sensation and headache on VIP days than on placebo days (Table 3).
Adverse events recorded and reported during 0–30 min and 30–120-min periods

Groups compared and tested with McNemar test.
Discussion
The major outcome of the present study is that systemic administration of VIP induces only a very mild and short-lasting headache, a long-lasting dilation of the extracerebral superficial temporal artery and a short-lasting increase in the diameter of the middle cerebral artery when calculated from the decreased blood flow velocity and unchanged global and regional CBF.
Can VIP induce headache?
Experimental vasodilation of the internal carotid artery and MCA may induce focal head pain in humans (37). In migraine patients, an attack was associated with MCA dilation and both pain and dilation were reversed by sumatriptan (38). A model has been proposed in which migraine is the result of activation of postganglionic parasympathetic neurons in the sphenopalatine ganglion, resulting in vasodilation and local release of inflammatory molecules that activate meningeal nociceptors (39). Activity in the parasympathetic nervous system decreases cerebrovascular tone (11, 40) and one of the most abundant peptides in the parasympathetic efferents, VIP, acts as a powerful vasodilator in animals and humans (16, 18, 41, 42). Immunohistochemical studies have shown that VIP is present in the parasympathetic nerve fibres surrounding human temporal (43–45) and middle cerebral artery (19). VIP receptors are also found in these arteries (46, 47). The binding of VIP to its receptors activates the adenylate cyclase, increases cellular cAMP (33) and causes vasodilation (48). However, VIP-induced vasodilation may not always be caused exclusively by cAMP (49). In addition to its direct effect, nitric oxide may contribute to parasympathetic vasodilation by regulating VIP release (50, 51), possibly acting as a second messenger system for VIP and acetylcholine (52). Another possible mechanism of generating headache is a direct activation and sensitization of sensory afferents around cerebral blood vessels by exogenous VIP. VIP has been found in rat spinal ganglia (53) and VIP receptors occur in the trigeminal ganglia (54). VIP may play an important role in sensory transmission in the dorsal root ganglia of the spinal cord (55) and in spinal dorsal horn neurons (56). Intrathecal administration of VIP facilitates a spinal nociceptive reflex in the rat (57) and VIP antagonists decrease the firing rate of DRG cells in animal models of neuropathic pain (58). Based on these data, we speculated that systemic administration of VIP might cause headache by inducing vasodilation and activation or sensitization of nociceptors. The present study shows, however, that although VIP induced a marked dilation of extracerebral arteries and a non-significant dilation of the MCA, it causes only a very mild and short-lasting headache. These results raise the question, why did VIP fail to induce significant dilation of the MCA and caused so little headache?
It could be argued that the VIP dosage used in the present study was too low to elicit headache and marked vasodilation of the MCA. In the present study, the plasma concentration of VIP was monitored and found to be high compared with endogenous levels (mean 111.4 pmol/l) during infusion. The effect of exogenous VIP is mediated through VPAC1 and VPAC2 receptors, which have a binding affinity (IC50) of 1–4 n
Slow passage of VIP across the blood–brain barrier (BBB) is another possibility. We observed a mild and short decrease in VMCA and no change in CBF, while the dilation of the STA was more pronounced and longer lasting. This difference in response between two vascular beds could be caused by the BBB since the STA is outside the barrier. If the BBB is bypassed and VIP injected intracisternally in dogs, a persistent vasodilation (120 min) is reported in the basilar artery (42). In mice, 0.15% of the injected VIP could be found in the brain 10 min after intravenous administration (60). In rats, after intracarotid infusion of a VIP analogue coupled to a transport vector, 3.2% of the perfusate concentration was found in the brain after 10 min (61). In baboons, intracarotid infusion of VIP after hypertonic opening of the BBB caused an increase in CBF of 37%, while no effect was seen under normal circumstances (62). In rats, VIP linked to a delivery vector increased CBF by 60% (63). Collectively, these data indicate that the BBB is very tight in arterioles, but less tight in the MCA. Dilation of STA seems without headache-inducing effect, as previously described (64, 65), while the mild and short-lasting effect on MCA corresponds to a mild and short-lasting headache.
In conclusion, this is the first double-blind cross-over study aimed at exploring the headache-eliciting effect of intravenous administration of the parasympathetic neurotransmitter VIP in healthy volunteers. In spite of marked vasodilator effect in the extracranial vessels and increased plasma VIP, healthy subjects developed only a very mild headache. Whether infusion of VIP may induce a more pronounced headache or migraine in migraineurs should be addressed in future studies.
Acknowledgements
The authors thank lab technicians Kirsten Brunsgaard, Lene Elkjær and Juliano Olsen for their dedicated and excellent assistance. The study was supported financially by University of Copenhagen, the Danish Headache Society and the Cool Sorption Foundation.
