Abstract
The aim of this study was to characterize the temporal course of phosphene thresholds (PT) using transcranial magnetic stimulation (TMS) in control subjects and in subjects with migraine and to observe whether changes in PT over time can predict a subsequent migraine attack. PTs were measured in 16 migraineurs [nine with aura (MA) and seven without aura (MoA)] and nine controls five times over an approximately 10-week period. Mean PTs were not significantly different between migraineurs and controls; however, there was a trend in MA showing lower thresholds. The minimum threshold values were also smaller in MA subjects than in MoA or control subjects. Generally, PTs had higher variance in migraineurs than in controls, revealing a significant increase in standard deviation of PTs in MA subjects. There was no significant difference of thresholds from the first to the last stimulation in controls and in MoA subjects, but the 3rd, 4th and 5th measurements of MA subjects were significantly lower than their first measurements. Four migraineurs experienced headache within 1 day after one of the measurements. They had either very low or very high PTs compared with the PT values which were not followed by a migraine attack. Our results imply that migraineurs show a higher variability among PT measurements over time than controls, revealing unstable excitability levels in these patients. Additionally, both particularly high and low PTs might predict a subsequent headache in some individuals.
Introduction
Studies of transcranial magnetic stimulation (TMS)-induced phosphenes in persons with migraine have produced conflicting results. Aurora et al. (1, 2) have demonstrated lowered phosphene thresholds (PTs) and increased ability to induce phosphenes in migraineurs with aura (MA), suggesting increased excitability of the visual cortex. However, lower prevalences of stimulation-induced phosphenes in MA subjects compared with healthy controls have been found (3) as well as the same prevalence but increased PTs, both of which suggest decreased excitability of the visual areas (4). Mülleners et al. (5) described reduced mean PT in MA subjects and migraine patients without aura (MoA) compared with controls but not different prevalences among groups concerning phosphene perception. Battelli et al. (6) observed lower PTs, not only for stationary but also for moving phosphenes in migraineurs. Finally, Valli et al. (7) have found no differences between migraineurs and controls concerning PTs.
Some of these discrepancies could be due to methodological differences, either TMS device or patient population dependent. For example, the size of the stimulated cortical area depends on the size and shape of the coil: larger cortical areas can be stimulated with circular coils than with figure-of-eight coils. Other technical differences that may contribute to varying results are the types of magnetic pulses (biphasic, monophasic, direction of the current (8)) which are sometimes not described in the papers. Additionally, it is possible that PTs in migraineurs show higher temporal variabilities than those in controls. In a recent study PTs were evaluated over 3 weeks in MA, MoA, patients with menstrual migraine and in controls (9). Lower PTs were found in migraineurs (but not in subjects with menstrual migraine) than in controls, with a consistent decrease in PTs from the first to the last measurement in all groups. The reason for this is that the subjects had probably learned to recognize even less bright, weaker phosphenes. The aim of the present study was to evaluate the stability of the PTs over time, including a practice session and five measurements with a break of at least 1 and a maximum of 3 weeks, and to observe if changes in PTs (i.e. a higher or lower threshold) predict a subsequent headache.
Methods
The study involved 16 subjects with migraine (mean age 25.8 years, range 18–54 years, SD 8.74; three men, nine with aura) and nine control subjects (mean age 28.6 years, range 20–42 years, SD 6.2; three men). Controls were recruited from the university staff. They had no family or personal history of migraine. Subjects were selected only if they could see phosphenes during the training session. All of the subjects gave their written informed consent according to the Declaration of Helsinki. The study was approved by the Ethics Committee of the University of Göttingen.
Migraine was diagnosed by expert neurologists according to the International Headache Society criteria (2003). Subjects with migraine were not on any prophylactic medication. Nine patients suffered from right-sided migraine, four from left-sided and three bilateral. The experiments were conducted in headache-free intervals and the measurements were conducted and implemented in the evaluation only if the subjects had no headache for at least 3 days before and after the experiment. In four subjects who had migraine attacks within 1 day after the experiment, the measurements were repeated later if possible or if they were not involved in the main analysis. These values were used for the prediction of migraine attacks. The frequency of migraine attacks was between 3 and 78/year (mean 25.5). Seven subjects reported migraine in their family history. There were various medications preferred by the subjects in case of a migraine attack: rizatriptan n = 1, sumatriptan n = 2, aspirin n = 4, paracetamol n = 2, ibuprofen n = 4, aspirin, paracetamol and caffeine n = 1, diclofenac n = 1 and propylphenzon, paracetamol and codeine n = 1.
TMS was performed with biphasic pulses with a posterior-anterior current flow in the coil, using a standard slightly bent figure-of-eight coil (outer radius of one half-coil was 9 cm) connected to a MagPro stimulator (Dantec S.A., Skovlunde, Denmark) with a maximum stimulator output of 147 A/µs. The handle of the coil was pointed upwards. The stationary PT was determined first in every subject. The subject was blindfolded. Single pulses were applied 2–4 cm above the inion initially with 50% of the maximum stimulator output. If the subject did not perceive phosphenes at this stimulation intensity, it was increased in steps of 5% until the subject perceived a phosphene, maximally at up to 80% of stimulator output. If the subject still failed to perceive a phosphene, the coil was moved 1 cm up or 1 cm left or right and the procedure was repeated. The location of the coil where stable phosphenes were perceived was measured and marked with a pen. PTs were determined as follows: starting from the stimulation intensity at which the subject perceived a stable phosphene in the same form and at the location every time when the same scalp area was stimulated, the intensity was reduced in steps of about 5% until phosphenes were no longer perceived. It was then increased again in steps of about 2% to the minimum intensity at which the subject could perceive a phosphene. These minimum final values were accepted only if the phosphenes were stable sensations, appearing in the same form at the same location in at least three cases when the same area was stimulated. At the beginning of the experiments a practice session was included. The PTs were measured over time using five measurements with a break of at least 1 and a maximum of 3 weeks. All of the measurements were done at the same time of day.
For all of the subjects mean, maximum and minimum PTs were calculated at each measurement time point individually (A/µs). Additionally, group means, minimum–maximum values and SDs were calculated. Repeated measurements of two-way or one-way
Results
Table 1 shows the mean and SDs of PT values in all of the three groups at the different time points. The mean PTs were not significantly different among the three groups (F 2,22 = 0.714, P = 0.50). The effect of the consecutive measurements (time points) was significant (F 4,88 = 4.087, P = 0.004), while the interaction between the groups and time points was not significant (F 8,88 = 1.525, P = 0.16). However, there was a trend towards lower thresholds among patients with aura (Fig. 1). We did not find a significant threshold decrease from the first trial to the last in the controls or in the MoA group; however, in the MA group there was a significant decrease in the 3rd, 4th and 5th measurements compared with their values in the first test (P < 0.05).

Mean phosphene thresholds levels (with SEs) in controls (•), migraine with aura (MA) (▵) and without aura subjects (▪) measured at the five consecutive time points with a break at least 1 week. Note the significant decline of phosphene thresholds in MA subjects. ∗Significant changes.
Mean (SDs) phosphene threshold values in all of the three groups at the different time points

Over all the time points minimum thresholds were 49.9 (SD 20.5) in the control, 38.18 (SD 19.68) in the MA and 46.4 (SD 14) in the MoA groups. Maximum thresholds were 63.26 (SD 19.6) in the control, 60.98 (SD 20.1) in the MA and 65.67 (SD 8.13) in the MoA groups. The threshold variabilities were greater for the minimum values. For the minimum thresholds the differences between migraineurs and controls were not statistically significant (F2,22 = 1.067, P = 0.36). However, there was a trend in MA subjects towards lower minimum values. The maximum thresholds were not statistically significantly different among the three groups (F2,22 = 0.162, P = 0.85). Differences of SDs were not significant among the three groups (F2,22 = 1.894, P = 0.174), but the difference between controls and MA subjects was significant (F1,16 = 4.27, P = 0.046)) (Fig. 2).

Minimum, maximum and SD phosphene threshold (PT) values (with SEs) in controls, in migraine with aura (MA) and migraine without aura (MoA) subjects. ∗Significant difference.
Four of the migraine patients experienced migraine within 1 day after one of the measurements. We compared the PT values to those that were not followed by headache. One subject had their individually highest (72.4 vs. 32.58), two subjects their lowest PTs compared with the mean PTs of the headache-free measurements (20.1 vs. 64.88; 38.0 vs. 57.18).
Figure 3 shows the individual PT values in the three groups at all measurements points.
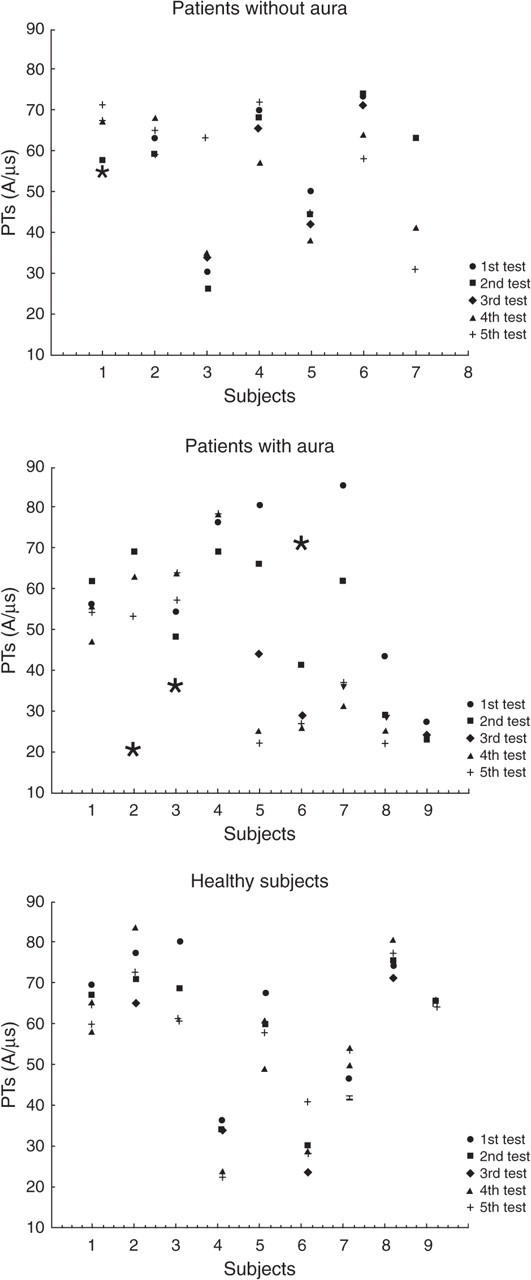
Individual phosphene threshold (PT) values in the three groups at the five measurement points. The stars show PT measurements that were followed by migraine attacks.
Discussion
We evaluated consecutive TMS-induced PT measurements and found that migraineurs, mainly subjects with aura, show higher intra- or interindividual variability concerning PTs over time than healthy subjects. Additionally, we observed that migraineurs who experienced headache after one of the measurements had higher or lower PTs compared with measurements not followed by a migraine attack.
A previous study (9) has also characterized the temporal course of PT changes in migraineurs and in controls. The authors observed lowered PTs over time in all of the groups, generally lower PTs in migraineurs than in controls (except subjects with menstrual migraine). In contrast, we did not find significantly lowered PTs over time between our control subjects and MoA patients, probably because the subjects had a practice session before the experiment and they had learned to recognize even weaker phosphenes. However, in MA subjects there was a decrease after the 3rd, 4th and 5th measurements compared with the first measurement, suggesting that in MA subjects the PT values are less stable even after practice. Nevertheless, it is difficult to explain why MA subjects had only the ability to ‘improve’ regarding phosphene perception over time.
Generally, we did not find significantly lower PTs in migraineurs compared with controls, in agreement with Valli et al. (7) but in discordant with other studies (1, 2, 5, 10). However, there was a trend towards lower thresholds among patients with aura. It has been suggested that in subjects with aura the cumulative physiological effects of many aura episodes and the occurrence of spreading depression (11) may cause stronger ultrastructural changes in the visual system (12) and therefore the visual changes observed in MA subjects are more pronounced that in MoA subjects.
Additionally, the individual PT variability over days or a week were higher in migraineurs (mainly MA subjects) than in controls. Therefore we made the assumption that the individual's high or low threshold might be a risk factor for an impending headache in some patients. Indeed, four of our subjects who had a migraine attack within 1 day after one of the measurements had higher or lower PTs compared with PT values which were not followed by migraine. The reason for this vulnerability is not clear, yet it is possible that regulatory mechanisms that stabilize neuronal activity within a homeostatic range in the cortex (13) are not working sufficiently before migraine attacks in both directions. A previous study found no relationship between a higher or lower threshold and the appearance of a subsequent headache in migraineurs (9). However, that study compared the number of days after/before the minimum–maximum PT values in MA and MoA subjects, which were not significantly different in the two groups, while we compared the individual PT values before the headache with those which were not followed by headache. Further studies including more patients would be necessary to clarify the reason for this finding.
Previous studies have demonstrated a change of cortical excitability during the menstrual cycle: an excitatory neuronal effect was associated with oestradiol and a neuronal inhibition was associated with progesterone (14, 15). However, these studies evaluated the excitability of the motor cortex using a paired-pulse paradigm in healthy women and not PTs in migraineurs. Additionally, there was no significant change in the motor threshold between the follicular and luteal phases. It is likely that PTs do change during the menstrual cycle, but there is no published study on this issue. In this study we did not record (similarly to many previous studies) in which phase of the menstrual cycle the actual measurement was performed. Only two of our subjects had menstrual migraine (migraine attacks occurring 2 days before to 5 days after the onset of menstruation). The PT values of these two subjects were not different from those of the other migraineurs, as in the study of Young et al. (9). Surprisingly, two of our male subjects had higher PT variability. Additionally, the control female subjects who had at least one or two menstruations during the time of the experiment showed minimal PT variability. Nevertheless, further studies should clarify this question.
Several studies using a wide range of tasks have suggested that migraine patients may have visual cortical anomalies between attacks. More intense illusions to grating patterns (16, 17) as well as greater visual masking effects (18), reduced perceptual suppression of simple targets (12, 19), altered motion perception (20) and increased motion after-effect duration (21) were observed by different studies. Differences in visual cortical function between migraineurs and controls have also been revealed using TMS. Lower PTs (1, 5, 10, 22) even after 1-Hz inhibitory repetitive TMS (rTMS) were detected (23). In contrast, other studies have found lower prevalences of phosphenes and increased PTs in migraineurs (3, 4). Electrophysiological studies have also implied abnormal primary visual processing by measuring the amplitude of visual evoked potentials (24, 25). From these studies an altered interictal excitability of the cerebral cortex in migraine, most pronounced in the visual areas, was proposed. However, it is not clear if these visual cortical anomalies are due to cortical hyper- or hypoexcitability. Our aim was not to resolve this hypo- vs. hyperexcitability problem. However, our consecutive measurements imply that the excitability of the visual cortex in migraineurs changes over a wider range than it does in controls, at least with regard to PT measurements. If in a given experiment one patient is measured usually only once it could result in either hyper- or hypoexcitability compared with controls. Therefore the conflicting results reported by different studies could be due not only to the different technical parameters but also to the temporal characteristics of the cortical excitability change in migraine.
Acknowledgements
We thank Petra Karacsonyi for the English corrections. This study was supported by the German Ministry of Research and Education within the Kompetenznetz Schmerz (FKZ: 01EM0117).
