Abstract
The issue of interictal excitability of cortical neurons in migraine patients is controversial: some studies have reported hypo-, others hyperexcitability. The aim of the present study was to observe the dynamics of this basic interictal state by further modulating the excitability level of the visual cortex using transcranial direct current stimulation (tDCS) in migraineurs with and without aura. In healthy subjects anodal tDCS decreases, cathodal stimulation increases transcranial magnetic stimulation (TMS)-elicited phosphene thresholds (PT), which is suggested as a representative value of visual cortex excitability. Compared with healthy controls, migraine patients tended to show lower baseline PT values, but this decrease failed to reach statistical significance. Anodal stimulation decreased phosphene threshold in migraineurs similarly to controls, having a larger effect in migraineurs with aura. Cathodal stimulation had no significant effect in the patient groups. This result strengthens the notion of deficient inhibitory processes in the cortex of migraineurs, which is selectively revealed by activity-modulating cortical input.
Introduction
The pathophysiological defect of non-hemiplegic migraine is still far from being understood. Cortical spreading depression (1), trigeminovascular inflammation (2), magnesium deficiency (3), dopaminergic hypersensitivity (4) and altered energy metabolism possibly due to mitochondrial defects (5) can all contribute to migraine pathogenesis. Due to the lack of an animal model to investigate neocortical processes, the study of cortical excitability in man is one of the closest approaches to pathophysiology at present. So far, an altered interictal cortical excitability has been claimed in migraine, but similar methodological approaches have led to opposing results, and similar results to opposing explanations. Most authors have found cortical hyperexcitability (6, 7), but data supporting hypoexcitability have also been reported (8–10).
Habituation is seen as a major factor contributing to these conflicting results. It is regarded as a fundamental, possibly protective mechanism of cortical processing. It has been shown to be present in visual, auditory, somatosensory and nociceptive domains. Cortical evoked potentials tend to decrease with increasing stimulus repetition in healthy subjects. In migraineurs, evoked potential experiments have revealed dishabituation in all of the studied domains. Visual evoked potentials (VEP), somatosensory evoked potentials (11), laser evoked nociceptive responses (12), auditory evoked potentials (AEV) (8) and mismatch negativity (13) studies have invariably shown dishabituation in migraineurs as opposed to normal habituation in healthy volunteers. While the results of this dysfunction are clear, most authors have explained them as due to hyperexcitability or impaired inhibitory processes of cortical circuitry (14). In favour of cortical hypoexcitability (15), interictal dishabituation was diminished by means of 10 Hz repetitive transcranial magnetic stimulation (rTMS) in migraine patients. Ten Hertz rTMS increases the excitability of cortical neurons in healthy subjects. Moreover, the authors reported disinhibition after 1 Hz rTMS in healthy volunteers, a finding very similar to the interictal VEP findings in migraineurs. Consequently, dishabituation has been interpreted as the result of a low baseline excitability in which stimulus repetition does not cause decreased response amplitudes due to a floor effect. This view has received further support by decreased thalamo-cortical activity between attacks, as shown by comparing the high-frequency oscillations in somatosensory evoked potentials in migraineurs and age-matched healthy volunteers (16). Experiments investigating phosphene threshold (PT), a representative of visual cortex excitability, have also led to controversial conclusions. Aurora et al. (17) found a much higher proportion of phosphene inducibility in migraineurs compared with age-matched healthy volunteers. Not only was the prevalence higher, but the PT was also significantly lower in migraineurs. Similar results have been achieved by stimulating the motion-sensitive visual area V5, where moving phosphenes can be elicited (18) with a lower threshold in migraine headache patients. Other groups have used very similar methodological approaches, but achieved opposite results. Afra et al. (19) reported lower prevalence of induced phosphenes in migraineurs, which favours cortical hypoexcitability. Possible reasons for these contradictory results, including different patient selection, paradigms and technical differences, have been discussed extensively (20). Most visual psychophysical studies point to enhanced interictal cortical excitability in migraineurs. High interictal sensitivity to light has been observed in patients compared with controls (21). Migraineurs are more prone to see stronger visual illusions (22) and have a lower critical fusion frequency threshold (23). This finding has been interpreted as a shorter cortical stimulation silent period in migraineurs.
A different approach to the study of excitability changes is the use of noisy visual stimuli. Migraineurs show impaired motion detection ability when they have to percieve coherent motion in a noisy background (6). However, without distractors, patients' performance was superior to that of healthy volunteers. This pattern of results is comparable to healthy volunteers' performance, when the motion-sensitive area MT/V5 was stimulated by means of anodal transcranial direct current stimulation (tDCS), which is known to cause reversible cortical hyperexcitability. While these observations are in favour of interictal hyperexcitability in migraine headache patients, other studies have shown a hypofunctioning visual system in migraineurs. Benedek et al. (24) investigated contrast sensitivity at different spatial frequencies and found decreased contrast sensitivity at low spatial frequencies in migraineurs without aura, compatible with decreased functioning of the magnocellular visual pathway in migraine.
We tested the range of up-and-down regulation of cortical excitability by using 'brain polarization' by tDCS. We assumed that a shift in the response range of PTs either to cathodal or anodal stimulation as measured by TMS reflects a shift in baseline excitability. tDCS applied through the skull directly modulates the excitability of the motor (25–27), somatosensory (28, 29) and visual (30–33) cortices of human subjects. In the visual cortex, tDCS has been shown to induce excitability changes that can be measured reliably by TMS-elicited PT. Animal studies suggest that cathodal tDCS decreases baseline firing rates of cortical neurons, probably by hyperpolarizing the axonal membrane of neurons. In contrast, anodal stimulation results in a reverse effect (34–36). The effect of stimulation can last up to 60 min after 10 min of transcranial stimulation (26, 27). In this study we administered 10 min anodal, cathodal and sham stimulation over the occipital cortex and compared baseline PTs with those measured after tDCS in control subjects and migraineurs with and without aura.
Methods and materials
The study involved nine healthy volunteers (HV) (mean age 28.6 years, range 20–42 years, SD 6.2; three men) fulfilling the following criteria: no metallic implants or electrical devices, no previous history of neurological or psychiatric disorders, drug abuse or alcoholism and no concurrent medication. The patient group included 16 migraine patients [mean age 25.8 years, range 18–54 years, SD 8.74; three men, nine with aura (MA)] diagnosed by a trained neurologist and fulfilling the criteria of the International Headache Society. Disease duration was between 1 and 12 years (mean 5.7). Attack frequency ranged between 0.1 and seven per month (mean 3.1). None of the subjects had had preventive antimigraine medication in the preceding year. All measurements were done in attack-free periods preceding and following at least 3 days of the experiment confirmed by interviews and telephone calls. In subjects who had had migraine attacks within 3 days after the experiment, the measurements were repeated later if possible, or were excluded from the analysis.
All subjects gave their written informed consent. The study conformed to the Declaration of Helsinki and was approved by the Ethics Committee of the University of Göttingen. The subjects in this study participated in other experiments.
Stimulation
TMS was performed with biphasic pulses with an initial current flow in the coil away from the handle using a slightly bent MC-B70 figure-of-eight coil (outer radius of one half-coil, 9 cm) and a MagPro stimulator (Dantec S.A., Skovlunde, Denmark). The maximum stimulator output was 147 A/μs. Subjects were seated in a comfortable armchair in a slightly illuminated room (10 cd/m2). Subjects were not dark adapted, but blindfolded during the phosphene measurements. Single pulses were delivered at 50% stimulator output on Oz (according to the 10–20 EEG coordinate system) five times with the handle pointing upwards. This resulted in an initial caudo-cranial current flow in the brain. The intertrial interval was approximately 5 s. If the subject did not perceive a phosphene at this intensity, it was increased in steps of 5% until the subject perceived a phosphene, maximally up to 80% of stimulator output. If the subject still failed to perceive a phosphene, the coil was moved 1 cm up or 1 cm left or right and the procedure was repeated. If the subject reliably perceived a phosphene at all of the trains applied and the phosphenes appeared in the same form at the same location, stimulation intensity was reduced in steps of 5% until the subject no longer reliably perceived a phosphene. It was then increased again in 2% steps until the minimum intensity at which the subject could perceive a phosphene was established. Subjects were asked to describe the shape, colour and position of phosphenes. The location of the coil on the skull was marked by a pen and the coordinates recorded in relation to Oz. After 2 min rest, the PTs were determined at this location three times with the method described above. The averages of these values were taken as baseline. After 10 min of anodal, cathodal or sham tDCS, PTs were determined at the same location three times.
tDCS was delivered by a battery-driven constant current stimulator (Schneider Electronic, Gleichen, Germany) using a pair of electrodes in a 5 × 7 cm water-soaked synthetic sponge. For cathodal stimulation the cathode was placed at Oz, the reference over Cz. For anodal stimulation the current flow was reversed. In the real tDCS conditions the current was applied for 10 min with an intensity of 1.0 mA. Constant current flow was controlled by a voltmeter. During sham stimulation the current was turned on only for 5 s at the beginning of the sham session and then turned off in a ramped-shaped fashion. None of the subjects was able to distinguish real tDCS from sham tDCS sessions. For each subject the separate experimental conditions were performed at least 1 week apart. The order of stimulation was counterbalanced both in HV and in patients.
Statistical analysis
Values of PT were entered into a three-way repeated measures analysis of variance (
Results
Baseline PT
The PT values obtained before and after 10 min tDCS are shown in Fig. 1 and reported in Table 1. The baseline PT of MA subjects tended to be lower compared with that of normal controls, although not statistically significantly different (Students' t-test: anodal, t = −1.54, P = 0.14; cathodal, t = −1.62, P = 0.12; sham, t = −1.41, P = 0.17). Baseline values were constant throughout sessions in every group, although it was shown in our laboratory previously that PT variability is significantly higher in migraineurs than in controls (37) and this variability might forecast forthcoming attacks. As measurements which were followed by headache attacks were excuded from the study, we rule out the possibility that this variability affected our results.

Transcranial magnetic stimulation-elicited phosphene threshold (PT) values in healthy volunteers (HV, ▪), migraine patients without aura (MoA, •) and migraine patients with aura (MA, ○) before and after anodal, cathodal and sham stimulation. Anodal stimulation significantly decreased PTs in all of the groups, whereas cathodal stimulation significantly increased PTs only in controls.
Transcranial magnetic stimulation-elicited phophene thresholds (PTs) before and after anodal, cathodal and sham stimulation for healthy volunteers (HV), migraine patients without aura (MoA) and migraine patients with aura (MA)
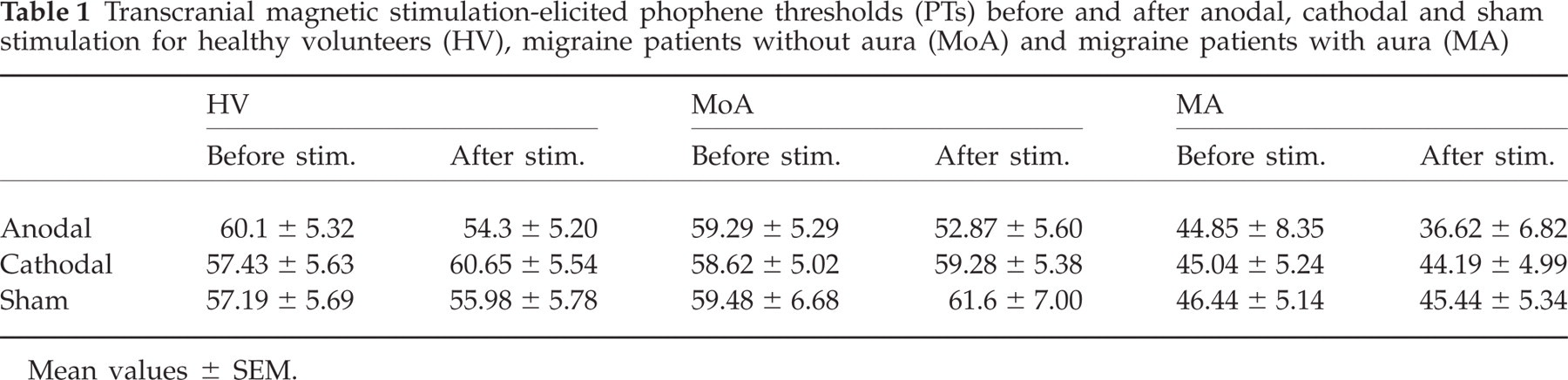
Mean values ± SEM.
The effect of stimulation
Three-way
Comparisons between groups
When the groups were compared with paired Student's t-tests, there were no significant differences observed between controls and the migraine without aura (MoA) group (0.53 < P < 0.91 for the three stimulation types, respectively). However, there was a significant difference between controls and MA subjects for anodal (t = 2.058, P < 0.05) and cathodal (t = 2.22, P < 0.05) stimulation. None of the groups differed from each other in the sham condition and in none of the groups did the before and after sham treatment values differ significantly.
Comparison within groups
Anodal stimulation increased cortical excitability significantly in all of the groups with a different magnitude (HV, t = 3.33, P < 0.01; MoA, t = 4.97, P < 0.005; MA, t = 3.68, P < 0.005). After cathodal stimulation, significantly increased PTs were observed only in the HV group (t = −3.29, P < 0.01); the PT values of MoA and MA subjects were not significantly affected by the stimulation (MoA, t = −0.49, P = 0.64; MA, t = 0.31, P = 0.76). Sham stimulation did not alter cortical excitability in any group (HV, t = 1.4, P = 0.2; MoA, t = −1.82, P = 0.12; MA, t = 0.74, P = 0.48).
Discussion
We have shown that the pattern of excitability changes induced by tDCS is different in migraineurs, particularly in those with aura compared with HV or MoA subjects. Anodal stimulation led to an increase of cortical excitability as measured by a decrease of PT in all experimental groups, which was largest in MA subjects. Cathodal stimulation had an opposite effect in HV, as expected based on previous experiments in our laboratory (30). In the MoA and MA groups it did not alter cortical excitability.
The present experiment, using a new experimental approach, adds data to the long-debated issue of interictal cortical excitability in migraineurs. PT was measured before and after 10 min of tDCS, which is known to alter cortical excitability bidirectionally in a polarity-dependent manner. Measuring the magnitude of changes of occipital cortex excitability induced by tDCS in migraineurs and contrasting them with those measured in HVs enabled us to estimate imbalances between excitation and inhibition in the patient group. Migraine aura, which is a prominent, transient cortical dysfunction found in migraineurs, is related to the experimental phenomenon known as cortical spreading depression (CSD), first reported by Leao (38). Observations in migraineurs with aura support this theory. Functional magnetic resonance imaging during induced visual aura showed a BOLD signal intensity progression at a speed which very closely resembled that of CSD (39). This study also showed the characteristic hyperactivity, followed by hypoactivity in the visual cortex. In animals CSD can be induced by stimulating the cortex mechanically or chemically. In humans the triggering event is unknown, but triggering an aura is possible in some subjects by flickering light, which is a strong activator of the visual system. In migraineurs a subnormal level of inhibitory processes during extensive cortical activation might induce a spreading hyperactivity.
Additionally, genetic studies and ion channel dysfunction caused by different mutations also help to explain the induction of CSD in migraine (40). CACNA1A gene mutations are found in 50–75% of cases with familiar hemiplegic migraine (FHM). Na-K ATPase, alpha2 (ATP1A2) gene is also a mutation found in some forms of migraine and in FHM (41). The products of these genes play a role in the normal functioning of neurons and control neuronal excitability. While there is no general agreement on the role of these mutations in migraine pathogenesis, indirect observations support it. Ambrosini et al. (42) treated migraineurs with acetazolamide, which is known to increase Ca2+ channel function. They observed an improvement in clinical measures and normalization of electromyography variables. Since the same type of channel is known to be present in neuromuscular transmission, these data indicate that Ca2+ channel abnormality indeed exists in non-FHM migraineurs. Furthermore, a recent study (43) found mutation of the neuronal voltage-gated sodium channel gene SCN1A in FHM. The excessive firing of the neurons expressing this mutation could initiate and facilitatate CSD and related aura symptoms.
Our findings are consistent with these results. We show that migraineurs, mostly those with aura, tend to respond to excitatory stimulation with an increased amount of PT reduction and fail to increase the threshold after cathodal stimulation, unlike normal subjects. Cortical hyperexcitability can be due to a higher activity of primary excitatory neurons or to reduced activity or efficacy of primary inhibitory neurons. Here, we support the hypothesis of cortical hyperexcitability in migraine to the latter, similar to some previous studies (44, 45). This dysfunctional pattern might also explain, at least in part, the conflicting results of previous studies: for example, why abnormal cortical excitability is not always manifested at rest, but is revealed only in dynamic states when the sum of extrinsic excitatory and inhibitory influences shifts from baseline. A study by Brighina et al. (46) has found that an inhibitory 1-Hz rTMS over the visual cortex of migraineurs results in increased excitability. The authors imply that increased excitability in migraineurs is probably due to the failure of cortical inhibitory mechanisms. Furthermore, a recent study has observed similar effect of 1 Hz rTMS over the motor cortex in migraineurs: the intracortical facilitation decreased significantly in controls after rTMS administration, whereas it increased in patients (47). Taking into account that the effect of 1 Hz rTMS depends on the preexisting level of cortical excitability in healthy subjects (48), these results are not suprising. In our study we found no increase in excitability after cathodal stimulation in migraineurs, although cortical excitability did not decrease as it did in controls. These differences are probably due to the different methods we applied: whereas rTMS induces externally triggered changes in the neuronal spiking pattern and interrupts or excites neuronal firing in a spatially and temporally restricted fashion, tDCS modulates the spontaneous firing rates of neurons by changing resting membrane potential. Therefore, tDCS is more like a subthreshold stimulation technique compared with rTMS.
Another possible explanation for the above-mentioned paradoxical effect (and our present results) could be a lower preactivation (hypoexcitability) level of visual cortical excitability and the impossibility of obtaining supplementary inhibition as implied by previous studies (8, 17). However, the baseline PT values of our migraine subjects tended to be lower than those of control subjects, as in previous studies (15, 41), Although the difference was not significant, probably due to the low number of subjects, the trend was clear and suggests more hyperexcitability than hypoexcitability.
The above-mentioned deficient inhibitory mechanisms explain why the cerebral cortex of some migraineurs is susceptible to CSD and, consequently, development of aura symptoms. Additionally, the fact that migraineurs without aura in our study group showed less pronounced deviations from the normal group raises the possibility that MA and MoA are more distinct than previously thought. A recent study (49) examined healthy subjects and different groups of migraineurs with single-fibre electromyography, a method very sensitive for access to neuromuscular transmission. It was suggested that abnormal neuromuscular transmission in an aura group of patients was due to the dysfunction of the P/Q Ca2+ channel, which was coded by the CACNA1A gene. However, another study did not support this hypothesis (50). Concerning epidemological studies, a recent population-based survey of twins with migraine (51) has implied that MA and MoA are distinct disorders. However, given the overlap of neurological symptoms and non-mutual exclusivity of aura symptoms recently studied (52), including twins and their parents, it is not yet proved that MoA and MA subtypes are aetiologically distinct. Further studies are necessary to clarify this question.
Taking all data together, we have shown inhomogeneity of migraine patients by investigating the dynamics of cortical excitability after external modulation. In our study, inhibitory dysfunction in MA was more pronounced than in MoA or control subjects. The results also strengthen the notion of deficient inhibitory processes in the cortex of MA patients, which was selectively revealed by activity-modulating cortical input.
Acknowledgements
This study was supported by the German Ministry of Research and Education within the Kompetenznetz Schmerz (FKZ: 01EM0117).
