Abstract
Migraine can induce ischaemic stroke, and is considered an independent risk factor for stroke in the young. To date, the nature of the link between migraine and stroke is essentially unknown. Forty-five children were studied. Homocysteine levels (fasting and post methionine load), vitamin B12 and plasma folate levels, factor V Leiden, factor II G20210A, methylenetetrahydrofolate reductase (MTHFR) C677T and A1298C mutations were examined. Compared with controls, patients with migraine had higher levels of post-methionine load homocysteine values (19.5 ± 4.9 vs. 16.9 ± 1.9; P = 0.025) and significantly lower folate levels (5.8 ± 2.6 vs. 7.5 ± 2.1; P = 0.002). We found a trend toward an increased risk of migraine in subjects carrying a homozygous mutant genotype for MTHFR C677T and MTHFR A1298C polymorphisms. Genetic prothrombotic conditions do not seem to be related to migraine in the young, whereas the biochemical differences between migrainous patients and controls are an appealing topic for further investigation.
Introduction
Migraine can induce ischaemic stroke and is considered an independent risk factor for stroke in the young (1, 2). Stroke is a rare complication of migraine with aura (MA), and it has also been described in patients with migraine without aura (MoA) (3).
The average annual incidence of migraine-induced infarction in adults is about 3.36/100 000, but in the absence of other risk factors it drops to 1.44/100 000 (4).
The International Classification of Headache disorders, 2nd edn (ICHD II) published in 2004 (5) defines ‘migrainous cerebral infarction’ as an ischaemic brain lesion demonstrated by neuroimaging, occurring during the course of a typical MA attack. According to the ICHD II, the following conditions must be present: (i) the attack should be typical of previous attacks, except that one or more aura symptoms persist for >60 min in a patient with MA; (ii) neuroimaging demonstrates ischaemic infarction in a relevant area; and (iii) it cannot be attributed to another disorder.
Data concerning migrainous stroke in children and adolescents are quite scarce. Furthermore, the paediatric migrainous stroke cases reported in the literature do not fulfil all ICHD II criteria, since most of them had never had previous MA, or previous migraine at all (6). However, it is well known that an attack of complicated migraine in children can be the beginning of a history of migraine (7).
The pathogenesis is still unknown, but it is believed that a common mechanism underlies the two diseases. Several physiological hypotheses have been suggested concerning what may lead young migraineurs to ischaemic stroke (with regards to vascular, neuronal or coagulation abnormalities) (8). Some authors detected an increase in platelet activation both during and between migraine attacks (9). Antiphospholipid antibodies have been reported in patients with migrainous stroke (10).
Besides the above-mentioned underlying triggering factors, acquired or inherited prothrombotic risk factors may play a role in the paediatric population with ischaemic cerebrovascular events.
A higher incidence of prothrombotic conditions, such as resistance to activated protein C (ACPR) as well as factor V Leiden G1691A (FVL) and factor II G20210A (PRT) mutations, have been found in migraine patients compared with controls (11, 12).
Recently, it has been suggested that folate metabolism may also be involved in the pathogenesis of migraine, with or without aura (13–15). Moreover, it is well established that homozygosity for the common C677T mutation in the methylentetrahydrofolate reductase (MTHFR) gene, which causes thermolability of the enzyme, is a risk factor for ischaemic stroke (16).
Another polymorphism in cis in the same gene, the A1298C mutation, seems to be a risk factor for migraine when associated with C677T in double heterozygosity, or by itself in homozygosity (17).
To date, no definite conclusion has been reached and the nature of the link between migraine and stroke is essentially unknown (18).
A group of patients attending the Out-patient Clinic of the Paediatric Neurology Unit at the G. Gaslini Institute were prospectively studied in order to investigate the incidence of metabolic alterations and molecular prothrombotic factors in migrainous children and teenagers. Homocysteine levels [fasting and post methionine load (PML)], vitamin B12 and plasma folate levels, FVL, PRT, MTHFR C677T and A1298C mutations were examined.
Patients and methods
Forty-five patients, 18 males (40%) and 27 females (60%), aged 5–17 years (mean 11.6 ± 2.7 years) affected by migraine, were consecutively enrolled between March 2000 and June 2003.
Diagnosis of migraine was made according to the 1st edn of the International Classification of Headache Disorders (ICHD-I) (19), although bilateral pain was admitted, and the minimum headache time requirement was dropped to 1 h, on the basis of the proposals made by several authors, which have now been accepted in the recent ICHD II classification (20–22).
Of our patients, 33 suffered from MA (12 males; 36.4%, aged 4.6–16.6 years; 21 females; 63.6%, aged 9.3–16.2 years) and 12 had MoA (6 females; 50%, aged 10.4–13.7 years; 6 males; 50%, aged 4.8–11.8 years).
Visual manifestations were the most common aura symptoms in the MA patients (24/33), and they often occurred as the only symptom (13/24). Table 1 shows the aura symptoms and their various associations in our patients.
Aura symptoms

A detailed family history regarding idiopathic headache and thromboembolic events was obtained for all patients through a specific structured interview, which had previously been adopted by the Italian Group for the Study of Juvenile Headache (20). Neurological and clinical examinations were performed in addition to ophthalmological and cardiological evaluation in all patients. All subjects underwent either brain computed tomographic (CT) scan or nuclear magnetic resonance (NMR) imaging. None of the patients smoked, drank alcohol or took drugs, except for possible symptomatic and/or prophylactic therapy of migraine attacks. All patients followed a balanced diet.
The control group consisted of 66 volunteer, regular blood bank donors (28 males, 43%; and 38 females, 57%; mean age 31.4 ± 5.7 years, range 15–41). The healthy adults had no history of migraine or thromboembolic disease either as adults or in childhhod. Furthermore, in this group we ruled out metabolic alterations and clinical conditions possibly inducing acquired increased homocysteine levels (chronic heart disease, stroke, chronic renal failure, immunocompromised subjects, hormonal treatments and vegetarian diets). All adult controls were enrolled after filling in a specific questionnaire, ruling out a previous history of migraine and/or thromboembolic diseases either in adulthood or in childhood.
Serum virological and immunological evaluation, lactate, rheumatoid factor, antiphospholipid antibodies, anticardiolipin antibodies, lupus anticoagulant (LA) and coagulation screening (prothrombin time and activated partial thromboplastin time) were performed on all patients and controls. Subjects with pathological results were not included in this study.
Since normal homocysteine values differ between adults and children, in order to avoid loading a new control group of children with homocysteine, we used data obtained from a previous study group of 45 healthy children (26 males, 58%; and 19 females, 42%; mean age 8.6 ± 3.4 years) to compare values of vitamin B12, folate and fasting and PML homocysteine levels. In these children, who had previously been evaluated at the axiological out-patient clinic due to suspected growth development abnormalities, genetic and acquired diseases had already been ruled out by the specialist examination.
Informed consent was obtained from all patients and from controls. Most of the participants were born and lived in Liguria and Southern Piedmont (Italy), none of them was on anticoagulant or vitamin therapy at the time of the study.
Patients were evaluated during the intercritical period, which was defined as at least 10 days after the end of an attack.
Biochemical and genetic analyses
Blood for DNA studies was collected into 0.1 vol. 0.129
Plasma homocysteine was measured while subjects were fasting and 4 h after a standard methionine loading test (100 mg/kg).
Samples for fasting and PML homocysteine were collected in EDTA, then immediately cooled on ice and centrifuged as soon as possible at 1542 g for 10 min at room temperature. Serum was left to clot for 30 min at room temperature, then centrifuged at 1542 g for 10 min. Specimens were stored at 4°C for analysis on the same day. The long-term storage temperature was −20°C.
Homocysteine assays were performed as previously described (27).
D,L-Homocysteine, L-cysteine, and Tri-n-butylphosphine were obtained from Sigma (St Louis, MO, USA). Cysteinyl-glycine was purchased from Serva Fine Chemicals (Westbury, NY, USA). Glutathione was bought from Boehringer (Mannheim, Germany), while SBD-F was supplied by Wako (Düsseldorf, Germany). All other reagents were obtained from Merck (Darmstadt, Germany).
Statistical analysis
Data are described as mean and standard deviation (SD) for continuous variables, and absolute and relative frequencies for categorical variables. PML homocysteine was transformed by natural logarithm in order to normalize the data.
Parameters of the two groups were compared using the one-way analysis of variance (
A P-value <0.05 was considered statistically significant, and all P-values were based upon two-tailed tests.
Statistical analysis was performed using SPSS for Windows (SPSS Inc., Chicago, IL, USA).
Results
The study population consisted of 45 children (18 males, 40%; 27 females, 60%) aged 5–17 years (mean 11.6 ± 2.7 years), 34 with MA and 11 with MoA.
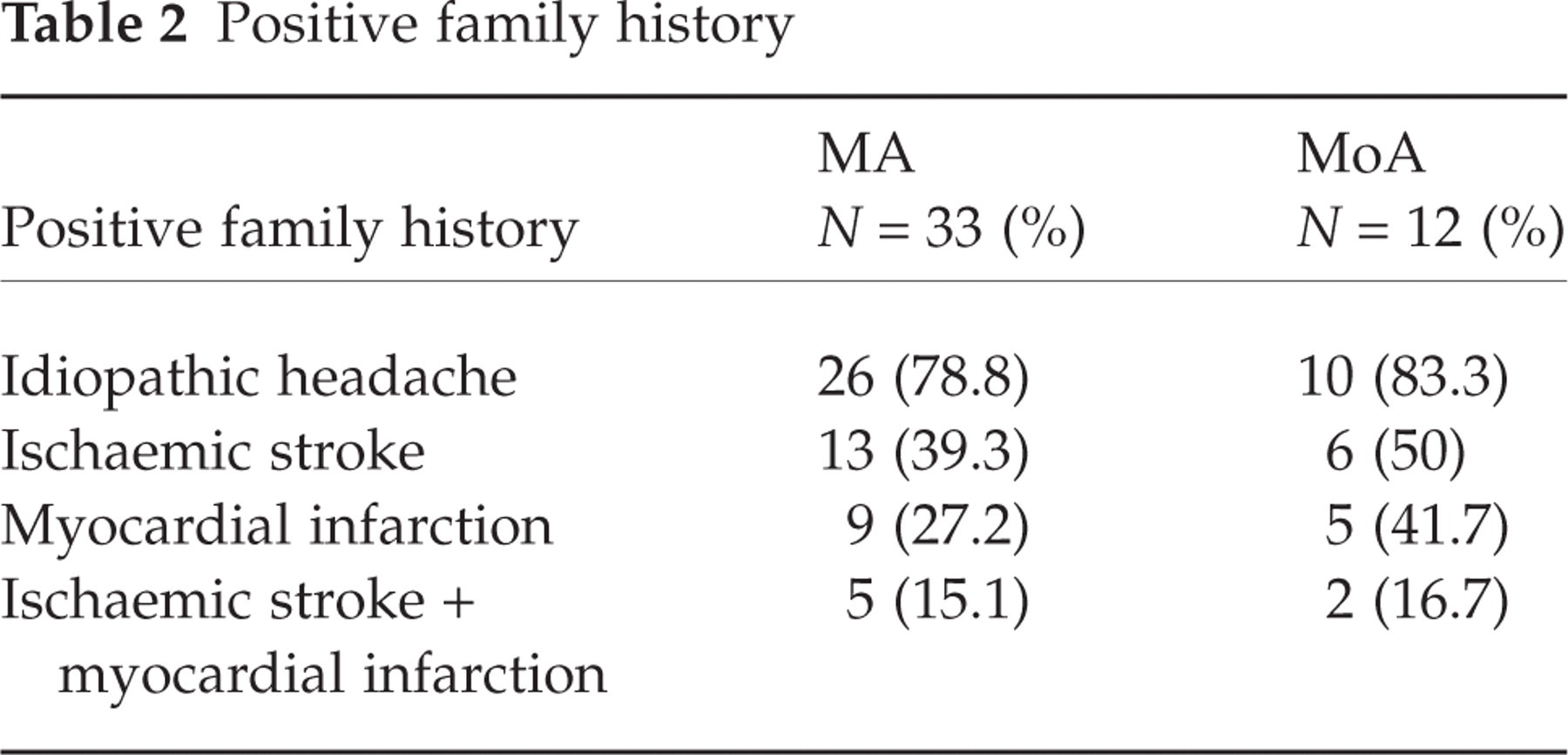
A family history of idiopathic headache was found in 26 MA patients and in 10 MoA patients. Table 2 shows the percentages of idiopathic headache, thromboembolic events and myocardial infarction recurrence in the patients’ families. As far as juvenile thromboembolic events are concerned (before 45 years of age), the only report involves the grandmother of an MoA patient who had an ischaemic stroke when she was 35 years old.
Positive family history
Cerebral CT and NMR were normal in all patients.
Homocysteine values, as well as folate and vitamin B12 levels of all patients and controls, are reported in Table 3.
Biochemical characteristics of patients and paediatric controls (mean ± SD)

Patients with migraine had higher levels of loading homocysteine values compared with controls (19.5 ± 4.9 vs. 16.9 ± 1.9; P = 0.025) and significantly lower folate levels (5.8 ± 2.6 vs. 7.5 ± 2.1; P = 0.002).
Genotype distribution and allele frequencies of MTHFR C677T, MTHFR A1298C, FVL and PRT mutations in patients and controls are shown in Table 4.
Genotype distribution and allele frequency of the MTHFR C677T and MTHFR A1298C, factor V A1698T and PRT G20210A polymorphisms in patients and adult controls

The genotype frequency distribution for MTHFR C677T and MTHFR A1298C mutations and FVL and PRT mutations was as expected, according to Hardy–Weinberg equilibrium (χ2 = 0.198, χ2 = 1.93, χ2 = 0.016, χ2 = 0.016).
The frequency of 677TT homozygotes in migraine patients (26.7%) was higher than in controls (13.6%). Similarly, the percentage of 1298CC homozygotes in migraine patients (14.3%) was higher than in controls (3.2%).
The FVL heterozygous mutation was found in only 3/44 migraine patients (6.7%) and in two controls (3%), which resulted in no significant difference. Moreover, the heterozygous form for the PRT mutation was found in 2/43 (4.4%) patients and 2/66 (3%) control subjects.
Among controls, the frequency of the T allele of the MTHFR C677T mutation was 38.6%, while the frequency of the C allele of the MTHFR A1298C mutation was 25%. As shown in Table 5a, we found no significantly increased risk for the heterozygous or homozygous 677 genotypes in migraine patients or in controls.

Unbold, not significant.
Regarding the A1298C mutation, we found no increased risk for the heterozygous genotype, but an increased risk, though not significant, for the homozygous genotypes (OR = 4.5, 95% CI 0.83, 24.36; P = 0.08). However, an increased risk was found when we compared the homozygous genotype of the patients with both wild-type and heterozygous genotypes of controls (OR = 4.92, 95% CI 0.82, 37; P = 0.05). These results were also confirmed when patients with MA were examined separately (Table 5b).
Finally, we investigated the combined genotype distribution of MTHFR C677T and MTHFR A1298C mutations (Table 6). Two or more MTHFR mutated alleles were present in 41% of controls and in 67% of patients.
Combinations of MTHFR C677T/A1298C genotypes in all migraine patients and adult controls

Discussion
The aim of this study was to analyse the possible relationships between migraine and prothrombotic risk factors and metabolic alterations in a young population in whom other environmental risk factors had been ruled out.
Our study showed a weak association between the MTHFR C677T polymorphism and migraine. In fact, we found an almost twofold increased risk for 677TT genotype, although this value was not statistically significant. Other authors have found an increased risk (OR = 6.5 and 3.05) in patients with aura carrying the 677TT genotype (13, 17). Furthermore, Oterino found an increased risk only in patients with MA compared with those without aura (OR = 2.34), but not vs. controls (15).
Concerning the association between MTHFR A1298C polymorphism and migraine, we found an OR = 4.5, 95% CI 0.83, 24.36, but this result was of borderline statistical significance, perhaps due to the small sample size. However, a significantly increased risk was found when the homozygous genotype of the patients was compared with both wild-type and heterozygous genotypes of the controls (OR = 4.92, 95% CI 0.82, 37; P = 0.05).
Kara found a significant association between the MTHFR 1298CC genotype and migraine in a larger cohort of patients (n = 102, OR = 8.9, 95% CI 1.9, 40.9; P = 0.001 (17).
In the present study, we observed that only 41% of controls showed a concomitant presence of MTHFR C677T mutant alleles and hetero- or homozygosity for MTHFR A1298C compared with 67% of patients with MA and 44% of patients with MoA. Moreover, we observed that among controls and migraine patients, none had double homozygosity for MTHFR 677TT/1298CC combined genotype, since both polymorphisms are in cis.
Among controls, the frequency of the T allele of the MTHFR C677T mutation was 38.6%, while the frequency of the C allele of the MTHFR A1298C mutation was 25%, comparable to that found in a previous study (28) and in accordance with other studies (17, 29, 30).
In our study, we found that prothrombotic genetic risk factors (FVL, PRT) did not differ significantly between patients with juvenile migraine and controls, in agreement with other authors (31, 32).
Increased homocysteine levels are associated with various pathological conditions, including stroke and ischaemic cardiovascular disorders. We observed that fasting homocysteine levels were similar in patients and controls while, on the other hand, PML homocysteine levels were significantly higher in patients. Moreover, the latter had significantly lower folic acid levels, despite their balanced diets. Thus, our data suggest that folate metabolism may play a role in the pathophysiology of migraine.
We found a trend toward an increased risk for migraine in subjects carrying a homozygous mutant genotype for MTHFR C677T or MTHFR A1298C polymorphisms.
Several authors agree that the presence of C677T and A1298C polymorphisms in the homozygous or in the double heterozygous state in the MTHFR gene can lead to a direct alteration of homocysteine and folate levels.
Owing to the small sample size we could not perform an analysis of gene–gene interaction. However, we attempted to study the combined genotype distribution of MTHFR C677T and MTHFR A1298C mutations. Interestingly, we observed that only 41% of controls had at least two or more MTHFR mutated alleles compared with 67% of patients.
In our experience, genetic prothrombotic conditions do not seem to be related to migraine in the young, whereas the biochemical differences between migrainous patients and controls are an appealing topic for further investigation in a larger cohort of patients.
