Abstract
Preference is a composite, patient-oriented endpoint incorporating efficacy, tolerability, formulation, and convenience of medications. The objective of this study was to compare patient preference for rizatriptan 10-mg wafer vs. eletriptan 40-mg tablet for acute treatment of migraine. In this multicentre, open-label, two-period, crossover study, out-patients were randomly assigned to treat the first of two moderate to severe migraines with rizatriptan or eletriptan and the second with the alternate therapy. Patients completed diary assessments at baseline and up to 24 h after taking study medication. At the last visit, patients completed a psychometrically validated preference questionnaire. A total of 372 patients (mean age 38 years, 85% female) treated two migraine attacks, and 342 patients (92%) expressed a preference for treatment. Significantly more (P ≤ 0.001) patients preferred rizatriptan 10-mg wafer [61.1%; 95% confidence interval (CI) 55.7, 66.3] to eletriptan 40-mg tablet (38.9%; 95% CI 33.7, 44.3). The most common reason given for preference of either treatment was speed of headache relief. At 2 h, 80% and 69% of patients reported that rizatriptan and eletriptan, respectively, was convenient or very convenient to take (mean convenience score 1.99 vs. 2.31, respectively; P ≤ 0.001). Both triptans were well tolerated. In this head-to-head study designed to evaluate global patient preference, significantly more patients preferred the rizatriptan 10-mg wafer to the eletriptan 40-mg tablet for acute treatment of migraine. The single most important reason for preference was speed of relief, consistent with results from previous preference studies.
Options for acute treatment of migraine have improved during the last decade with development of selective agonists for serotonin 5-HT1B/1D receptors, a class of drug known as the triptans (1). These agents, including sumatriptan, naratriptan, zolmitriptan, rizatriptan, almotriptan, eletriptan and frovatriptan, have established efficacy for treating migraine based on results of controlled clinical trials (1–3). However, each varies in details of pharmacology, efficacy, and tolerability, characteristics that can influence patient preference for therapy. Preference is a clinically relevant endpoint that is understandable and meaningful to patients.
Rizatriptan is a potent, selective 5-HT1B/1D-receptor agonist with rapid onset of action after oral administration; time to peak plasma levels (T max) during migraine attacks is 1 h (2, 4–6). The recommended dose of rizatriptan for acute treatment of migraine is 10 mg; for patients also taking propranolol, the dose is 5 mg. A rapidly dissolving formulation of rizatriptan, known as a wafer, was developed to improve patient convenience by eliminating the need for fluid intake with medication. The wafer disintegrates on the tongue within seconds of administration and is swallowed with saliva and then absorbed in the gastrointestinal tract. In a double-blind, placebo-controlled clinical trial (7), significantly more (P ≤ 0.008) patients experienced pain relief (22% vs. 11% for placebo) and became pain-free (4% vs. 0%) as early as 30 min after administration of rizatriptan 10-mg wafer compared with placebo. At 2 h, 42% and 10% of patients in rizatriptan and placebo groups, respectively, were pain free. While individual patients tend to prefer one rizatriptan formulation over the other, there is no difference in patient preference rates for rizatriptan wafer vs. tablet (8).
Eletriptan is also rapidly absorbed after oral administration and has a T max of 1.5 h that is delayed to 2 h during a migraine attack (2). The recommended single dose for acute treatment of migraine is eletriptan 40 mg. In a recent double-blind, controlled clinical trial, significantly more patients reported relief of pain at 30 min after receiving eletriptan 40 mg than after placebo (8% vs. 2%) (9). Rates of freedom from pain at 2 h were 32% and 3% in eletriptan and placebo groups, respectively.
The objective of this multicentre, randomized, open-label, two-period crossover study was to compare the proportion of patients who preferred the rizatriptan 10-mg wafer with the proportion of patients who preferred the eletriptan 40-mg tablet for treating moderate to severe migraine. Preference is a patient-oriented endpoint that incorporates many aspects of treatment experience, including efficacy, tolerability, formulation and convenience of medication use. The primary hypothesis was that patients would report greater preference for rizatriptan than for eletriptan. This was based on the superiority of preference for rizatriptan compared with sumatriptan in two studies (10, 11), using a validated global patient preference questionnaire (12) administered at the final post-treatment visit. The secondary hypotheses were that both rizatriptan and eletriptan would be effective, safe, and well tolerated.
Patients and methods
Patients
Patients were adults aged 18–65 years with a history of at least 6 months of migraine, with or without aura, as defined by International Headache Society criteria (13). Eligible patients were judged to be in good health other than migraine. Migraine headache pain was scored on a 4-point scale (0, no headache; 1, mild pain; 2, moderate pain; 3, severe pain). Patients were excluded from study participation if they had less than one or more than eight attacks of migraine per month, if they had a preponderance of mild attacks (grade 1), if they had basilar or hemiplegic migraines, or if they had difficulty distinguishing migraine from tension or other interval headaches.
Other exclusion criteria were as follows: history or clinical evidence of cardiovascular disease; history of clinically significant electrocardiographic abnormality; uncontrolled hypertension; or clinical or laboratory evidence of renal, hepatic, or other systemic disease. For patients with risk factors for occult coronary artery disease, such as hypercholesterolaemia or diabetes, additional cardiovascular assessments were recommended before enrolment. Pregnant or lactating women were excluded from study participation.
Patients who had previously taken either rizatriptan or eletriptan were not eligible. Medications prohibited from 2 weeks before until 2 weeks after the study included erythromycin, ketoconazole, itraconazole, clarithromycin, josamycin, indinavir, ritonavir, nelfinavir (because of a potential interaction with eletriptan owing to CYP3A4 inhibition), monoamine oxidase inhibitors (because of a possible elevation in rizatriptan levels), and methysergide. Treatment with other triptans, ergot derivatives, or opiates was not permitted from 24 h before study treatment until 24 h after the last dose of study medication in a treatment period. Medications used for migraine prevention were permitted with the exception of non-steroidal anti-inflammatory drugs (NSAIDs) and propranolol, which was to be discontinued from at least 3 days before the first study treatment until 14 days after the last study treatment.
All patients signed a written consent form before participating in the study, which listed the potential benefits and side-effects of both medications. The study protocol was approved by the appropriate institutional review board or ethical review committee. Study medication was provided to patients without charge.
Study design
This randomized, two-period, open-label crossover study to compare preference for rizatriptan or eletriptan for treating a single migraine attack was conducted from December 2001 until September 2002 at 46 centres in Germany, Norway, Poland, Israel, Spain, Italy and Colombia. There were three scheduled clinic visits, the first at screening and the second and third within 5–7 days after each of the two treatment periods. Patients treated a single migraine attack and up to one recurrence in each of the two treatment periods, which were defined as the 24-h period after the initial dose of study medication. Recurrence was defined as the return of a grade 2–3 headache within 24 h after the initial dose in a patient who had responded by 2 h after taking study medication. Treatment periods were separated by at least 5 days and no more than 2 months.
Eligible patients were randomly assigned in a 1 : 1 ratio by a computer-generated random allocation schedule to one of two treatment sequences: (i) rizatriptan 10-mg wafer for treating the first migraine attack, then eletriptan 40-mg tablet for the second migraine attack (rizatriptan–eletriptan sequence); or (ii) eletriptan 40-mg tablet, then rizatriptan 10-mg wafer for first and second migraine attacks, respectively (eletriptan–rizatriptan sequence). At the prestudy screening visit and at the first post-treatment visit, patients received two doses of the medication assigned for first and second treatment periods, respectively. Treatment of the second migraine was not permitted until patients had completed the first post-treatment visit. Patients were also provided with a diary for each treatment period and with timers to facilitate entry of timed data into the diary.
During each treatment period, patients were instructed to take the randomly assigned study medication to treat moderate to severe (grade 2–3) migraine pain. Patients whose migraine severity did not improve to grade 0–1 within 2 h (non-responders) were permitted to take an additional analgesic, such as an NSAID or opiate, and an antiemetic. Those whose migraine severity improved to grade 0–1 within 2 h (responders) but then recurred within 24 h after treatment were to take the second dose of study medication. Any further recurrences during the 24-h treatment period could be treated with an additional analgesic and antiemetic.
Outcome measures
Preference questionnaire
At the last post-treatment visit after the second migraine attack (visit 3), patients completed a validated global preference questionnaire (10), answering the following question: If you had to treat your migraine headache with one of these two headache medications, would you prefer the wafer/tablet medication (form of first study drug) or the tablet/wafer medication (form of second study drug)? There were three possible answers: (i) I prefer taking the wafer medication, (ii) I prefer taking the tablet medication, and (iii) I have no preference. If patients expressed a preference, they were asked to select the one most important reason for their preference; if they had other reasons for preference, they were asked to write them in at the bottom of the questionnaire (Table 1).
Questionnaire regarding reasons for preference

The first part of each question was asked of patients who preferred the rizatriptan wafer; the second part of each question was asked of patients who preferred the eletriptan tablet.
Patient diary
Other outcome measures were derived from information recorded in patient diaries. Immediately before taking study medication (0 h, baseline) and again at 15, 30, 60, 90 and 120 min after taking study medication, patients rated headache severity and the ability to perform daily activities, and they recorded the presence or absence of associated symptoms (nausea, vomiting, photophobia and phonophobia). Headache severity was rated on the 4-point scale described above. Patients rated their functional disability also on a 4-point scale (0, normal; 1, mildly impaired; 2, severely impaired; and 3, unable to do activities, require bed rest).
At 2 h after the first dose of study medication in each treatment period, patients completed the question of satisfaction with medication using a 7-point scale (1, completely satisfied, could not be better; 2, very satisfied; 3, somewhat satisfied; 4, neither satisfied nor dissatisfied; 5, somewhat dissatisfied; 6, very dissatisfied; and 7, completely dissatisfied, could not be worse). At this time, they also rated the convenience of study medication (7-point scale: 1, very convenient; 2, convenient; 3, somewhat convenient; 4, neither convenient nor inconvenient; 5, somewhat inconvenient; 6, inconvenient; and 7, very inconvenient).
From 2 to 24 h after intake of study medication, patients recorded the use of additional analgesics or antiemetics (name and dosage) as well as the time and maximum severity of headache recurrence.
The following parameters were derived from data in patient diaries:
Headache relief: the proportion of patients reporting reduction of headache severity from grade 2–3 to grade 0–1 at each time point.
Time to headache relief within 2 h: the first time that each patient reported headache relief within the first 2 h after medication administration.
Freedom from pain: the proportion of patients reporting complete abolition of headache pain (grade 0) at each time point.
Time to freedom from pain within 2 h: the first time that each patient reported the complete abolition of headache pain in the first 2 h.
Freedom from pain and associated symptoms at 2 h: the absence of headache, nausea, vomiting, photophobia, phonophobia, and functional disability at 2 h after intake of study medication.
Sustained pain freedom at 24 h: freedom from pain at 2 h, no recurrence of headache, as well as no use of additional medication from 2 to 24 h after the initial treatment.
Sustained headache relief at 24 h: relief from headache at 2 h, no recurrence of headache, as well as no use of additional medication from 2 to 24 h after the initial treatment.
At each of the two post-treatment visits, the investigator reviewed the diary card with the patient and questioned the patient about the presence of aura at the time of the migraine attack. Female patients were asked about onset date of their last menstrual period to identify headaches that could be categorized as ‘menstrually associated migraine’, defined as a migraine beginning between 3 days before to 3 days after the onset of menstrual flow.
Safety evaluations
At screening, medical and migraine histories were recorded, and a complete laboratory examination was performed, including serum biochemical analysis, complete blood count, urinalysis, and, for women of childbearing potential, a pregnancy test (serum β-human chorionic gonadotropin levels). Physical examinations were performed and vital signs recorded at each visit.
Sample size calculations
Assuming a total of 320 patients (160 per sequence), this two-period crossover trial had approximately 95% power to detect a 20% point difference in preference (60% vs. 40%) for the wafer compared with the tablet. Approximately 450 patients (225 per sequence) were randomized to account for dropouts and respondents with no preference.
Statistical analysis
The primary efficacy endpoint, patient preference, was analysed for all patients who treated both attacks and who expressed a preference for one medication over the other. The Mainland–Gart procedure (14) was used to compare the number of patients preferring the rizatriptan wafer with the number of patients preferring the eletriptan tablet. The association in the table constructed following the Mainland–Gart procedure was assessed using the Fisher exact test. The 95% confidence intervals for the proportions of patients preferring rizatriptan wafer and for those preferring eletriptan tablet were obtained using the binomial distribution. The number of patients expressing no preference was summarized but not included in the main analysis. However, a prespecified exploratory analysis including these patients was also performed with the Prescott test, which does not discard any information from patients with no preference.
Secondary efficacy analyses included all patients who took at least one dose of study medication and recorded at least one efficacy measurement after the treatment dose. Missing data from a treatment period were estimated by carrying forward the preceding data in that treatment period. Baseline data were not used for imputation and were not carried forward, and data from the first treatment period were not carried forward to the second.
The percentages of patients at each time point with headache relief, freedom from pain, associated symptoms, and functional disability and those who took additional medication were analysed with logistic regression using the generalized estimating equation (GEE) method with exchangeable correlation structure. The main model included factors for treatment, period, baseline headache severity and geographical region. This model was used also for the analyses of the proportion of patients with freedom from pain and associated symptoms at 2 h and those reporting sustained pain freedom and sustained headache relief at 24 h. The ratings of functional disability were also dichotomized to ‘normal’ (grade 0) or ‘abnormal’ (grades 1, 2 or 3) and were analysed similarly to the analysis of headache relief.
For the analysis of time to headache relief, patients were considered censored either at 2 h if their headache severity was grade 2–3 at 2 h or at an earlier time if headache relief was not yet achieved and was followed by missing pain severity up to 2 h. A binary regression model adapted to the analysis of interval censored data (15) was used for the comparison of time to headache relief. The intracorrelations were allowed in the model with the GEE method and exchangeable correlation structure. The main model included factors for treatment, baseline severity, time interval, geographical region and period. Similar analyses were performed for time to freedom from pain within 2 h.
The seven scores for satisfaction with and convenience of medication at 2 h were considered continuous and analysed using mixed models. The main model included factors for treatment, baseline headache severity, period and geographical region as fixed effects and patient as random effect.
To summarize the efficacy of rizatriptan and eletriptan for menstrually associated migraine, the proportion of female patients with headache relief from menstrually associated migraine at 2 h was tabulated by treatment group. In order to evaluate the consistency of treatment effects across attacks associated with menstruation vs. those not associated with menstruation, the interaction between treatment and menstrual migraine was tested. This interaction was also evaluated for the proportion of female patients with freedom from pain at 2 h. Logistic regression was performed using data from the first treatment period only.
A per-protocol analysis, excluding patients with important protocol violations based on a set of prespecified criteria, was performed for the primary (preference) and key secondary endpoints (headache relief and pain free). The results of the per-protocol approach were consistent with those of the analysis including all patients treated.
The sequence effect was examined for all statistical models. If the sequence effect was significant (at a significance level of 0.10), then the nature of this effect was investigated to see whether the treatment effect was consistent across treatment periods and sequences. All probability values were two-sided, and statistical significance of treatment comparisons was declared at a P-value of ≤0.05.
All patients who took at least one dose of study medication were included in the safety analyses. The proportions of patients with adverse experiences among those who treated both attacks were summarized and compared between treatments using the McNemar test. Secondary analyses were based on all patients receiving at least one dose of the study medication, using logistic regression with the GEE approach and unstructured correlation structure. The model included factors for treatment and period.
Results
Patients
A total of 509 patients were screened for the study, and 506 patients were randomly assigned to treatment, of whom 439 (87%) received treatment and were included in the safety analyses. Forty patients received a reversed sequence of the two treatments in error; however, sensitivity analyses based on the randomized treatment sequence confirmed the results that were based on the actual treatment sequence. The results presented here are thus for the actual sequence: 260 patients took the rizatriptan–eletriptan sequence and 246 took the eletriptan–rizatriptan sequence. Patient disposition according to actual sequence taken is summarized in Fig. 1. Most patients who did not receive treatment had no migraine attack within 2 months, and most patients who discontinued the study after visit 2 did so because they did not have a second migraine attack within the allotted time interval.
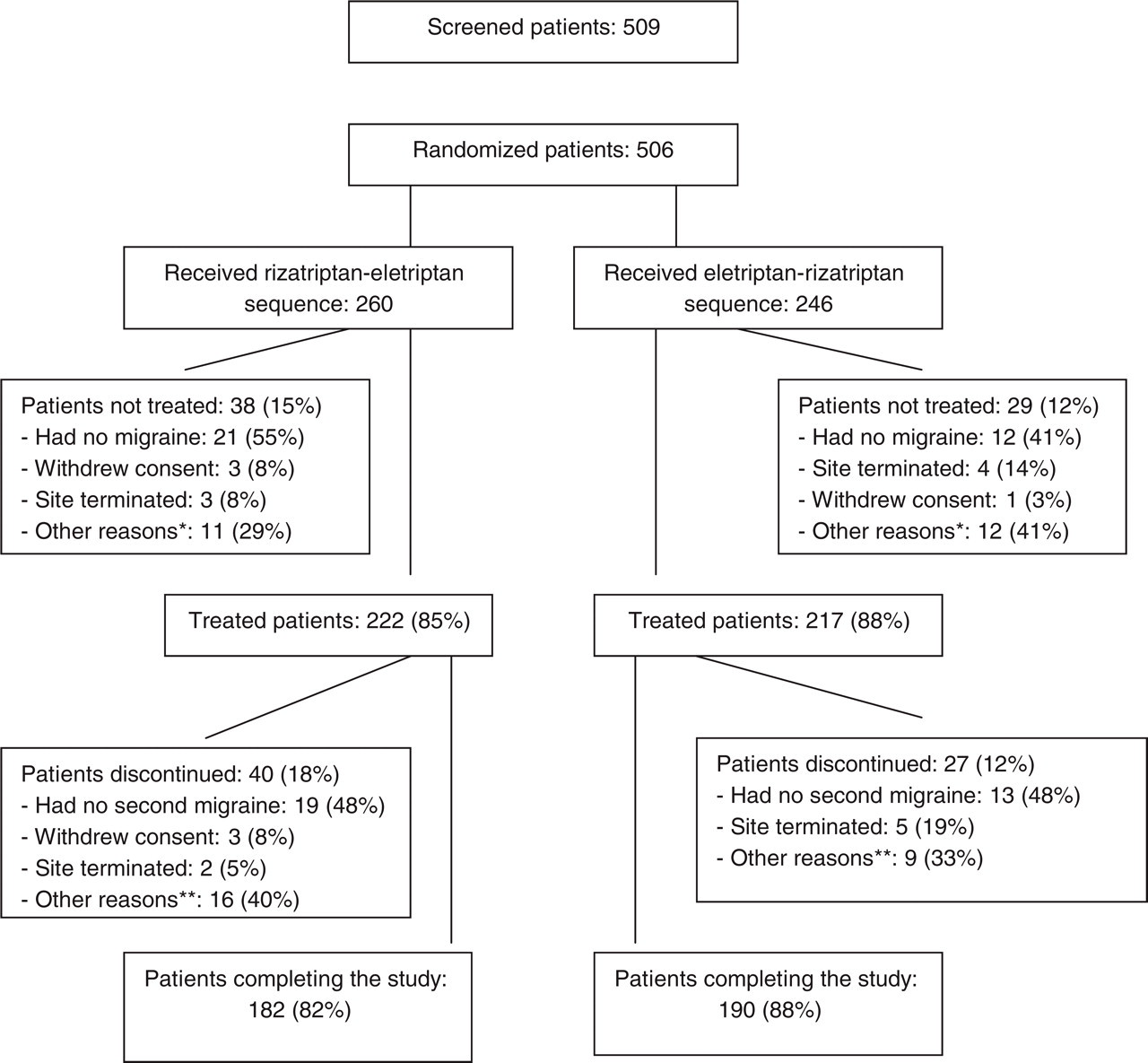
Profile of patient enrolment. ∗Other reasons for patients not receiving treatment included patient non-compliance, protocol deviation, patient moving away and loss to follow-up. ∗∗Other reasons for patients discontinuing from the study included adverse events, lack of efficacy, misdiagnosis, patient moving away, patient non-compliance and protocol deviation.
A total of 372 patients (85% of the patients treated) completed both treatment periods and were asked to complete the preference questionnaire. The mean age of treated patients was 38 years and most were female (85%) and white (88%). Baseline patient demographics and migraine features were similar between the two treatment sequences (Table 2).
Baseline demographic and migraine characteristics of patients who received treatment

NSAID, Non-steroidal anti-inflammatory drug.
Includes black, Asian, and multiracial patients.
Last migraine attack before inclusion in the study.
Characteristic of the first migraine attack of the study.
Percentage of female patients (no information was available for 23% and 20% of patients in the rizatriptan–eletriptan and eletriptan–rizatriptan sequences, respectively).
Primary endpoint—treatment preference
Of the 372 patients who completed both treatment periods, 342 (92%) expressed a preference for one of the study drugs and were included in the preference analysis; 29 patients (8%) did not express a preference; and one did not complete the preference questionnaire. Significantly more (P ≤ 0.001) patients preferred treatment with rizatriptan 10-mg wafer [61.1%; 95% confidence interval (CI) 55.7, 66.3] to treatment with eletriptan 40-mg tablet (38.9%; 95% CI 33.7, 44.3). These findings were consistent for each treatment sequence (Fig. 2), and there was no sequence effect (P = 0.739). Moreover, the results of the prespecified exploratory analysis including all 371 patients who completed the questionnaire were consistent with the results from the main analysis: rizatriptan 10-mg wafer was still preferred over eletriptan 40-mg tablet (56.3% vs. 35.8%, respectively, P ≤ 0.001).

Percentages of patients in each treatment sequence who reported preference for one medication over the other. Sequence 1 was rizatriptan for the first migraine attack and eletriptan for the second; sequence 2 was eletriptan for the first and rizatriptan for the second. The comparison favoured rizatriptan (P ≤ 0.001). □, Prefer rizatriptan; ▪, prefer eletriptan.
Faster relief of headache pain was the most common reason for preference, cited both by patients who preferred rizatriptan (54%) and by those who preferred eletriptan (51%). The second most common reason for preference was ‘returned to my normal activities quicker’, which was cited by 12% and 10% of patients, respectively. Reasons cited for treatment preference are summarized in Table 3.
Secondary endpoints
Satisfaction with medication and convenience of medication at 2 h
Overall satisfaction with medication at 2 h was significantly greater after rizatriptan treatment than after eletriptan treatment (mean score 3.12 vs. 3.33; P = 0.048 for the comparison; a lower score indicates greater satisfaction). At 2 h, 45% of rizatriptan-treated patients and 40% of eletriptan-treated patients reported that they were completely or very satisfied with study medication (score of 1 or 2). There was a strong association between preference and satisfaction. Patients who were completely or very satisfied with rizatriptan or eletriptan tended to prefer that triptan (95.6% vs. 88.7%, respectively).
Mean scores for convenience of medication at 2 h were also significantly lower (indicating better convenience) for rizatriptan (1.99) than for eletriptan (2.31; P ≤ 0.001). At 2 h, 80% and 69% of patients reported that rizatriptan and eletriptan, respectively, was very convenient or convenient to take.
There was no sequence effect for either satisfaction or convenience scores or for any other secondary endpoint; however, there was a period effect for the convenience score (P = 0.023).
Headache relief and freedom from pain up to 2 h
The proportions of patients reporting headache relief and freedom from pain at each time point during the first 2 h were similar after treatment with rizatriptan or eletriptan. Relief of headache at 2 h was reported by 77% of patients receiving either treatment. At 2 h, 52% of rizatriptan-treated patients and 50% of eletriptan-treated patients were pain free. The onset of relief also influenced preference. In the subgroup of patients citing an early response as the reason for preference, 97.6% of patients who experienced earlier relief with rizatriptan preferred it, compared with 79.5% for eletriptan.
Time to headache relief and time to freedom from pain up to 2 h
The times reported by patients at which they experienced headache relief and freedom from pain were similar after treatment with rizatriptan and eletriptan, although a borderline significant treatment effect of rizatriptan compared with eletriptan was detected from 30 min onward with respect to time to headache relief (P = 0.08). The cumulative percentages of patients reporting relief of headache pain at from 31 to 60 min were 47% and 43% after rizatriptan and eletriptan, respectively; at from 61 to 90 min, 69% and 66%, respectively; and at from 91 to 120 min, 81% and 79%, respectively. The cumulative percentages of patients reporting that they were pain free at from 31 to 60 min were 17% and 14% after rizatriptan and eletriptan, respectively; at from 61 to 90 min, 30% and 28%, respectively; and at from 91 to 120 min, 54% and 51%, respectively.
Sustained pain freedom
At 24 h, 39% and 37% of patients receiving rizatriptan and eletriptan, respectively, reported sustained pain freedom. These results were not statistically different.
Associated symptoms
At baseline and at 2 h after treatment with rizatriptan or eletriptan, similar percentages of patients were experiencing symptoms associated with migraine, including nausea, photophobia, phonophobia and vomiting (Table 4). These percentages were not statistically different at other time points with the exception of the following time points, when significantly fewer patients experienced symptoms after rizatriptan than after eletriptan intake: nausea at 60 min (30% vs. 37%; P = 0.005), photophobia at 30 min (63% vs. 70%; P = 0.014); phonophobia at 15 min (64% vs. 69%; P = 0.031); and phonophobia at 30 min (54% vs. 60%; P = 0.008).
Most important reasons for treatment preference

Secondary efficacy endpoints

Functional disability
Most patients (97%) reported functional disability at baseline before treatment with either rizatriptan or eletriptan. At 2 h, percentages of patients reporting resolution of functional disability were similar after either treatment (Table 4).
Freedom from pain and associated symptoms at 2 h
Similar percentages of patients were free from pain and all symptoms associated with migraine at 2 h after treatment with rizatriptan and eletriptan, namely, 43% and 41%, respectively.
Need for additional medication from 2 to 24 h or second dose of study medication
The percentages of patients who took additional analgesia or antiemetics from 2 to 24 h after treatment were 23% after rizatriptan compared with 19% after eletriptan (P = 0.194). Those who took a second dose of study medication to treat a headache recurrence from 2 to 24 h after treatment were 19% and 16%, respectively.
Efficacy in menstrually associated migraine
Approximately one-third of migraine attacks among female patients during the study were associated with menstruation. The proportions of patients with headache relief or freedom from pain at 2 h were not significantly different whether or not migraine attacks were associated with menstruation (P = 0.585 for interaction of treatment with menstrually associated migraine). The percentages of patients who reported headache relief at 2 h after rizatriptan were 81% and 75% with and without association of menstruation with migraine, respectively; the percentages after eletriptan were 71% and 78%, respectively. Those who were pain free at 2 h after rizatriptan were 52% and 50%, respectively, and after eletriptan 44% and 49%, respectively.
Safety results
On the 372 patients who treated both attacks, 14% reported an adverse event only after treatment with rizatriptan, 18% reported an adverse event only after treatment with eletriptan, and 8% reported at least one adverse event after both treatments; there was no significant difference between treatment groups (P = 0.197) with respect to the proportions of patients with any clinical adverse event. Overall, adverse events were reported by 22% of patients after rizatriptan therapy and 27% of patients after eletriptan therapy. The most common adverse events after rizatriptan and eletriptan, respectively, were somnolence (3.9% and 2.0%), asthenia/fatigue (2.7% and 5.3%), nausea (2.4% and 4.3%) and dizziness (1.9% and 3.8%). Chest pain was reported for 1.2% of the patients after rizatriptan 10 mg and for 1.8% of the patients after treatment with eletriptan 40 mg. The only serious adverse event during the study (gastric disorder leading to hospitalization) started 10 days after rizatriptan intake and was considered definitely not drug-related by the physician.
Discussion
Significantly more patients in this open-label, crossover study preferred the rizatriptan 10-mg wafer to the eletriptan 40-mg tablet for the acute treatment of migraine. Preference is a patient-focused endpoint that reflects the totality of patient experience with a medication. This endpoint can incorporate efficacy, tolerability and convenience dimensions, including formulation. Faster relief of headache pain was reported as the most important reason for treatment preference both by patients who preferred rizatriptan (54%) and by those who preferred eletriptan (51%). Another 12% and 10%, respectively, reported the most important reason for treatment preference to be a faster return to normal activities.
Results of prior preference studies comparing rizatriptan and sumatriptan have indicated that preference for migraine medication is usually driven by speed of relief (10, 11). In these two open-label crossover studies, designed similarly to the present study, significantly greater proportions of patients preferred the 10-mg rizatriptan wafer (10, 11) over the 50-mg sumatriptan tablet for acute treatment of migraine. The most important reason for preference, cited by approximately half of patients expressing a preference in these two studies, was faster relief of headache pain.
These findings are consistent with what patients have reported to be most important in an acute migraine treatment. In two telephone surveys of self-reported migraineurs (16, 17), patients listed rapid and effective relief of headache pain as among the most important features of a migraine medication. Moreover, rapid onset of complete pain relief is an important determinant of patient satisfaction with treatment (18, 19).
There are no prior published studies directly comparing rizatriptan with eletriptan. However, Ferrari and coworkers (2, 3) have published a meta-analysis examining raw data from 24 089 patients enrolled in 53 clinical trials of oral triptans in migraine; they used the data for sumatriptan 100 mg as the basis for comparison of data for other triptans. In their published meta-analysis, eletriptan 40 mg showed efficacy, consistency, and tolerability results similar to those of sumatriptan 100 mg. Rizatriptan 10 mg showed similar tolerability but better efficacy and better consistency than sumatriptan 100 mg. Moreover, the intrapatient consistency rates for pain response and pain free over three attacks were the highest for rizatriptan 10 mg of all the triptans. The authors comment that differences among the triptans are in general relatively small but are clinically relevant for individual patients (2). The current study evaluated only a single attack, and it would be interesting to explore preference over multiple attacks in future research.
Convenience of taking the wafer formulation of rizatriptan, without the need for liquid, may also have been a factor contributing to patient preference for rizatriptan in the present study, as the comparison of scores for convenience was highly significant in favour of rizatriptan. However, although the wafer formulation may have been a factor, there were no differences in the percentages of patients who listed the formulation or the ease of administration as the reason for preferring rizatriptan or eletriptan. This is consistent with results of a prior retrospective evaluation that found no difference in patient preference rates for rizatriptan wafer vs. tablet (8).
A limitation of the present study was that it was not a blinded comparison. The results for a sequence effect to identify potential bias in patient answers on the preference questionnaire were analysed, and there was no evidence for a sequence effect on any of the multiple endpoints examined.
Both rizatriptan and eletriptan were well tolerated in this study. The overall incidence of adverse events was similar for both treatments and there was no clinically significant difference in incidence of individual adverse events, the most common of which were somnolence, asthenia/fatigue, nausea and dizziness.
In conclusion, rizatriptan 10-mg wafer was preferred to eletriptan 40-mg tablet for the acute treatment of migraine. The most important reason for patient preference in this study was speed of relief, consistent with prior preference studies. Given the similar results for both treatments on traditional headache endpoints, the findings emphasize the utility of preference as a clinically relevant outcome measure. Preference is a patient-oriented endpoint that captures the sum total of treatment experience, allowing the results to be driven from a patient's perspective. Being aware of this perspective could assist physicians in their selection of acute migraine therapy.
Conflict of interest
M.J.A.L. has received grant/research support from, has been a consultant/scientific advisor for/or has received honoraria for oral presentations from Almirall Prodesfarma, AstraZeneca, Böhringer Ingelheim, Bristol-Myers Squibb, Elan Pharmaceuticals, GlaxoSmithKline, Janssen Cilag, Johnson & Johnson, MSD, Novartis, Pierre Fabre, and Sanofi-Synthelabo. C.A., N.A.R., R.M. and K.L. are full time employees of Merck & Co. Inc., USA.
Footnotes
Acknowledgements
Editorial assistance was provided by Elizabeth V. Hillyer DVM, ELS.
