Abstract
A meta-analysis of pooled individual patient data from four randomized, placebo-controlled, double-blind trials comparing several doses of almotriptan (n = 1908) with placebo (n = 386) was used to investigate the efficacy, speed of onset and tolerability of almotriptan in the acute treatment of migraine. As early as 30 min after dosing, almotriptan 12.5 mg was significantly more effective than placebo for pain relief (14.9% vs. 8.2%; P < 0.05) and pain free (2.5% vs. 0.7%; P < 0.05). At 2 h, pain-relief rates were 56.0%, 63.7% and 66.0% for almotriptan 6.25, 12.5 and 25 mg, respectively, compared with 35% for placebo; 2-h pain-free rates were 26.7%, 36.4% and 43.4% compared with 13.9% for placebo. All almotriptan dosages were significantly more effective than placebo in eliminating migraine-associated symptoms (P < 0.05) and in achieving sustained pain relief up to 24 h (P < 0.05). The incidence of adverse events after almotriptan 6.25 mg and 12.5 mg was not significantly different from that of placebo. This meta- analysis confirms the findings of individual clinical trials, while demonstrating for the first time, significant pain-free efficacy at 30 min compared with placebo.
Introduction
Migraine is a common, painful and often disabling condition with major social, financial, and personal ramifications (1). The main objectives of acute migraine therapy are to reduce and eliminate pain and associated symptoms, rapidly restore the patient to normal function without side-effects and prevent recurrence of the attack within 24 h (2). In recent years, the triptans, selective serotonin 5-HT1B/1D receptor agonists have become the drugs of choice for the acute treatment of moderate and severe migraine attacks (3, 4). Worldwide, seven triptans are currently marketed for the acute treatment of migraine: almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan and zolmitriptan.
Although triptans as a class are highly effective for the acute treatment of migraine (5, 6), a meta-analysis of the results of 53 double-blind, placebo-controlled single attack studies, which compared six oral triptans with sumatriptan 100 mg with regard to efficacy, tolerability and consistency of treatment response, found that some triptans differ from others (7). In addition, patients also report personal preferences for individual agents in this class (8). Patients indicate a strong preference for acute agents that provide quick, complete and lasting relief from the headache and other migraine symptoms (9–12). They describe quick as ≤1 h (11, 13). Moreover, in several comparative clinical trials, the principal reason given by patients for preferring one drug over another was speed of headache relief (13, 14). Therefore, the ‘ideal’ triptan can be defined as one that restores the patient's ability to function by swiftly and completely relieving the pain and associated symptoms with a minimum of side-effects and without recurrence of symptoms.
Almotriptan is a newer triptan shown to be consistently effective and well tolerated in controlled clinical trials (15–18). Almotriptan 12.5 mg has the highest bioavailability of all the oral triptans (5) and a rapid onset of action, with pain relief superior to placebo as early as 30 min after administration (18). The efficacy of almotriptan 12.5 mg is comparable to that of sumatriptan 50 and 100 mg (16, 17), but almotriptan is associated with better tolerability (16–18).
The consistency of almotriptan has been demonstrated in a clinical trial measuring patients’ responses across three consecutive migraine attacks (15). Considering only patients who had three attacks, 74.5% of those who treated with almotriptan 12.5 mg achieved 2-h pain relief in at least two of three attacks, 50% achieved 2-h pain relief in three of three attacks; the equivalent values in the placebo group were 35.7% and 16%, respectively (15).
Although controlled clinical trials have been useful for establishing the efficacy and tolerability profile of almotriptan, pooling patient data from multiple trials increases the amount of information in the analysis and provides additional statistical power that typically cannot be derived from a single clinical trial. The improved statistical precision confers the ability to explore almotriptan treatment outcomes in greater detail and over a larger population base. The aim of these analyses was to pool patient data from four randomized, double blind, placebo-controlled studies of almotriptan to assess the global efficacy and tolerability of almotriptan in the acute treatment of migraine.
Methods
Studies included in the analysis
Individual patient data derived from four prospective studies conducted by Almirall between 1995 and 1997 examining the use of almotriptan in adult patients with migraine were pooled for this meta-analysis (Table 1). Three of the studies have been published previously as primary manuscripts in peer-reviewed journals; one was published in abstract form but additional unpublished data were requested and received from Almirall (15, 17–19). In one of these trials, three attacks were treated (15); for consistency with the other trials pooled for this meta-analysis, only the first attack was included.
Description of patients and studies

All study drugs were encapsulated.
This trial treated three attacks; only the first attack is included to be consistent with the other trials pooled for this meta-analysis.
This trial also contained a sumatriptan 100-mg arm to which 194 patients were randomized; these patients are not included in the analysis presented here.
Study designs
All studies employed identical randomized, double-blind, placebo-controlled, parallel-group designs and assessed the efficacy and safety of single doses of almotriptan in patients with migraine. One also included a comparative treatment arm with sumatriptan 100 mg but the results from this arm are not included in the meta-analysis (17). All studies were conducted at multiple sites in Europe in accordance with the Declaration of Helsinki and Good Clinical Practice and approved by the relevant Ethics Committees. All patients provided written informed consent before entering the studies.
Patients
All trials had broadly similar eligibility criteria for patients. Studies included male and female patients between 18 and 65 years of age who had at least a 6-month history of migraine according to the criteria of the International Headache Society, and typically experienced one to six migraine attacks per month.
Patients were excluded if they had clinical evidence of cardiovascular disease including significant ECG abnormality, or if they had a history within 1 year or current evidence of drug or alcohol abuse. Other exclusion criteria included: migraine with prolonged aura, familial hemiplegic migraine, migrainous infarction or vertebrobasilar migraine; the occurrence of more than six migraine attacks per month; time between screening and migraine attack longer than 8 weeks; prophylactic medications for migraine taken during the 2 weeks prior to the study; pregnant women or nursing mothers without adequate contraceptive protection; and intolerance or known hypersensitivity to 5-HT1B/1D agonists.
Treatment and assessments
Eligible patients were randomized to receive placebo or one of several doses of almotriptan, which were assessed for their efficacy and tolerability. Patients took a single oral dose of active drug or placebo during a migraine attack of moderate or severe intensity. Patients were permitted to take rescue medication chosen by the investigator according to the patient's characteristics and preferences, if migraine pain persisted 2 h after taking study medication. However, ergot derivates or 5-HT1B/1D agonists were not allowed as rescue medication. A second dose of study medication was permitted only for the treatment of recurrence of a moderate or severe headache within 24 h in a patient who had experienced initial pain relief at 2 h.
At baseline, and at 0.5, 1, 1.5 and 2 h after treatment, patients recorded their headache severity on self-assessment cards using a standard 4-point headache pain scale (0 = no pain, 1 = mild pain, 2 = moderate pain, 3 = severe pain). At the same time points, patients also recorded the presence or absence of migraine-associated symptoms (nausea, vomiting, photophobia and phonophobia).
Efficacy measures
The following efficacy outcomes that were measured for this analysis include:
Percentage (proportion) of patients achieving pain relief: the percentage of patients with a reduction of headache pain severity from moderate or severe to mild or no pain (score of 2 or 3 reducing to 1 or 0) at 0.5, 1.0, 1.5 and 2.0 h postdose
Percentage (proportion) of patients achieving pain free: the percentage of patients with a reduction of headache pain severity from moderate or severe to none (score of 2 or 3 reducing to 0) at 0.5, 1.0, 1.5 and 2.0 h postdose
Percentage (proportion) of patients achieving sustained pain relief: the percentage of patients who had pain relief at 2 h with no return of moderate or severe headache pain within 24 h
Percentage (proportion) of patients experiencing headache recurrence: the percentage of patients with return of moderate or severe headache pain within 24 h following relief at 2 h
Percentage (proportion) of patients using rescue medication
Percentage (proportion) of patients achieving relief from migraine-associated symptoms: the percentage of patients with photophobia, phonophobia, nausea or vomiting at 2 h postdose in patients who had each symptom at baseline
Percentage (proportion) of patients achieving sustained pain free (SPF): the percentage of patients who were pain free at 2 h with no recurrence of moderate or severe headache pain and no use of rescue medication from 2 to 24 h postdose
Percentage (proportion) of patients achieving sustained pain free with no adverse events (SNAE): the percentage of patients who achieved SPF and did not report any adverse events (AEs)
Safety assessments
Safety and tolerability were assessed by physical examinations, standard laboratory tests, vital signs and ECGs at baseline and final visit, and self-recorded AEs. All postdose AEs were collected using diary cards and evaluated by the investigator. The physician also asked patients about AEs in an open-ended fashion during post-treatment visits.
Statistical analysis
The efficacy analysis was performed on an intent-to-treat (ITT) basis that included all patients who provided a baseline evaluation and recorded at least one efficacy rating within 2 h after dosing. In case of missing values, last observation carried forward (LOCF) data were used with the proviso that no imputations were made using baseline data.
The differences in efficacy outcomes between the almotriptan- and placebo-treated groups were assessed on the pooled estimates. Both fixed- and random-effects models were used to obtain pooled estimates across the studies. The fixed-effects model leads to valid inferences about treatment effect that is conditional on the available trial data (20), whereas the random-effects model assumes that the particular study samples were drawn from a larger universe of possible studies, and leads to inferences about all studies in that hypothetical population of studies (21, 22). If heterogeneity is not present, the fixed-effects and random-effects models provide similar results.
Comparisons between almotriptan doses and placebo were performed but not between almotriptan doses. No adjustments were made for multiple comparisons. The odds ratios (OR) and their 95% confidence intervals (CIs) for relative changes in efficacy/safety between a given almotriptan dose and placebo were computed using logistic regression. The adjustment variables were dose, study and patients’ baseline pain severity levels. For each efficacy parameter, the relationship between dose and response was established by modelling dose either as a continuous or discrete variable.
Therapeutic gain is defined as the net effect of treatment, i.e. the proportion of patients responding to almotriptan minus the proportion of patients responding to placebo. The number needed to treat (NNT) to obtain a positive response in one patient (defined as the inverse of the therapeutic gain) and 95% CIs were calculated for the pooled data using the fixed-effects model. A significant improvement over placebo was assumed when the upper limit of the 95% CI of the NNT did not include infinity. Therapeutic penalty is defined as the incidence of AEs in the almotriptan group minus the incidence of AEs in the placebo group. Therapeutic gain and therapeutic penalty were obtained using the random-effects model that takes intrastudy variation into account.
The Q statistic was calculated to assess if significant heterogeneity was present among the included trials (23). This statistic tests the null hypothesis that the underlying effect measured by the pooled studies is equivalent. For all statistical tests, the overall type I error rate was set at α= 0.05.
Results
In all, the four studies represented 2294 patients randomized to treatment: 386 in the placebo group and 1908 in the almotriptan group. The mean age of the study population was 41 years and 86% of the patients were female. Details of study characteristics and sample sizes are summarized in Table 1. For the purposes of this study, efficacy and safety analyses were performed only with the placebo patients and the 1635 patients treated with the marketed doses of almotriptan (6.25, 12.5 and 25 mg). The Q statistic test indicated that the results of the studies were homogeneous with respect to treatment effects. Therefore, patient-level data of all studies were pooled for this meta-analysis.
Pain relief and pain free
The proportion of patients who reported pain relief and pain free at various time points up to 2 h after dosing are shown in Figs 1 and 2. At all time points and for each almotriptan dosage, a greater percentage of patients reported pain relief and pain free with almotriptan than with placebo.

Percentage of patients who reported pain relief in the 2-h interval following dosing with placebo (▪, n = 355), almotriptan 6.25 mg (□, n = 527), almotriptan 12.5 mg (▵, n = 721) and almotriptan 25 mg (•, n = 352) using fixed-effects model. P ≤ 0.05 for almotriptan 12.5 mg vs. placebo at all time points and for almotriptan 6.25 mg and 25 mg vs. placebo at 60, 90, and 120 min.

Percentage of patients who reported pain free in the 2-h interval following dosing with placebo (▪, n = 355), almotriptan 6.25 mg (□, n = 527), almotriptan 12.5 mg (▵, n = 721) and almotriptan 25 mg (•, n = 352) using fixed-effects model. P ≤ 0.05 for almotriptan 12.5 mg vs. placebo at all time points, for almotriptan 6.25 mg vs. placebo at 90 and 120 min and for almotriptan 25 mg vs. placebo at 60, 90, and 120 min.
The pain-relief rate with almotriptan 12.5 mg was statistically superior to placebo beginning at 0.5 h postdose (14.9% vs. 8.2%; P < 0.05), and remained superior to placebo at all time points measured (Fig. 1). At 2 h, 55.5%, 61.3% and 63.9% of patients receiving 6.25, 12.5 and 25 mg of almotriptan had pain relief, respectively, compared with 35.2% of patients receiving placebo (all P < 0.05 compared with placebo, using fixed-effects model).
A similar pattern was seen in the pain-free analysis (Fig. 2). Almotriptan 12.5 mg was significantly superior to placebo beginning at 0.5 h after dosing (2.49% vs. 0.69%; P < 0.05). At 2 h, 28.7%, 35.4% and 40.4% of patients were pain free in the almotriptan 6.25-mg, 12.5-mg and 25-mg groups, respectively, compared with 14.5% of patients in the placebo group (all P < 0.05 compared with placebo, using fixed-effects model).
Other efficacy assessments (Table 2)
Effect of almotriptan treatment on various efficacy measurements

Calculated using fixed-effects model; SE, standard error.
P ≤ 0.05 vs. placebo;
P ≤ 0.05 vs. 6.25 mg.
Use of rescue medication
Rescue medication was used from 2 h after initial dosing by 55.5% of patients in the placebo group compared with 38.7%, 31.5% and 31.4% of patients in the 6.25-mg, 12.5-mg and 25-mg almotriptan groups, respectively. The use of rescue medication was significantly lower in all almotriptan-treated groups than in the placebo group (P < 0.05).
Recurrence from 2 to 24 h
In the subsets of patients achieving pain relief by 2 h, the proportion who experienced recurrence was 22.3% in the placebo group, 26.3% in the almotriptan 6.25 mg group, 25.6% in the almotriptan 12.5 mg group and 16.6% in the almotriptan 25 mg group, with no significant differences between the groups. However, a high proportion of these patients in the almotriptan 6.25 mg group (75.4%) and in the almotriptan 12.5 mg group (79.3%) in comparison with those in the placebo group (47.1%) responded to a second dose of almotriptan (P < 0.05).
Sustained pain relief at 24 h
The percentage of patients with pain relief at 2 h and no return of moderate or severe headache pain within 24 h was 26.8% for the placebo group, and 40.9%, 45% and 51.4% for the almotriptan 6.25 mg, 12.5 mg and 25 mg groups, respectively. All active treatments were significantly superior to placebo (P < 0.05).
Migraine-associated symptoms
All three active treatments were significantly more effective than placebo in reducing the incidence of migraine-associated symptoms of nausea, vomiting, photophobia and phonophobia at 2 h postdose (P < 0.05) Across all active treatment groups, a dose-dependent decrease in the incidence of migraine-associated symptoms of nausea, photophobia and phonophobia was observed.
SPF and SNAE
A further analysis of the almotriptan 12.5 mg group was performed to determine the SPF and SNAE rates (Fig. 3). Almotriptan 12.5 mg was associated with a significantly better rate compared with placebo (P < 0.0001) for both SPF (25.8% vs. 11.1%) and SNAE (22.2% vs. 10.4%).

Percentage of patients achieving sustained pain free (SPF) and sustained pain free with no adverse events (SNAE) with almotriptan 12.5 mg (▪) compared with placebo (□). ∗P < 0.0001 vs. placebo.
Safety and tolerability
The incidence of AEs occurring in more than 1% of patients after treatment with study medication is shown in Table 3. There was a dose-related increase in the incidence of AEs across the treatment groups. In the placebo group, 13.5% (52/386) of patients reported AEs compared with 14% (74/527), 16.1% (116/721), and 21.7% (84/387) in the almotriptan 6.25 mg, 12.5 mg, and 25 mg groups, respectively. Almotriptan 6.25 mg and 12.5 mg were as well tolerated as placebo; however, the overall incidence of AEs in the almotriptan 25 mg group was significantly higher than in the placebo group (P < 0.05).
Adverse events reported in >1% of patients

P < 0.05 from a two-tailed exact test for difference with placebo.
The most common AEs in the almotriptan 12.5 mg-treated patients were nausea (1.7% vs. 1.3% for placebo), dizziness (1.4% vs. 1.8% for placebo), paraesthesia (1.4% vs. 0.8% for placebo) and somnolence (1.2% vs. 1.0% for placebo). No AE with almotriptan 12.5 mg occurred in more than 2% of patients. The incidence of chest pain with almotriptan 12.5 mg was very low (0.1%) and lower than that of placebo (0.3%).
The withdrawal rates due to AEs in the almotriptan-treated groups were 0.95%, 0.28% and 1.03% in the almotriptan 6.25 mg, 12.5 mg and 25 mg groups, respectively, compared with 1.04% in the placebo group.
Meta-analysis
To explore the effects of treatment on a range of outcomes, statistical meta-analysis was performed using logistic regression models and the data are summarized in Table 4.
Summary measures of efficacy and safety for almotriptan
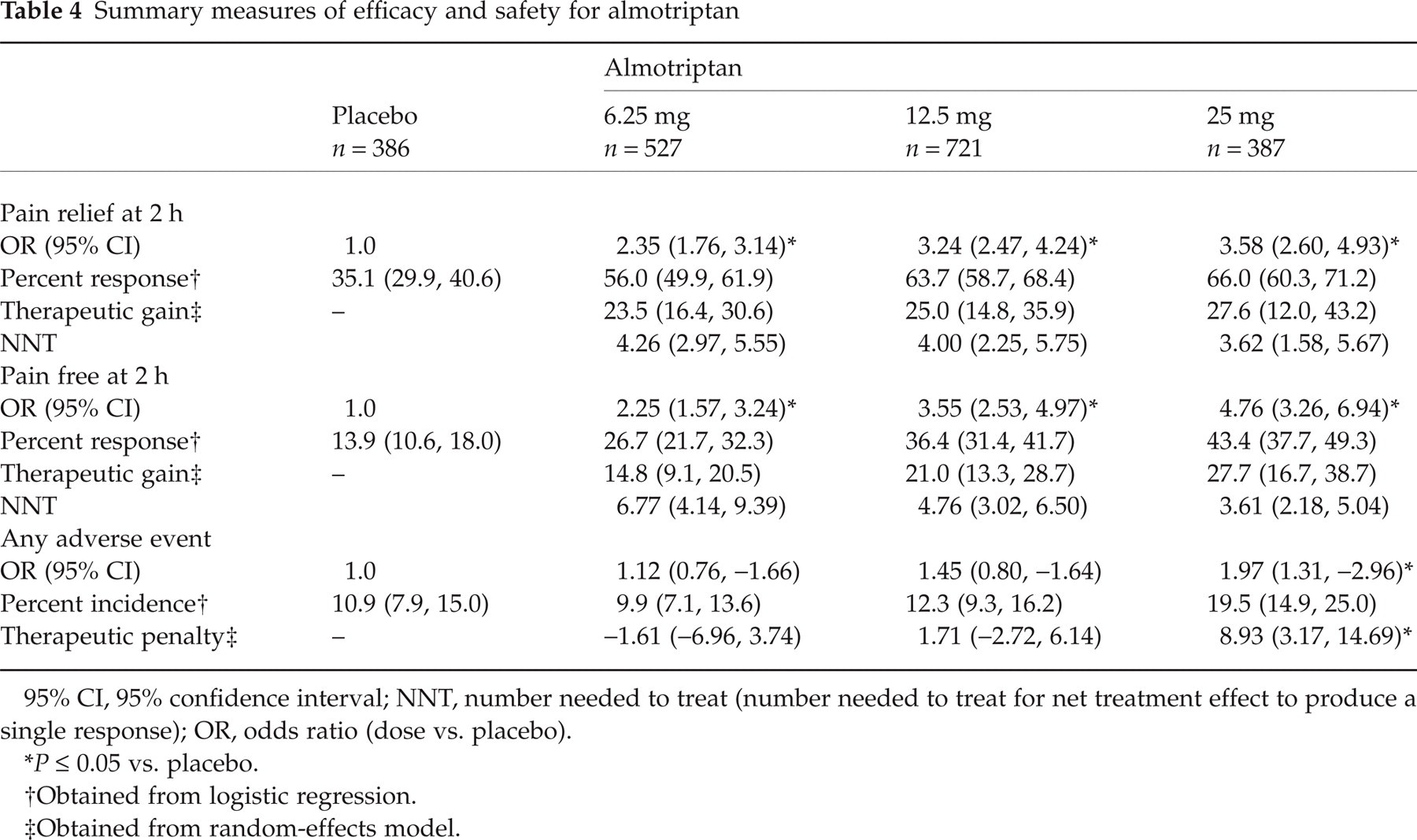
95% CI, 95% confidence interval; NNT, number needed to treat (number needed to treat for net treatment effect to produce a single response); OR, odds ratio (dose vs. placebo).
P ≤ 0.05 vs. placebo.
Obtained from logistic regression.
Obtained from random-effects model.
The ORs of response rate for pain relief at 2 h increased with dose and were significantly different from placebo (P < 0.05). The absolute response rate was 35.1% (95% CI 29.9, 40.6) for placebo, and 56% (95% CI 49.9, 61.9), 63.7% (95% CI 58.7, 68.4) and 66% (95% CI 60.3, 71.2) for almotriptan 6.25 mg, 12.5 mg and 25 mg, respectively. The placebo-subtracted response rates adjusted for the covariables, trial and baseline pain intensity, estimated using the logistic regression model, were 20.9%, 28.6% and 30.9% for almotriptan 6.25 mg, 12.5 mg and 25 mg, respectively. The corresponding values obtained using the random-effects model that do not take into account the baseline pain intensity of the patients were slightly different: 23.5%, 25% and 27.6%, respectively. The NNTs for achieving headache relief at 2 h decreased from 4.26 for almotriptan 6.25 mg to 4.00 for 12.5 mg and to 3.62 for 25 mg, indicating that almotriptan 12.5 mg and 25 mg were more effective than almotriptan 6.25 mg.
Compared with placebo, the ORs of response rate for pain free at 2 h were significantly different for all three almotriptan doses (P < 0.05), with absolute rates of 26.7% (95% CI 21.7, 32.3), 36.4% (95% CI 31.4, 41.7) and 43.4% (95% CI 37.7, 49.3) for almotriptan 6.25 mg, 12.5 mg and 25 mg compared with 13.9% (95% CI 10.6, 18.0) for the placebo group. The covariables-adjusted therapeutic gain using the logistic regression model was 12.8%, 22.5% and 29.5% for almotriptan 6.25 mg, 12.5 mg and 25 mg, respectively. The corresponding random-effects-adjusted estimates were 14.8%, 21.0% and 27.7%. The NNTs for achieving pain-free status at 2 h decreased from 6.77 for almotriptan 6.25 mg to 4.76 for 12.5 mg and to 3.61 for 25 mg, indicating that almotriptan 12.5 mg and 25 mg were more effective than almotriptan 6.25 mg.
Compared with placebo, the ORs for increase in incidence of any AE were not significantly different for almotriptan for 6.25 mg (OR 1.12; 95% CI 0.76, −1.66) or almotriptan 12.5 mg (OR 1.45; 95% CI 0.80, −1.64).
Discussion
In this study, individual patient data from four randomized, double-blind, placebo-controlled trials that assessed the efficacy and tolerability of almotriptan in the treatment of acute migraine, involving 2294 patients, were pooled and subjected to a meta-analysis. Almotriptan was shown to be significantly more effective than placebo on the outcome measures of pain free at 2 h, elimination of migraine-associated symptoms at 2 h and sustained pain relief up to 24 h across the 6.25-mg to 25-mg dosage range investigated. Almotriptan 12.5 mg, assessed for SPF and SNAE, was found to be significantly better than placebo. In addition, a statistically significant advantage to almotriptan 12.5 mg compared with placebo in providing pain relief and pain free was observed to begin at 30 min after dosing.
The strength of this meta-analysis lies in the quality of its data. The four randomized, controlled trials included in this analysis were clinically homogeneous with all having (i) fixed and clearly defined inclusion criteria and (ii) fixed and clearly defined outcomes or outcome measures. It was possible therefore to ensure that all variables had common definitions across studies. Also, the Q statistic test for heterogeneity between studies was not significant, suggesting that all trials were homogeneous and that the treatment effect measured by the pooled studies was equivalent. In short, the circumstances of this meta-analysis assured that comparable data were pooled from all four studies.
The primary conclusion supported by this pooled analysis of individual patient data from four trials is a statistically significant advantage to almotriptan 12.5 mg compared with placebo in providing pain relief and pain-free status beginning at 30 min after dosing. This finding corroborates the 30-min efficacy for pain relief already reported for almotriptan (18). However, the 30-min pain-free results for almotriptan demonstrated in this meta-analysis are significant and robust compared with placebo and have not been previously reported. From a patient perspective, rapid onset of action is the single most important attribute of an acute antimigraine drug (9, 11).
Among the oral triptans, rizatriptan has been shown to be significantly more effective than placebo using pain-relief and pain-free outcomes beginning at 0.5 h postdose. In a meta-analysis of seven randomized, placebo-controlled clinical trials evaluating the efficacy of rizatriptan 10 mg (n = 2068) vs. placebo (n = 1260), the placebo-subtracted response rates for pain relief and pain free at 0.5 h were 6% and 1%, respectively (24). The therapeutic gains for almotriptan 12.5 mg at 0.5 h in the current meta-analysis, 6.7% for headache relief and 1.8% for pain free, are comparable to those observed with rizatriptan 10 mg.
The therapeutic gain at 2 h obtained for all three dosages of almotriptan was robust because both the logistic regression and random-effects models yielded comparable results, again reinforcing the homogeneity of the studies included in this meta-analysis. The therapeutic gain of 25% for pain relief and 21% for pain free with almotriptan 12.5 mg at 2 h is comparable to that reported for other oral triptans (7, 24, 25).
While SPF has been suggested to be the ultimate endpoint for acute migraine trials, it does not take into account tolerability. Side-effects are important to patients, often causing them to delay or avoid taking their prescription medication (26). This delay in treatment has been shown to be associated with greater intensity and longer duration of pain and negative impacts on social functioning. A new composite endpoint, SNAE, has been proposed (27). It is an inclusive endpoint that encompasses all of the attributes that patients desire of their acute migraine medication. In this meta-analysis, the proportion of patients achieving SNAE was significantly better (P < 0.0001) with almotriptan 12.5 mg (22.2%) compared with placebo (10.4%).
Conclusion
This meta-analysis supports and extends findings from previous randomized controlled trials in demonstrating that almotriptan treatment results in significant pain-free rates as early as 30 min after dosing. The excellent tolerability demonstrated in individual trials is supported by this meta-analysis and augmented by the superior SNAE rates compared with placebo.
Footnotes
Acknowledgements
The authors thank Sharon Schaier for providing editorial assistance.
Conflict of interest
C.G.D. has served as a consultant/scientific advisor for advisory boards, clinical trials, and investigator-initiated trials and has been a speaker for Allergan, Almirall, AstraZeneca, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Janssen-Cilag, Merck, Novartis, Ortho-McNeil, Pierre Fabre and Pfizer. J.P. has served as speaker for Almirall, Allergan, AstraZeneca, Eisai, GlaxoSmithKline, Jansen, Menarini, Merck and Pfizer and on the advisory boards for Allergan, Almirall and Merck. D.W.D. has served as an advisory board consultant for Advanced Bionics, Allergan, Almirall, Cierra, Eisai, Endo, GlaxoSmithKline, Medpointe, Medtronic, Merck, Ortho McNeil, Pfizer and Velocimed. He has also received honoraria for speaking at CME events sponsored by Allergan, Endo, Medpointe, Ortho McNeil and Pfizer. A.J.D. has served as a consultant/scientific advisor for advisory boards, clinical trials, and investigator-initiated trials and has been a speaker for Allergan, Almirall, AstraZeneca, Bristol-Myers Squibb, Eisai, GlaxoSmithKline, Janssen-Cilag, Janssen Pharmaceutica, Merck, Medpointe, Menarini, NMT Medical, Novartis, Ortho-McNeil, Pierre Fabre and Pfizer.
