Abstract
Patients expect their acute migraine treatment to have a rapid onset of action, achieve complete pain relief that is sustained for 24 h, and to have a good tolerability profile. Almotriptan has a favourable pharmacokinetic profile that translates clinically to a rapid onset of action and consistent absorption regardless of age, sex, food intake and status of the acute migraine attack. In addition, almotriptan is not associated with any clinically relevant drug-drug interactions. Pain-free status at 2 h postdose is achieved by approximately 39% of patients receiving almotriptan in clinical trials. Recurrence of headaches within 24 h is low with almotriptan (< 22%). Almotriptan has a sustained pain-free rate of 25-27%, which in a meta-analysis of triptans was superior to sumatriptan 100 mg. Almotriptan therapy is associated with a low incidence of adverse events, including those affecting the central nervous system and chest.
Introduction
A survey of patient expectations of acute migraine therapy has revealed that migraineurs value rapid onset of effect, complete pain relief and low rate of headache recurrence (1). Patient satisfaction with treatment is reduced when pain relief persists or returns (2). Good tolerability of treatment is also rated highly by the majority of patients (3). A recent study has found that patients may delay or avoid taking their medications if they have concerns over tolerability (4).
The US Headache Consortium guidelines support these ideals by recommending that migraine therapy should treat attacks rapidly and consistently; restore the patient's ability to function; minimize the use of backup and rescue medications; and have minimal or no adverse events (5).
Data from more than 3000 patients – from both controlled clinical trials and clinical experience studies – have shown that almotriptan has a favourable pharmacokinetic profile, and provides migraine patients with rapid, complete, sustained and reliable relief of pain with excellent tolerability (6).
Selective action
Almotriptan is a selective 5-HT1B/1D receptor agonist, exerting potent vasoconstrictor activity in meningeal arteries (responsible for migraine attacks), with little effect on coronary and pulmonary arteries (linked to certain triptan adverse events) (7,8). Almotriptan has a 60-fold lower affinity for 5-HT1A receptors than for 5-HT1B and 5-HT1D receptors, and no appreciable affinity for non-serotonergic receptors (7). Almotriptan inhibits neurogenic extravasation implicated in headache pathophysiology more potently than sumatriptan (8).
No clinically significant cardiovascular effects
The effects of almotriptan on the cardiovascular system have been investigated in healthy volunteers, migraineurs and hypertensive subjects. Results of several clinical studies (with doses of almotriptan ranging from 5 to 200 mg) have demonstrated no clinically significant effect of almotriptan on heart rate, blood pressure, ECG or other cardiovascular parameters (9–11). However, as a class, triptans are contraindicated in patients with ischaemic heart disease, previous myocardial infarction, coronary vasospasm or uncontrolled or severe hypertension (12).
Favourable pharmacokinetic profile
Rapid absorption is important for a migraine medication given patients’ desire for fast pain relief. Almotriptan is rapidly absorbed after an oral dose; 75% of the dose is absorbed within the first hour after administration, and peak plasma concentrations are reached at 1.5–2 h postdose (13). Absorption of almotriptan is consistent, regardless of whether it is administered during or after a migraine attack (14–17). Among the other triptans, naratriptan takes 3.5 h to reach maximum plasma drug concentration during an attack (tmax) and frovatriptan has a tmax which ranges between 2 and 4 h. Absorption of almotriptan is unaffected by food, and is similar both in men and women, and in adolescents and adults. No dose adjustment is necessary in the elderly.
As with all triptans, almotriptan must be administered with caution in patients with renal or hepatic impairment. In patients with severe renal disease, the starting dose of almotriptan is 6.25 mg with a maximum daily dose of 12.5 mg.
Oral almotriptan has one of the highest bioavailability scores of all the triptans (70%) – much higher than that of oral sumatriptan (14%) indicating consistent efficacy (18). Migraine attacks are frequently accompanied by gastric stasis, which can impede absorption. Therefore, high bioavailability is a desirable attribute for an acute migraine medication. The half-life of almotriptan is 3.0–3.7 h, which is longer than that of sumatriptan (approximately 2 h). No active metabolites of almotriptan have been identified, and this may explain the low incidence of adverse events observed with this agent. As almotriptan is degraded via three routes it is less prone to adverse drug interactions than other triptans. For example, unlike sumatriptan, rizatriptan and zolmitriptan, almotriptan is not contraindicated in patients taking monoamine oxidase inhibitors. Furthermore, unlike eletriptan (which should not be used within 72 h of administration of ketoconazole, itraconazole, nefazodone, troleandomycin, clarithromycin, ritonavir and nelfinavir) no dose adjustments are required with concomitant cytochrome CYP3A4 inhibitors (19). Almotriptan has been coadministered with fluoxetine, verapamil, propanolol, moclobemide and ketoconazole in studies with healthy volunteers, to identify whether almotriptan interacts with any of these commonly used agents (20–23). No clinically relevant changes in the pharmacokinetics of almotriptan were observed during these studies, and no clinically relevant changes in ECG or laboratory parameters were reported. These findings mean that almotriptan can be taken concomitantly with monoamine oxidase inhibitors, selective serotonin re-uptake inhibitors and other drugs (with the exception of other 5-HT1B/1D agonists or ergot-containing medications) without the need for dose modification (6,24). Similarly, almotriptan has no clinically relevant interaction with alcohol or oral contraceptives, which may potentially be used concomitantly by patients (25,26).
Rapid and complete relief of pain restoring the patient's ability to function
Almotriptan has a rapid onset of action showing significantly greater efficacy than placebo at relieving pain within 30 min (27). In contrast, the onset of action for both naratriptan and frovatriptan is markedly longer (estimated at 3 h and 2 h, respectively) (Fig. 1) (13,28).

Rapid onset of efficacy (pain relief) is an important attribute for triptan therapy. Reproduced from (13) with permission.
Placebo-controlled studies have shown the 2-h response rate of almotriptan to be around 60–70% (27,29–31). In an early dose-finding study of treatment for a single migraine attack of moderate or severe intensity in 742 patients, 59% of patients treated with almotriptan 12.5 mg vs 33% of those in the placebo group showed headache response at 2 h (P < 0.05) (29). Higher response rates were found in a different trial involving 722 patients treating multiple attacks of moderate or severe intensity with almotriptan (27). In this study, response rates at 2 h were 70% with almotriptan 12.5 mg vs 38% with placebo (P < 0.001). Furthermore, a meta-analysis of placebo-controlled trials of almotriptan in moderate to severe migraine in 2294 patients found response rates of 61% with almotriptan 12.5 mg compared with 35% with placebo (P < 0.01) (31).
Pain-free efficacy is a key criterion for patient satisfaction. In the dose-finding single-attack study in 742 patients with moderate to severe intensity migraine mentioned above, the pain-free rate with almotriptan 12.5 mg was 38%, compared with 11.3% with placebo (P < 0.0001) (29). Similarly, the placebo-controlled multiple-attack study in 722 patients with moderate to severe migraine found a pain-free rate across attacks of 39% for almotriptan vs 16% for placebo (P < 0.001) (27).
In a meta-analysis of 53 randomized, double-blind, controlled clinical trials of oral triptans involving more than 24 000 patients, almotriptan 12.5 mg was more effective than sumatriptan 100 mg at achieving pain-free status at 2 h, as were eletriptan 80 mg and rizatriptan 10 mg (32). Naratriptan 2.5 mg and eletriptan 20 mg were not as effective as sumatriptan 100 mg, while the remaining triptan doses evaluated (i.e. sumatriptan 25 mg and 50 mg; zolmitriptan 2.5 mg and 5 mg; rizatriptan 5 mg and eletriptan 40 mg) were not significantly different from sumatriptan 100 mg.
Furthermore, in a 1-year open-label study, 762 patients experiencing 13 751 acute migraine attacks of any intensity were evaluated for their response to treatment with almotriptan 12.5 mg. The 2-h pain-free rate for almotriptan found in this long-term study was 58% (33).
A recent postmarketing surveillance study has evaluated the efficacy of almotriptan in routine clinical practice, and provided further evidence of the rapid and complete pain-relieving efficacy of this triptan. The study, performed in Germany, included 2131 migraine attacks treated with almotriptan among 899 patients (34). Relief of pain at only 1 h postdose was observed in 84.5% of patients. In addition, by 1 h postdose 41.4% of patients were pain free (Fig. 2). This cohort may provide a more accurate reflection of the type of patients observed in daily practice, i.e. a broader patient population, than those selected for participation in clinical trials.

Pain relief and pain-free rates with almotriptan recorded in clinical practice. Reproduced from (34) with permission.
Minimizing the need for rescue medication
Headache recurrence can be a problem for both patients and clinicians. Although patients can take a second dose of medication within 24 h, this may cause a loss of confidence in the treatment, or encourage medication overuse that can lead to further headaches. Repeat dosing also increases the per-attack cost of treatment.
Rates of headache recurrence vary, probably due to pharmacokinetic differences between the triptans, such as half-life (35). In a recent randomized, double-blind, placebo-controlled study, single doses of almotriptan 12.5 mg were compared with sumatriptan 100 mg in single migraine attacks. In patients experiencing moderate pain, the rate of headache recurrence within 24 h with almotriptan was similar to that of sumatriptan (22.7 vs 22.4%; vs 16.7% for placebo). However, in those patients with severe headache, recurrence was lower with almotriptan than with sumatriptan or placebo (8.8 vs 28.9 vs 27.3%, respectively). Overall, headache recurrence occurred at a rate of 18.0% with almotriptan, compared with 24.6% with sumatriptan and 19.5% in the placebo group (30).
In the triptan meta-analysis, headache recurrence with almotriptan was either similar to or lower than that of sumatriptan 100 mg (approximately 30%). In contrast, rizatriptan 5 mg and 10 mg were associated with significantly higher rates of headache recurrence than sumatriptan 100 mg (Fig. 3) (32).

Headache recurrence rates of oral triptans (within 2–24 hours post-dose). Reproduced from (32) with permission from Elsevier.
The almotriptan clinical experience study also recorded a low rate of headache recurrence with almotriptan in the wider patient community (34). From 2131 attacks, headache recurrence occurred at a rate of 22% with the first dose of almotriptan. In patients taking a second dose of almotriptan, only 12% experienced a recurrence of headache.
Sustained pain-free efficacy
Sustained pain-free efficacy measures the propensity of a treatment to not only achieve a pain-free state at 2 h postdose but also sustain this effect by preventing headache recurrence and the need for rescue medication over 24 h. This outcome is patient-centred as it accounts for the important patient requirement of being completely free of pain over a sustained period, and is predictive of patient satisfaction with treatment. The International Headache Society (IHS) has described sustained pain-free as the ideal outcome for assessing response to acute migraine therapy (36), although it is the most difficult to achieve.
In three randomized, double-blind, placebo-controlled trials with almotriptan 12.5 mg, sustained pain-free rates of 25–27% have been recorded in patients with moderate to severe migraine headache (n = 1791 across studies) (27,37–39). These rates are significantly higher than those achieved with placebo (8–12%) and similar to the rate achieved with sumatriptan 100 mg (22%) (Fig. 4).

In the triptan meta-analysis, significantly more patients treated with almotriptan 12.5 mg achieved a sustained pain-free state than those treated with sumatriptan 100 mg (which had a rate of approximately 20%). Rizatriptan 10 mg and eletriptan 80 mg also showed favourable sustained pain-free rates while eletriptan 20 mg had significantly lower sustained pain-free rates and the other triptan doses studied were not significantly different from sumatriptan 100 mg (32) (Fig. 5).
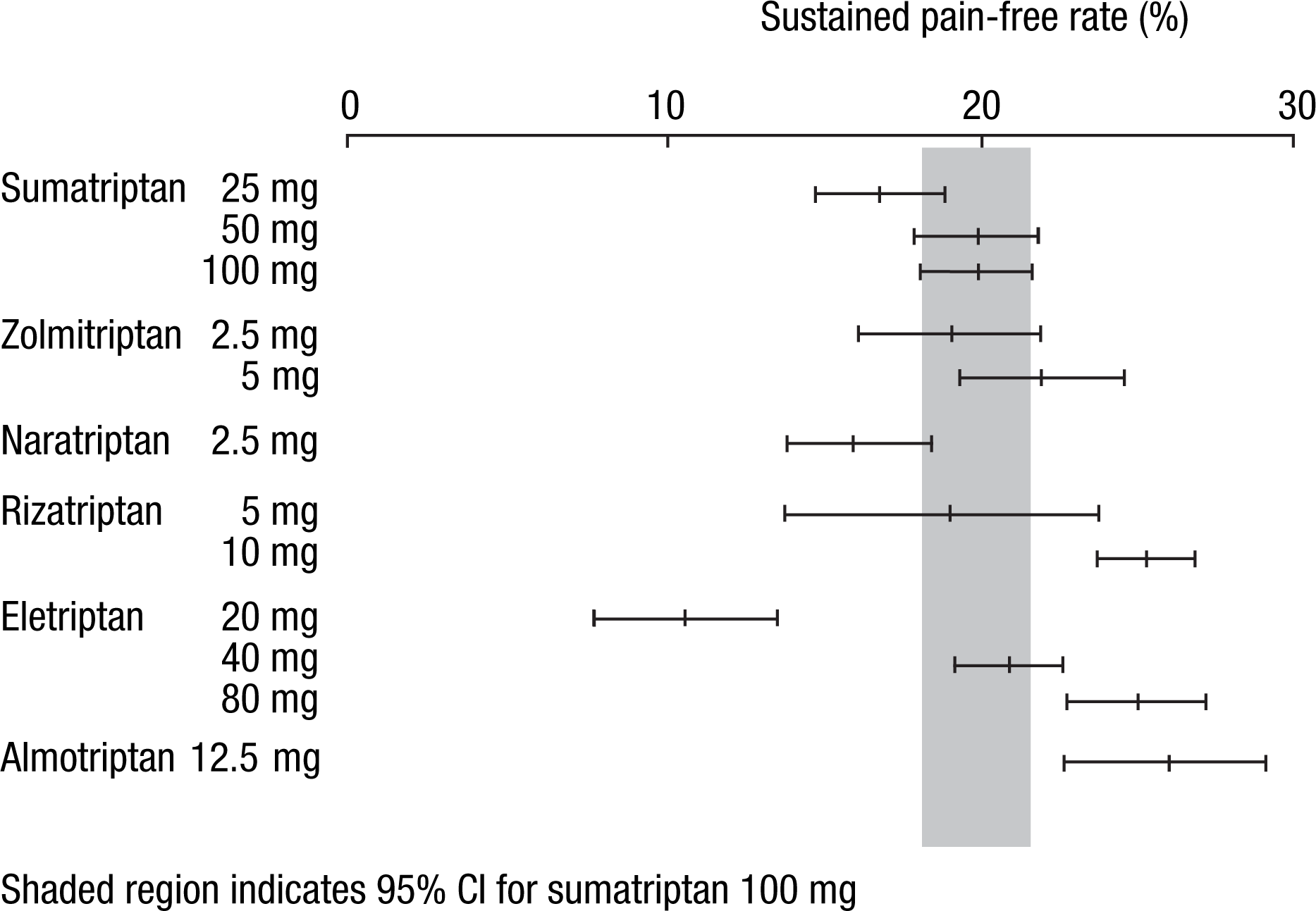
Sustained pain-free rates of the oral triptans. Reproduced from (32) with permission from Elsevier.
Treatment with minimal or no adverse events
Allaying concerns of triptan-related adverse events is important for patient confidence in therapy. Patients may delay their treatment because of fear of side-effects, which leads to reduced treatment response rates and worse outcomes, including more intense pain, a need to lie down and an extended duration of headache (4). Clearly, an acute migraine medication that combines rapid and sustained symptom relief with good tolerability would be preferred by patients.
Triptan-related adverse events include central nervous system (CNS) symptoms and symptoms that are often considered as ‘chest’ symptoms. CNS adverse events (such as dizziness, somnolence, asthenia) can be troublesome to the patient, especially those who need to remain active. Although studies have shown chest symptoms, such as palpitations, chest and throat tightness to be benign and not due to coronary vasoconstriction or cardiac ischaemia, they can still be anxiety-provoking for patients (12).
As previously discussed, differences in the pharmacokinetic profiles of the various triptans translate to a differing propensity to cause adverse events. In a randomized, double-blind, placebo-controlled study of 688 patients comparing almotriptan 12.5 mg with sumatriptan 100 mg, the incidence of adverse events reported showed that almotriptan was as well tolerated as placebo (8.7 vs 6.1%; P = 0.493 (30). However, the incidence of adverse events in the sumatriptan group was significantly higher than that for almotriptan (8.7 vs 22.2%; P < 0.001).
The triptan meta-analysis concluded that only naratriptan 2.5 mg and almotriptan 12.5 mg have a lower incidence of adverse events overall than sumatriptan 100 mg, and that almotriptan and naratriptan have an incidence of adverse events not significantly different from that seen with placebo (32). However, like frovatriptan, naratriptan has a slow onset of action; these two agents also have a lower 2-h headache response rate than the other oral triptans (13,28). In addition, almotriptan 12.5 mg was the only triptan with a lower incidence of ‘CNS’ and ‘chest’ adverse events than sumatriptan 100 mg (Fig. 6) (32).

Comparative tolerability of the oral triptans. Placebo-subtracted incidence of: (a) any adverse event; (b) CNS adverse events; and (c) chest symptoms. Reproduced from (32) with permission from Elsevier.
Satisfying patient needs
Patients have high expectations for acute migraine therapy. The majority of patients desire an agent that provides rapid, complete and sustained pain relief, but which causes few or no adverse events.
Considering tolerability and sustained pain-free efficacy together, the composite endpoint of sustained pain-free with no adverse events (SNAE), provides a useful assessment of how closely a triptan can meet these patient expectations. When using this measure, almotriptan has the most favourable profile of all the oral triptans (Fig. 7).

Sustained pain-free with no adverse events (SNAE) profiles versus sumatriptan 100 mg for the oral triptans. Modified from (32).
TRIPSTAR examined the relative importance of prespecified treatment attributes of acute migraine medication among 206 triptan users and 209 triptan-naïve patients with high migraine-related disability (40,41). The treatment attributes included pain-free rates at 1 and 2 h, sustained pain-free rates, consistency of effect, and incidence of CNS, chest and other adverse events. These importance weightings were then combined with data on relative performance of the oral triptans on these attributes (32) and inserted into a multi-attribute decision model to identify the preferred triptans. At the group level of analysis, only almotriptan 12.5 mg, eletriptan 80 mg and rizatriptan 10 mg were significantly closer to the hypothetical ideal triptan than the reference product sumatriptan 100 mg.
Migraine headaches can significantly reduce the health-related quality of life, physical and social functioning and productivity of sufferers due to the associated severe pain and headache-related disability. A double-blind, parallel-group preference study compared the ability of almotriptan and sumatriptan to improve these measures (42). A total of 1173 patients with moderate to severe migraine with no previous history of triptan therapy were randomized to receive either almotriptan 12.5 mg or sumatriptan 50 mg. In each patient group, measures were taken of health-related quality of life (HRQoL), satisfaction with treatment and functional status. A 15-question HRQoL questionnaire translated to a score between 3 and 21, where 21 was the highest HRQoL, while treatment satisfaction and functional status were measured on a scale from 0 to 100 where 100 represented most satisfied / best functioning. All patients reported substantial impairment in HRQoL due to their migraine pain and symptoms, which improved with treatment response. At 48 h after treatment of an acute attack, there was no significant difference between the two treatment groups in terms of satisfaction with pain relief (mean score approximately 50 in both groups) or functional status, which returned to almost 90% functioning at 24 h postdose compared with baseline. Patients taking almotriptan, however, were more satisfied with the tolerability of their treatment than patients taking sumatriptan (a mean satisfaction score of 81.29 was reported with almotriptan vs only 77.46 with sumatriptan; P = 0.016). Significantly more patients receiving sumatriptan were ‘extremely bothered’ by side-effects, compared with those receiving almotriptan (4.0 vs 1.7%; P = 0.02).
Conclusions
Almotriptan exhibits a well-balanced profile in terms of efficacy and tolerability that has been demonstrated in both clinical trials and routine clinical practice. Importantly, its pharmacokinetic profile is unaffected by sex, age, food intake or the time of administration within the acute migraine attack – thus maintaining a consistent effect. Almotriptan is not associated with any clinically significant drug–drug interactions, which can be important for the migraineur population who may be receiving other medications, for example for migraine prevention.
Key attributes for an acute migraine therapy are rapid onset, sustained pain-free efficacy and good tolerability. Almotriptan begins to achieve a response within 30 min of administration. Data from individual clinical trials, a major meta-analysis of clinical trials of oral triptans and from studies in routine clinical practice have shown that almotriptan achieves pain-free status at 2 h postdose in a greater proportion of patients than that observed with sumatriptan 100 mg. Recurrence of headache can be a significant problem for both patients and physicians. Almotriptan 12.5 mg is associated with rates of recurrence similar or lower to those seen with sumatriptan 100 mg (while rizatriptan 5 mg and 10 mg have higher rates of recurrence than sumatriptan 100 mg) (32). Sustained pain-free status is the ideal outcome of acute migraine therapy, and is achieved by 25–30% of patients receiving almotriptan therapy, which compares favourably with sumatriptan.
Tolerability of acute migraine therapy is extremely important to patients. Almotriptan has an excellent tolerability profile, with an incidence of adverse events reported in clinical trials similar to that of placebo. In the triptan meta-analysis, almotriptan had a lower incidence of adverse events than sumatriptan. In addition, almotriptan was the only triptan with a lower incidence of both CNS and chest adverse events than sumatriptan. The importance of tolerability to patients has been confirmed by a preference study where patients favoured almotriptan over sumatriptan due to its superior tolerability.
When all the key attributes of an acute therapy for migraine are evaluated, almotriptan compares favourably with the other triptans. Indeed, the triptan meta-analysis of oral triptans identified almotriptan 12.5 mg to be among the three most likely to provide treatment success on these measures overall (32). The two others triptans identified were the higher doses of eletriptan (80 mg) and rizatriptan (10 mg). Therefore, almotriptan should be considered as a first-line therapy, especially when good tolerability and proven, consistent efficacy are important – attributes of particular relevance to patients.
