Abstract
Almotriptan is a novel and specific serotonin 5-HT1B/1D agonist for the acute treatment of migraine. This randomized, single-dose, double-blind, multicentre, study assessed the efficacy and safety of oral almotriptan (12.5 mg and 25 mg) in patients with migraine, and compared it with the standard treatment (sumatriptan 100 mg) and placebo. A total of 668 patients treated one migraine attack of moderate or severe intensity with study medication. The primary efficacy assessment was migraine pain relief, improvement from severe or moderate pain to mild or no pain, at 2 h after treatment. Response rates, stratified for variation in baseline pain levels, for both almotriptan doses were equivalent to sumatriptan and significantly better than placebo. Other efficacy assessments confirmed the equivalence of the almotriptan groups with the sumatriptan group. Almotriptan 12.5 mg was as well tolerated as placebo (P = 0.493) and significantly better tolerated than sumatriptan (P < 0.001), in terms of the overall incidence of adverse events. There was no statistically significant difference in the incidence of adverse events between almotriptan 25 mg and sumatriptan 100 mg (P = 0.376). The results from this large clinical study indicate that the new, specific 5-HT1B/1D agonist, almotriptan, is an effective and well-tolerated treatment for migraine pain.
Introduction
Migraine is a common disorder characterized by episodic headache, often accompanied by disturbed vision and gastrointestinal complaints. The 1-year prevalence of migraine is approximately 15–18% in women and 6% in men; thus it can create a large socio-economic problem (1). Surveys have shown that migraine results in a significant economic burden in the form of missed workdays and reduced work performance (2).
Migraine pain arises, in part, from excessive dilatation of cranial blood vessels. Serotonin (5-HT) is a brain neurotransmitter that has a potent vasoconstrictor action. It is effective against migraine, but is nonspecific in its action (3). Serotonin acts on several receptor subtypes (4), including 5-HT1, the type most implicated in migraine. Agonists of the 5-HT1 receptors have been developed to provide specific symptomatic relief of migraine (5). Although it can be severely debilitating during attacks, migraine can be dramatically improved by effective acute therapies (6, 7). Sumatriptan was the first of the 5-HT1 agonists to be successfully developed for the treatment of migraine (6, 8, 9). However, it also has some disadvantages including recurrence of headaches (10), poor oral bioavailability (14%) and occasional induction of cardiovascular adverse events (9).
There is therefore a need to develop compounds with improved efficacy, tolerability and safety profiles relative to sumatriptan. Other triptan compounds have been developed to meet this need, and these include almotriptan, a specific 5-HT1B/1D agonist. The pharmacokinetics of almotriptan have been investigated in healthy volunteers. Almotriptan has been shown to have high oral bioavailability (70%) and an elimination half-life of 3–4 h (11). Clinical studies show that oral almotriptan exhibits good efficacy and tolerability in the acute treatment of migraine attacks, characterized by a rapid onset of action and a low recurrence rate (12–15). Almotriptan may be better tolerated than sumatriptan. Fewer chest symptoms were reported with almotriptan than with sumatriptan in clinical trials (16).
The objective of this study was to assess the efficacy and safety of oral almotriptan at two different doses (12.5 mg and 25 mg) in a large number of patients with migraine, and compare it with the standard treatment (sumatriptan 100 mg) and placebo.
Methods
Design
This randomised, single-dose, double-blind, parallel-group, multicentre clinical study was conducted predominantly in primary care practices in five European countries (Spain, Germany, UK, France and the Netherlands). The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice and approved by the relevant Ethics Committees. Patients gave their written informed consent.
Patients
Male and female migraine patients aged between 18 and 65 years who met the International Headache Society criteria for migraine with or without aura (17) were eligible to participate in the study. Patients had at least a 1-year history of one to six migraine attacks per month, with an age of onset of migraine of less than 50 years and at least 24 h free from headache between their attacks. Female patients of childbearing age were not pregnant on testing and practised contraception. Patients were tested with an electrocardiogram (ECG) and were enrolled if there were no abnormalities of clinical relevance.
Patients were excluded from the study if they had migraine with prolonged aura, familial hemiplegic migraine, migrainous infarction, vertebrobasilar migraine or Raynaud's phenomenon associated with migraine, or any other significant medical condition. In addition, patients suffering from cardiovascular disease (cardiac ischaemia, atherosclerosis, cardiac arrhythmia or hypertension (blood pressure; systolic >160 mmHg, diastolic >95 mmHg)), alcoholism, drug abuse or mental retardation were also excluded from the study. Patients taking certain drugs were not permitted to enter the study (any investigational drug 1 month before study medication; monoamine oxidase inhibitors, lithium, selective serotonin reuptake inhibitors, ergots or derivatives or methysergide in the 2 weeks prior to study medication).
Patients were withdrawn from the study if they did not experience a migraine attack within 8 weeks of screening or needed symptomatic medication for migraine in the 2 days before taking the study medication.
Treatment
Patients were randomized in a 2 : 1 ratio (active treatment: placebo) to receive a single oral dose of almotriptan (12.5 mg or 25 mg), sumatriptan (100 mg) or placebo capsules to treat one migraine attack. Patients took their study medication when the attack was severe or moderate in intensity.
Escape medication, chosen by the investigator with the patient, could be taken if migraine pain did not disappear or become mild within 2 h of treatment. However, ergot derivatives were not allowed as escape medication. A second dose of the study medication was allowed only for the treatment of relapse after recurrence of headache (patient had mild or no pain at 2 h after treatment but moderate or severe pain reappeared within 24 h) Concomitant treatments permitted as migraine prophylaxis included valproic acid, beta-blockers and calcium antagonists.
Assessments
Patients recorded efficacy assessments on self-assessment cards 1, 2, and 24 h after treatment. The primary efficacy assessment was migraine pain relief, defined as improvement from severe or moderate pain to mild or no pain at 2 h after treatment, according to the following 4-point scale:
no headache.
mild headache allowing normal activity.
moderate headache, disturbing but not prohibiting normal activity, which does not require bed rest.
severe headache, prohibiting normal activity, which may require bed rest.
Secondary efficacy assessments were pain relief at 1 h, pain free at 1 and 2 h, use of escape medication, relief of migraine-associated symptoms (nausea, vomiting, photophobia and phonophobia) and migraine recurrence.
Safety and tolerability were assessed by physical examinations, standard laboratory tests, vital signs, ECGs and recording of adverse events. Adverse events were collected using diary cards and by personal communication between the patient and physician during clinic visits. The incidence of chest symptoms (including pain, tightness and discomfort) was collected during the study.
Statistical analysis
It was assumed from the available data that a decrease of migraine pain would occur in approximately 30% of patients treated with placebo and 70% of patients treated with either almotriptan 25 mg or sumatriptan after 2 h. With this estimation, and assuming a 10% drop-out rate, the required sample size was calculated as 95 patients in the placebo group and 190 patients in each of the active treatment groups. A total of 665 patients were required in order to obtain 600 evaluable patients.
Categorical demographic data were compared between the four treatment groups using χ2 tests. In order to validate the study, the pooled patients who received almotriptan 25 mg or sumatriptan were compared with the patients who received placebo using a one-sided Fisher's exact test at the 5% level. The confirmatory part of the study consisted of a predefined sequence of equivalence tests, the null hypothesis for the first being that almotriptan 25 mg was not equivalent to sumatriptan, the second that almotriptan 12.5 mg was not equivalent to sumatriptan. Both tests were performed at the 5% level. A 90% confidence interval (CI) for the difference of the probability for success was obtained using asymptotic methods (18) and equivalence was assumed if the CI was within −0.15 and 0.15. The analysis was performed on the intention-to-treat population. In addition, two-sided 95% CI according to Pearson-Clopper (based on an F-distribution) were provided for the response rates of the four treatment groups.
As the pain before intake of the test drug was not balanced within the four treatment groups, exploratory equivalence analyses (90% CI adjusted for baseline pain) were performed for the primary endpoint as well as the following secondary endpoints: response rate at 1 h, pain free at 1 and 2 h, presence of nausea, vomiting, photophobia and phonophobia at 2 h, and migraine recurrence.
Results
Disposition of patients
A total of 668 patients aged between 17 and 66 years were recruited to the study (184 patients received almotriptan 12.5 mg, 191 received almotriptan 25 mg, 194 received sumatriptan 100 mg, and 99 received placebo). Eight patients withdrew from the study (one patient receiving almotriptan 12.5 mg, six receiving almotriptan 25 mg, and one receiving sumatriptan). The reasons for withdrawal were intake of prohibited medication (one patient receiving almotriptan 25 mg) and protocol violations (one patient receiving almotriptan 12.5 mg, five receiving almotriptan 25 mg, and one receiving sumatriptan).
The four treatment groups were comparable with respect to age, sex, height and weight (Table 1). However, there were significant differences in baseline pain between the almotriptan groups and the placebo group (P=0.006 and P=0.037 for placebo vs. almotriptan 12.5 mg and 25 mg, respectively), with a higher proportion of patients with severe pain in the almotriptan groups.
Patient characteristics

Efficacy assessments
Pain relief at 2 h after treatment
Considerably more patients reported pain relief at 2 h following treatment with almotriptan 12.5 mg, almotriptan 25 mg or sumatriptan than with placebo treatment. The 2-h response rates were 56.8% for almotriptan 12.5 mg, 56.5% for almotriptan 25 mg and 63.7% for sumatriptan, compared with 42.4% for placebo (Table 2). A higher percentage of patients responded when the pain was moderate than when it was severe (Table 2 and Fig. 1). Nevertheless, all the three active treatment groups were markedly better than placebo. Both doses of almotriptan were equivalent to sumatriptan 100 mg with the 90% CI interval inside the range of the equivalence region (Fig. 2).
Response to treatment assessed by migraine pain relief (to mild or no pain) at 2 h after treatment, according to baseline pain levels (moderate plus severe, moderate, and severe)
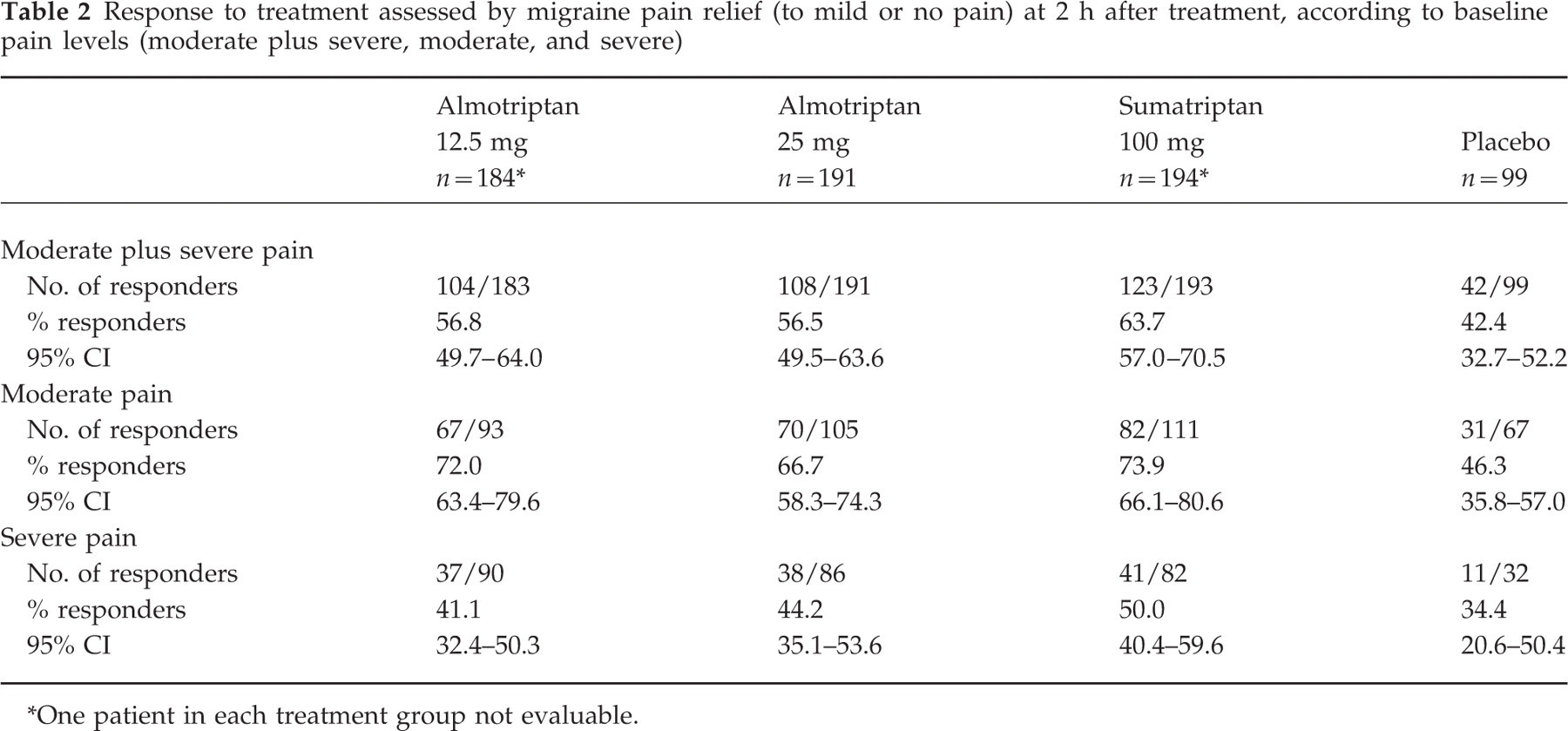
∗One patient in each treatment group not evaluable.

Proportion of patients (95% CI) reporting migraine pain relief (to mild or no pain) at 2 hours after treatment, stratified for baseline pain (moderate and severe). moderate pain; □ severe pain.
moderate pain; □ severe pain.

90% CIs for the difference between almotriptan (12.5 mg and 25 mg) and sumatriptan response rates, stratified for baseline pain (moderate and severe).
Pain free at 2 h after treatment
The proportion of patients who were pain free at 2 h after treatment, stratified by baseline pain level, was substantially higher in the active treatment groups than in the placebo group (Table 3). There were no differences between the active treatment groups.
Migraine pain relief (to mild or no pain) or pain free response at 1 and 2 h after treatment, stratified for baseline pain levels (moderate and severe pain)

Other efficacy assessments
The response to treatment stratified by baseline pain level (reduction in migraine pain to a mild intensity or pain free) at 1 h after treatment was comparable across all three active treatment groups (Table 3).
There was no significant difference between the treatment groups in the proportion of responders who had a recurrence of their migraine attack within 24 h of drug administration for baseline moderate pain (almotriptan 12.5 mg=22.7%, almotriptan 25 mg=14.9%, sumatriptan=22.4%, placebo=16.7%). However, for treatment of severe pain, there was an advantage for both almotriptan 12.5 mg and almotriptan 25 mg over sumatriptan and placebo (almotriptan 12.5 mg=8.8%, almotriptan 25 mg=16.2%, sumatriptan=28.9%, placebo=27.3%). Overall levels of recurrence were 18.0 (CI: 12.09–25.76), 15.4 (CI: 9.89–22.42), 24.6 (CI: 18.05–32.09) and 19.5% (CI: 10.10–32.46) for almotriptan 12.5 mg, almotriptan 25 mg, sumatriptan and placebo, respectively.
The three active treatment groups did not differ in the incidence of migraine-associated symptoms after treatment with study medication (Table 4). All comparisons between almotriptan and sumatriptan, adjusted for baseline pain severity, had 90% CIs within the equivalence region. All three active treatments reduced the incidence of migraine-associated symptoms compared with placebo.
Migraine-associated symptoms at baseline and at 2 h after treatment, stratified for baseline pain levels (moderate and severe)

Escape medication was taken more frequently by patients in the placebo group than by those in the active treatment groups (almotriptan 12.5 mg=38.6%; almotriptan 25 mg=38.2%; sumatriptan=32.4%; placebo=55.5%).
Safety and tolerability
All study medications were well tolerated and there were no withdrawals due to adverse events. The incidence of adverse events occurring in more than 2% of patients after treatment with study medication is shown in Table 5. Almotriptan 12.5 mg was as well tolerated as placebo (adverse event incidence: almotriptan=8.7%, placebo=6.1%; P=0.493) and was significantly better tolerated than sumatriptan (adverse event incidence: almotriptan 12.5 mg=8.7%, sumatriptan 100 mg=22.2%; P<0.001). There was no statistically significant difference in adverse event incidence following treatment with almotriptan 25 mg and sumatriptan 100 mg (P=0.376).
Incidence of all adverse events after medication and those occurring in more than 2% of patients in any treatment group
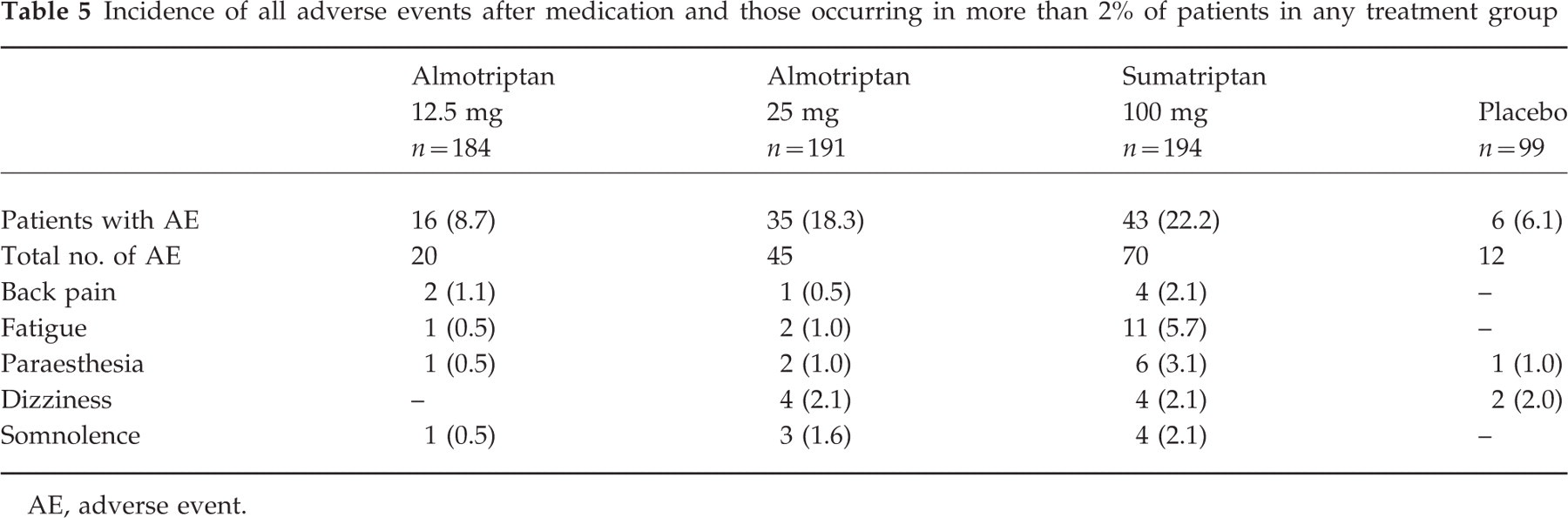
AE, adverse event.
The most frequently reported adverse events in the almotriptan 25 mg group were dizziness (2.1%) and somnolence (1.6%). The commonest adverse events in the sumatriptan group were fatigue (5.7%), paraesthesia (3.1%), dizziness (2.1%) and somnolence (2.1%). No adverse event with almotriptan 12.5 mg occurred in more than 2% of patients. The incidence of chest symptoms was 0% with almotriptan 12.5 mg, 1.6% with almotriptan 25 mg, 1% with sumatripan 100 mg and 0% with placebo.
No clinically relevant differences were observed between the four groups in physical examinations, vital signs, ECGs and laboratory analyses.
Discussion
The results from this large clinical study indicate that the new, specific 5-HT1B/1D agonist, almotriptan, is an effective and well-tolerated treatment for migraine pain. This randomized, single-dose, double-blind, parallel-group, multicentre clinical trial was conducted in five European countries and included 668 patients with migraine. The primary efficacy assessment was migraine pain relief, improvement from severe or moderate pain to mild or no pain, at 2 h of treatment. Secondary efficacy assessments included pain relief at 1 h, pain free at 1 and 2 h, use of escape medication, relief of migraine-associated symptoms and migraine recurrence. Although the treatment groups were comparable with regard to demographic details, the groups differed significantly with respect to the level of baseline pain, with more patients reporting severe pain in the almotriptan groups. Therefore, the pain relief was evaluated after stratification for baseline pain levels.
Overall response rates for almotriptan at 2 h were equivalent to that for sumatriptan (56.8%, 56.5% and 63.7% for almotriptan 12.5 mg, almotriptan 25 mg and sumatriptan, respectively). The proportion of patients reporting a response at 2 h after treatment was higher for patients with moderate pain than for those with severe pain. However, the three active treatment groups were superior to placebo for treatment of both moderate and severe pain. The response rates for the two almotriptan groups were: moderate pain—almotriptan 12.5 mg=72.0%, almotriptan 25 mg=66.7%; severe pain—almotriptan 12.5 mg=41.1%, almotriptan 25 mg=44.2%.
Published overall 2-h response rates for the oral triptans, sumatriptan, zolmitriptan. naratriptan and rizatriptan, range between 43 and 77%, with response rates for placebo ranging between 16 and 47% (19). Similar response rates were observed in this study. Although the placebo response rate was towards the high end of the triptan range (42.4% at 2 h), the active treatments were significantly superior to placebo for many of the endpoints assessed. This high placebo response may be due to the study being conducted predominantly in primary care practices, where patients may have had higher expectations of the efficacy of the treatment they were given than if they had been treated in specialist centres. It should be noted that, in accordance with the protocol, patients were instructed to take the study medication when the migraine attack was moderate or severe in intensity. In normal practice, patients are likely to take their medication as soon as the attack starts and more favourable response rates to almotriptan would be expected.
The pain-free response at 2 h after treatment was much lower in the placebo group than in the active treatment groups, which all had similar response rates. Other efficacy assessments did not reveal any clinically significant differences between the almotriptan groups and the sumatriptan group. Nevertheless, the proportion of patients who had a recurrence of their migraine within 24 h of the original attack was substantially smaller for the almotriptan groups compared with the sumatriptan group when the pain was severe. For these patients, the proportion of responders who had a recurrence was 8.8% for almotriptan 12.5 mg, 16.2% for almotriptan 25 mg, 28.9% for sumatriptan and 27.3% for placebo. The global recurrence rate of migraine for both almotriptan doses in patients with moderate and severe pain (18.0% and 15.4% for 12.5 and 25 mg doses, respectively) was lower than that reported previously for rizatriptan 10 mg (41–47%) (20), naratriptan 2.5 mg (27–28%) (21, 22), zolmitriptan 1–10 mg (26–36%) (23) and sumatriptan 100 mg (28–57%) (9, 24).
Both doses of almotriptan were well tolerated during the study, although the 12.5 mg dose was better tolerated than the 25 mg dose. Almotriptan 12.5 mg had a tolerability profile similar to that of placebo, whereas sumatriptan was associated with significantly more adverse events than placebo and almotriptan 12.5 mg (P<0.001 for both comparisons). There were no statistically significant differences between almotriptan 25 mg and sumatriptan (P=0.376) in the overall incidence of adverse events. No adverse event with almotriptan 12.5 mg occurred in more than 2% of patients. The most frequently reported adverse events in the almotriptan 25 mg group were dizziness (2.1%) and somnolence, (1.6%). The most common adverse events in the sumatriptan group were fatigue (5.7%), paraesthesia (3.1%), dizziness (2.1%) and somnolence (2.1%). The incidence of chest symptoms for the almotriptan 25 mg and sumatriptan groups was low but similar. However, these symptoms were not reported for the almotriptan 12.5 mg and placebo groups. The superior tolerability of almotriptan 12.5 mg vs. sumatriptan has been reported previously (16), and may be due to almotriptan's high specificity as a 5-HT1B/1D receptor agonist (11). Almotriptan has shown high and specific affinity for human 5-HT1B/1D receptors in binding studies. In addition, almotriptan has been shown to have high oral bioavailability (70%) (11), compared with sumatriptan where oral bioavailability has been quoted as 14% (9).
It appears therefore that almotriptan 12.5 mg provides effective relief for migraine, similar to that provided by the standard treatment (sumatriptan), but is better tolerated. Further comparative studies of the pharmacokinetics and tolerability of almotriptan and other triptans are required to reinforce the findings from this study.
In conclusion, almotriptan 12.5 mg provides effective and reliable relief from migraine attacks and is well tolerated. Almotriptan 12.5 mg is as effective as sumatriptan 100 mg in providing relief from migraine, and is significantly better tolerated.
Footnotes
Acknowledgements
This article was supported by funding from Laboratorios Almirall SA, the manufacturers of almotriptan. Drs Dowson, Massiou and Laínez have received funding from Laboratorios Almirall to perform studies.
