Abstract
The endogenous cannabinoid anandamide (AEA) plays important roles in modulating pain. Head pain is an almost universal human experience, yet primary headache disorders, such as migraine without aura (MoA) or episodic tension-type headache (ETTH), can represent a serious threat to well-being when frequent and disabling. We assessed the discriminating role of endocannabinoids among patients with ETTH or MoA, and control subjects. We measured the activity of AEA hydrolase and AEA transporter, and the level of cannabinoid receptors in peripheral platelets from MoA, ETTH and healthy controls. Sixty-nine headache patients and 36 controls were selected. Diagnosis of headache type was made according to the International Headache Society criteria. We observed significant sex differences concerning AEA membrane transporter and fatty acid amide hydrolase activity in all groups. An increase in the activity of AEA hydrolase and AEA transporter was found in female but not male migraineurs. Cannabinoid receptors were the same in all groups. Here we show that the endocannabinoid system in human platelets is altered in female but not male migraneurs. Our results suggest that in migraineur women an increased AEA degradation by platelets, and hence a reduced concentration of AEA in blood, might reduce the pain threshold and possibly explain the prevalence of migraine in women. The involvement of the endocannabinoid system in migraine is new and broadens our knowledge of this widespread and multifactorial disease.
Introduction
The role of endocannabinoids in the modulation of pain has come under intense investigation (1–3), but the role of endocannabinoids in primary headache disorders have not yet been investigated. Endogenous cannabinoid receptor agonists (‘endocannabinoids’) are recently identified lipid mediators thought to modulate central and peripheral neural functions, and to mimic several effects of cannabinoids (1–3). Anandamide [arachidonoylethanolamide (AEA)] and 2-arachidonoylglycerol are the most biologically active endocannabinoids, which bind to both central and peripheral cannabinoid (CB) receptors (1–3). The level of AEA in the extracellular space is controlled by cellular uptake via a specific AEA membrane transporter (AMT) (4), followed by intracellular degradation by the enzyme AEA hydrolase [fatty acid amide hydrolase (FAAH)] (5). Mice lacking FAAH have 15-fold higher levels of endogenous AEA and show reduced pain sensation compared with wild-type mice (6). More recently it has been suggested that central and peripheral endocannabinoids regulate discrete behavioural processes and may be targeted for distinct therapeutic gain (7). In particular, it has been proposed that not only central but also peripheral actions of endocannabinoids may regulate pain. Interestingly, anti-inflammatory peripheral effects of endocannabinoids may also diminish hyperalgesia (7, 8).
Recently, functional CB receptors, AMT and FAAH, i.e. the proteins forming the ‘endocannabinoid system’, have been shown in human platelets (9, 10). The release of inflammatory mediators by platelets at the site of tissue injury can modulate the development of afferent sensitization (11). Endocannabinoids also exert a critical control on cerebrovascular tone, by interacting with serotonergic transmission, nitric oxide (NO) production, and calcitonin gene-related peptide (CGRP) release (3). Since these factors are implicated in the pathophysiology of migraine (12), a dysfunction of the endocannabinoid system might also play a role in the development of migraine. An abnormal pain threshold is also implicated in the development of tension-type headache (13).
We have analysed possible anomalies of the endocannabinoid system in two groups of headache subjects [migraine without aura (MoA) or episodic tension-type headache (ETTH) patients], and compared them with healthy controls. In particular, we measured the activity of FAAH, as well as that of AMT and the level of CB receptors, in platelets isolated from our subjects.
We have previously shown that blood levels of AEA in women are regulated by FAAH
activity of blood cells (14). Since there is growing evidence for the influence of sex hormones
on endocannabinoids (15–17), we collected blood samples
from all females during the same menstrual phase
Subjects and methods
Headache assessment
Subjects eligible for the study were those aged 20–50 years describing a minimum of two but fewer than 10 headaches per month. Participants were required to maintain a daily diary during the study period, recording information on headache attacks and medication intake on a standard form. Diagnosis of headache type was made according to the 2004 International Headache Society criteria (20). Sixty-nine headache patients were selected from consenting women attending the Headache Centre in the Neurology out-patients department of the S. Eugenio Hospital. Thirty-six healthy subjects of medical and paramedical staff were also included as controls. Controls were carefully interviewed to exclude even very infrequent migraine. A structured medical history was utilized during a face-to-face interview by one neurologist specialist in headaches.
All subjects either had never been treated with any prophylactic drug or had discontinued treatment at least 6 months prior to enrolment. None of the subjects included in the present study was suffering from major psychiatric or medical illnesses, was under drug treatment or used any medication 72 h prior to collection of blood samples. Women included in the study had regular menstrual periods, were not pregnant at enrolment, and were not under oral contraceptive treatment.
Collection of blood samples
The study was conducted in a comfortable room, blood sample collection was performed by venepucture from the antecubital vein between 08.30 and 09.30 h. Washed platelets were prepared from blood as reported (9, 10). All women included in the present study were tested during the same menstrual phase, i.e. the late follicular phase. To check the menstrual phase and the venepuncture stress, serum concentrations of 17β-oestradiol, progesterone, follicle-stimulating hormone (FSH), luteinizing hormone (LH), and prolactin (PRL) were measured using a standard radioimmunoassay method. The reagents used in assays of PRL, FSH, and LH were obtained from DiaSorin (Saluggia, Italy), whereas 17β-oestradiol and progesterone were obtained from DPC (Medical Systems, Genoa, Italy). The interval between the last headache attack and the blood sample collection was at least 3 days. All subjects gave their informed consent and the study was approved by the local Ethics Committee.
Biochemical assays
The uptake of 3H-AEA (223 Ci/mmol, from NEN DuPont de Nemours, Köln, Germany) by the selective AMT of intact platelets was measured as reported (10, 14). The hydrolysis of 3H-AEA by the FAAH (E.C. 3.5.1.4) activity was assayed in platelet extracts by reversed-phase high-performance liquid chromatography (10, 14). The binding of 3H-CP55,940 (126 Ci/mmol, from NEN DuPont de Nemours) to platelet membrane fractions was determined by rapid filtration assays, as described (10, 14).
Statistical analysis
The main objective of this study was to assess the discriminating role of endocannabinoids among patients with ETTH or MoA, and control subjects, taking into account possible differences in terms of demographic characteristics, smoking habits, hormonal status, headache frequency, headache history duration and symptomatic drug intake. The relationship between AMT, FAAH, CB receptors and groups was investigated by one-way analysis of variance. Generalized linear model was performed to better understand the influence of age, smoking status, concentration of PRL, FSH, LH, 17β-oestradiol or progesterone on AMT, FAAH, and CB receptors. Finally, multiple regression analysis allowed us to determine which variables could be considered as independent factors.
Statistical analysis was performed with SPSS 11.0 statistical package (SPSS Inc., Chicago, IL, USA).
Results
Patients’ characteristics are shown in Table 1. No significant differences
were noted in age or smoking habit among the groups
Characteristics of subjects suffering from migraine without aura (MoA) or episodic tension-type headache (ETTH), and of healthy controls
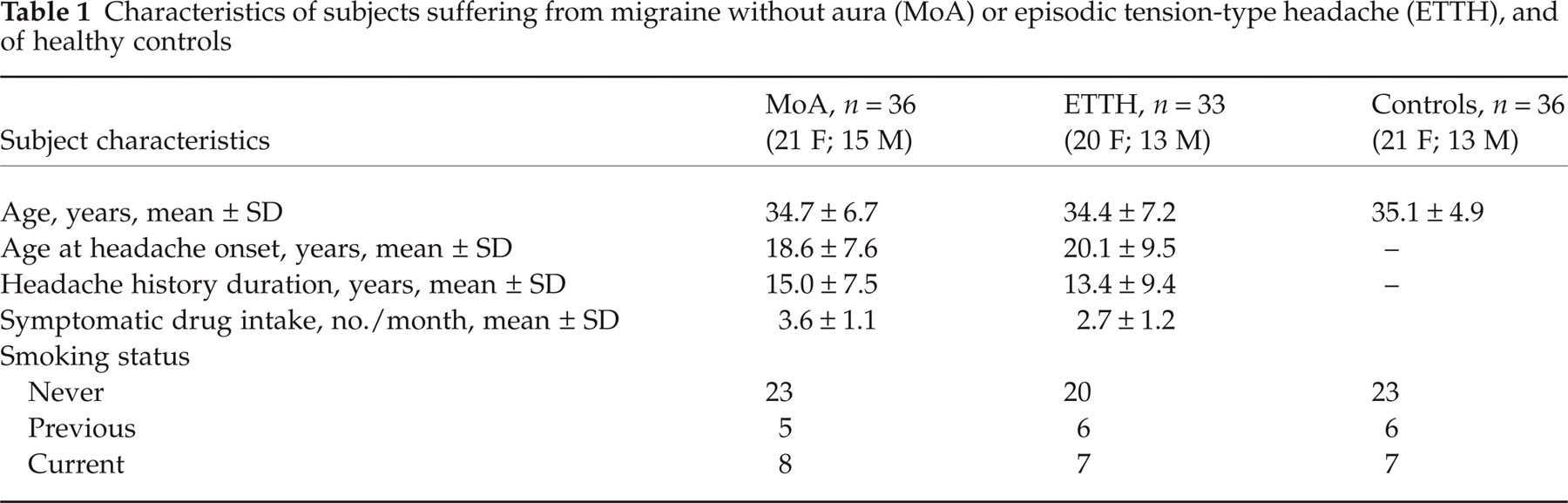

Bar graphs showing the activity of arachidonoylethanolamide (AEA) membrane
transporter (AMT) and of fatty acid amide hydrolase (FAAH) in platelets
isolated from subjects suffering from migraine without aura (MoA) or
episodic tension-type headache (ETTH), and from healthy controls.
▪, Females; □, males. AMT and FAAH activity (both
expressed as pmol/min per mg protein) were assayed using 400
n
Headache frequency and history duration, and extent of symptomatic drug intake did not significantly differ between episodic ETTH and MoA. The three female groups did not differ significantly in terms of serum concentrations of 17β-oestradiol, progesterone, FSH, LH or PRL (data not shown).
The multiple regression analysis performed separately on each test group of females showed that in the control group, but not in ETTH and MoA, there was a positive relationship between progesterone and both AMT and FAAH, and a negative relationship between LH, FSH, and both AMT and FAAH. Moreover, the generalized linear model showed that progesterone is a powerful covariate of both FAAH and AMT, extending to platelets our previous observation of a strong relationship between progesterone and FAAH in human lymphocytes (15). Even after adjustment for hormonal levels, FAAH and AMT activities were variables independently associated with migraine. In addition, multiple regression analyses revealed that in the ETTH group, unlike controls, AMT and FAAH levels are independent of hormone levels.
Discussion
In the present study we show that the endocannabinoid system in human platelets is altered in female but not male migraineurs. Human platelets share several receptors and transduction signalling pathways with neuronal cells (21, 22) and they have CB receptors and the biochemical machinery to degrade AEA (9, 10).
In these cells we found an increase in the activity of AEA hydrolase and AEA transporter in female but not male migraineurs. Conversely, we did not find significant differences in CB receptor levels in platelets among groups. Our clinical findings suggest a relationship between endocannabinoids and migraine. This relationship has been already suggested by the experimental data of Goadsby's group. In fact, anandamide has been shown to inhibit trigeminovascular neurons and prevent vasodilation (23, 24).
Recent advances into the function of endogenous cannabinoids have provided important insights into the mechanism of antinociception (1–3). Endogenous AEA plays an important role in the cannabinergic pain suppression system existing within the dorsal and lateral periaqueductal grey (PAG) (25). Indeed, the PAG has a recognized role in pain modulation (26). In addition, the brainstem and the striatum contain the highest amounts of AEA within the central nervous system (27). In this regard, brainstem activation has been observed by positron emission tomography analysis in the periacqueductal grey matter, dorsal raphe nucleus and locus coeruleus, during spontaneous attacks of migraine in patients with MoA (28). This finding strengthens the hypothesis that the pathogenesis of migraine is related to an imbalance between the activity of brainstem nuclei regulating antinoception and vascular control (28).
Circulating blood levels of AEA were not directly measured in this study, but an inverse relationship between FAAH activity and AEA levels in blood has been demonstrated previously (14). As we did not find significant differences in CB receptor levels in the platelets of the three groups of subjects, the increased activity of FAAH and AMT may represent an imbalance of endocannabinoid degradation. Accordingly, it is interesting to note that the genetic ablation of FAAH dramatically increases AEA levels and increases nociceptive thresholds in mouse models of pain (6).
Some notes of caution are due in the interpretation of these data. It is not known whether the apparent association between female migraineurs and peripheral levels of AMT and FAAH is causative, as it is also unclear whether gender differences in AMT and FAAH have a direct relationship to the known gender differences in pain perception (29). In addition, it is also important to note that peripheral and central endocannabinoids are inactivated by distinct FAAH genes (7), so that we cannot draw any conclusions about the central regulation of endocannabinoids from this study. However, it is possible that peripheral inflammation related to endocannabinoids may also play a role in the observed effects on pain (7). As previous findings favour a role for FAAH in regulating pain perception by modulating endogenous cannabinoid tone (2), these findings may open the way for FAAH inhibitors as novel pain-killing drugs.
In this study we tested a relatively small number of subjects. However, it must be stressed that our population was highly selected. In fact, all subjects were studied outside of headache attacks and divorced from analgesic drug intake, which could influence endocannabinoid levels. In addition, none of the subjects included had any medical or major psychiatric comorbidity. Furthermore, we followed severe inclusion criteria concerning the sex hormonal status.
In conclusion, while the endocannabinoid system does not appear to be causative of either MoA or ETTH, it may act as a sensitizer making women more sensitive to ache.
Footnotes
Acknowledgements
The authors thank the patients who took part in this study and the nurses Maria Petroni and Stefania Tomassini for assisting with blood sample collection. We also thank Dr Emanuele Fabrizi for his expert assistance with statistical analysis.
