Abstract
Cytokines have been measured in cerebrospinal fluid (CSF) from headache patients [infrequent episodic tension-type headache (TTH) and migraine with or without aura, all during attack, and cervicogenic headache] and compared with levels in pain-free individuals. Both proinflammatory [interleukin (IL)-1β, tumour necrosis factor-α and monocyte chemoattractant protein-1 (MCP-1)] and anti-inflammatory cytokines IL-1 receptor antagonist (IL-1ra), IL-4, IL-10 and transforming growth factor-β1 (TGF-β1)] were included. There were significant group differences in IL-1ra, TGF-β1 and MCP-1 in episodic TTH and migraine compared with controls, and a significant difference in MCP-1 between cervicogenic headache and migraine with aura. Intrathecal MCP-1 correlated with IL-1ra, IL-10 and TGF-β1 in episodic TTH, and MCP-1 with IL-10 in migraine with aura. Cytokine increases were modest compared with those often accompanying serious neurological conditions, and may represent a mild response to pain. We believe this to be the first comparative study of CSF cytokine levels in connection with headache.
Introduction
Several studies have shown that it is difficult to distinguish between subarachnoid haemorrhage (SAH) and severe acute, benign headache attacks if based only on the clinical presentation (1, 2). A large proportion of these benign attacks are primary headaches such as migraine and episodic tension-type headache (TTH), frequent disorders with a number of symptoms in common, possibly sharing some common pathophysiology (3) and sometimes difficult to distinguish (4–6).
Migraine is often triggered by stress factors (7, 8), and migraine pain is believed to result from neuronal nociceptive activity in the trigeminal vascular system, whereby neuropeptides released from trigeminal fibres, putatively within the meningeal vasculature, induce sterile neurogenic inflammation (9, 10). Although these effects are likely to be local, changes in cytokines (pro- or anti-inflammatory) causing or resulting from headache pathophysiology might be detectable in cerebrospinal fluid (CSF). Intrathecal levels of such substances are thought to reflect autochthonous production in the presence of an intact blood–brain barrier (11).
A number of studies have examined cytokine levels in blood in connection with headache, although the majority concern migraine during or outside attack. The proinflammatory cytokines interleukin (IL)-1β and tumour necrosis factor (TNF)-α have been investigated most frequently, with attack levels being found to be elevated (12, 13) or unchanged (14, 15). These cytokines have also been reported to be increased in the serum of patients with cervicogenic headache relative to migraineurs during attack or to controls (16). The anti-inflammatory cytokines IL-4 and IL-10 have also been studied in migraine, with several reports indicating a reduced level of IL-4 during an attack (13, 17, 18), whereas IL-10 has been found to increase during an attack (12, 18). Significantly increased plasma transforming growth factor (TGF)-β1 has been found in migraine during the headache-free period, but not TTH, compared with controls (19). A meta-analysis of the literature concerning immunological aspects of migraine from 2001 concluded that there was no clear-cut evidence of immune dysfunction in migraineurs, but that altered immune function could not be totally excluded (20).
By comparison, very little has been reported in the literature concerning cytokine levels in CSF in connection with common headaches, even though it is a medium more likely to reflect cytokine changes in the nervous system. Although ethical considerations limit lumbar puncture, CSF is sometimes obtained from headache patients when serious neurological conditions are initially suspected. Ironically, the little that is published about intrathecal levels of cytokines during TTH and other headaches is as a reference group for conditions such as meningitis, stroke, amyotrophic lateral sclerosis or multiple sclerosis (21–24). All of these can be accompanied by substantially increased CSF levels of pro- and anti-inflammatory cytokines, whereas patients with a common headache are expected to have CSF cytokine levels closer to, or within, the physiological range. To our knowledge, no reference values exist for CSF cytokine levels in healthy individuals.
In the present study, cytokines were studied in CSF from patients admitted to hospital in order to rule out or verify SAH. They had acute headache, thereby being in an ictal phase, and a lumbar puncture was carried out as part of the clinical investigation. Comparable patterns of cytokines in the CSF might be expected if migraine and episodic TTH share a common pathophysiology. The cytokines chosen were representative of proinflammatory cytokines [TNF-α, IL-1β and monocyte chemoattractant protein (MCP)-1], as well as several with predominantly immunosuppressive properties [IL-1 receptor antagonist (IL-1ra), IL-4, IL-10, and TGF-β1] (25). For comparison, cytokines were also measured in CSF from two other groups: patients with cervicogenic headache, a secondary headache attributed to a disorder in the neck sharing many clinical features with both migraine and TTH, prior to radiofrequency treatment; and pain-free patients who had been given a spinal puncture to exclude serious neurological conditions, but where none had subsequently been found.
Patients and methods
Patients
The CSF samples were obtained from two Norwegian hospitals. Most patients in the present study (n = 97) were part of a larger group of 433 patients who had arrived at hospital with acute headache (referred by general practitioners or taken directly to hospital by relatives or by ambulance), described as their first or worst headache ever, reaching maximum pain within seconds to hours, without known cause and with clinical features suspicious of SAH.
Emergency Departments are not separate entities in Norway, so these patients were examined directly by doctors from the Neurology Department. All patients were given a full clinical and neurological examination. Blood tests, electrocardiograms and cerebral computed tomography (CT) scans were taken without delay. As part of the diagnostic procedure to rule out or verify SAH, the patients underwent lumbar puncture if the initial cerebral CT was normal or inconclusive. CSF was analysed for cell count, levels of protein and glucose, spectrophotometry to detect bilirubin and oxyhaemoglobin, and a selection of cytokines.
The patients' headache attacks were diagnosed retrospectively, separately by two neurologists reviewing the same case histories, using as guidelines the International Classification of Headache Disorders (26). Ultimately, 30 different conditions were decided as underlying the acute headache attacks in the group of 433 patients. The first 166 of these samples were analysed for a series of cytokines. In this smaller material, only three patients were subsequently diagnosed with SAH (because if bleeding was evident by CT then no lumbar puncture was made), whereas 123 were diagnosed as having attacks of a primary headache, of whom 97 were diagnosed with either infrequent episodic TTH or migraine (with or without aura). Other primary headache groups were too small to warrant statistical analysis. Diagnoses were based on the dominating symptoms and features in the clinical presentation and how the symptoms progressed. Most patients (70%) did not have a previously diagnosed headache condition.
Patients with cervicogenic headache (n = 10) were recruited at an out-patient neurology and headache clinic and were diagnosed according to criteria described by the Cervicogenic Headache International Study Group (27). They were lumbar punctured after inclusion in a study of the effect of radiofrequency denervation (28). Blockade of the greater occipital nerve relieved pain in all these patients, and they did not have other forms of headache.
CSF samples from 20 patients who had been lumbar punctured to eliminate serious neurological conditions, but where none had been subsequently found, symptoms at referral including paraesthesias, numbness, unspecific vertigo, anxiety or stress, were used as controls. The patients were considered to be pain free, as none of them had any pain or used any drugs at the time of puncture, and there was no mention of any painful condition in their medical records. The age of the groups did not vary significantly except between the control and cervicogenic headache groups.
Diagnosis of the patients and analysis of the samples were carried out by separate collaborators, and the diagnostic code was broken only after all analyses were completed. The study was approved by the Regional Committee for Medical Research Ethics. Demographic and routine CSF data are shown in Table 1.
Demographic and routine cerebrospinal fluid (CSF) data

Results are expressed as the mean ±
∗ P < 0.0005;
† P < 0.05 (Kruskal–Wallis);
‡significantly different from control group, P < 0.01 (Mann–Whitney).
§Only one patient with pleocytosis.
Lumbar puncture
The patients admitted to hospital because of acute headache were lumbar punctured when 12 h had elapsed from headache onset. For a proportion of the patients the period from headache onset to lumbar puncture was > 12 h, due to delayed referral to the hospital emergency room. Lumbar puncture was carried out with patients lying on their side in a horizontal position, at the level of the lower lumbar spinous processes, mostly L4/L5. Patients with accidental bleeding during the procedure were excluded. CSF samples were obtained immediately after the lumbar puncture, placed in ice-water and stored at −80 °C. Cell number, protein and glucose were analysed routinely by the respective University Hospital laboratories (Table 1).
Cytokine analysis
CSF samples were thawed in an ice-bath and analysed for IL-1β, IL-1ra, IL-4, IL-10, TNF-α, MCP-1 and TGF-β1 (total). No sample had more than one freeze–thaw cycle. Cytokines were assayed in duplicate in undiluted CSF by specific enzyme-linked immunosorbent assay kits (R&D Systems Europe, Abingdon, UK) according to the manufacturer's instructions. In the case of TGF-β1, CSF was acidified by the addition of 60 μl 1 N HCl to 300 μl CSF, incubated at room temperature for 10 min and then neutralized by the addition of 60 μl 1.2 N NaOH in 0.5 M HEPES buffer before use. Cytokines were measured colorimetrically on a microplate reader (Multiscan Spectrum; Thermo Labsystems, Helsinki, Finland). Lower detection levels for the cytokines were: IL-1β, 0.1 pg/ml; IL-1ra, 16 pg/ml; TNF-α 0.32 pg/ml; IL-4, 0.13 pg/ml; IL-10, 0.2 pg/ml; MCP-1, 5 pg/ml; TGF-β1, 7 pg/ml. Peripheral blood samples were not analysed with respect to cytokines. Results of cytokine analysis from the minor diagnostic groups in our material are not reported here.
Statistical analysis
Not all the data from the cytokine assays were normally distributed, and statistical analysis was therefore performed with non-parametric methods using the programme
Results
Of the seven cytokines investigated, measurable amounts of only three, IL-1ra, MCP-1 and TGF-β1, were found in the CSF from all patients (Figs. 1–3). The Kruskal–Wallis test indicated significant differences between the groups in the levels of all these cytokines. The main differences were found between the group of pain-free control individuals, and patients with episodic TTH, and migraine without aura. In both these headache groups, IL-1ra, MCP-1 and TGF-β1 were all significantly elevated compared with corresponding levels in the control group. In the case of migraine with aura, there were significant differences in the levels of IL-1ra and MCP-1, but no significant difference in the level of TGF-β1. There were no significant differences between the control and cervicogenic headache groups. No other significant differences in cytokine levels were found between the headache groups, except in the level of MCP-1 between the cervicogenic headache and migraine with aura groups (P = 0.007). Similarly, trends towards a difference in the level of MCP-1 between the cervicogenic headache and migraine without aura (P = 0.031) or episodic TTH (P = 0.044) groups were found. IL-10 was detected in most patients, but there was only a trend towards a difference between the groups (Fig. 4).

Distribution of interleukin-1 receptor antagonist (IL-1ra) levels in cerebrospinal fluid from patients without headache (n = 20), or with episodic tension-type headache (TTH, n = 39), migraine without aura (n = 34), migraine with aura (n = 24) or cervicogenic headache (n = 10). Lines indicate the median of the group. There are significant differences between the groups, P < 0.0005 (Kruskal–Wallis); Mann–Whitney indicates significant differences between the pain-free control group and groups of patients with episodic TTH (P < 0.0005), migraine without (P < 0.0005) or with (P = 0.001) aura (∗).
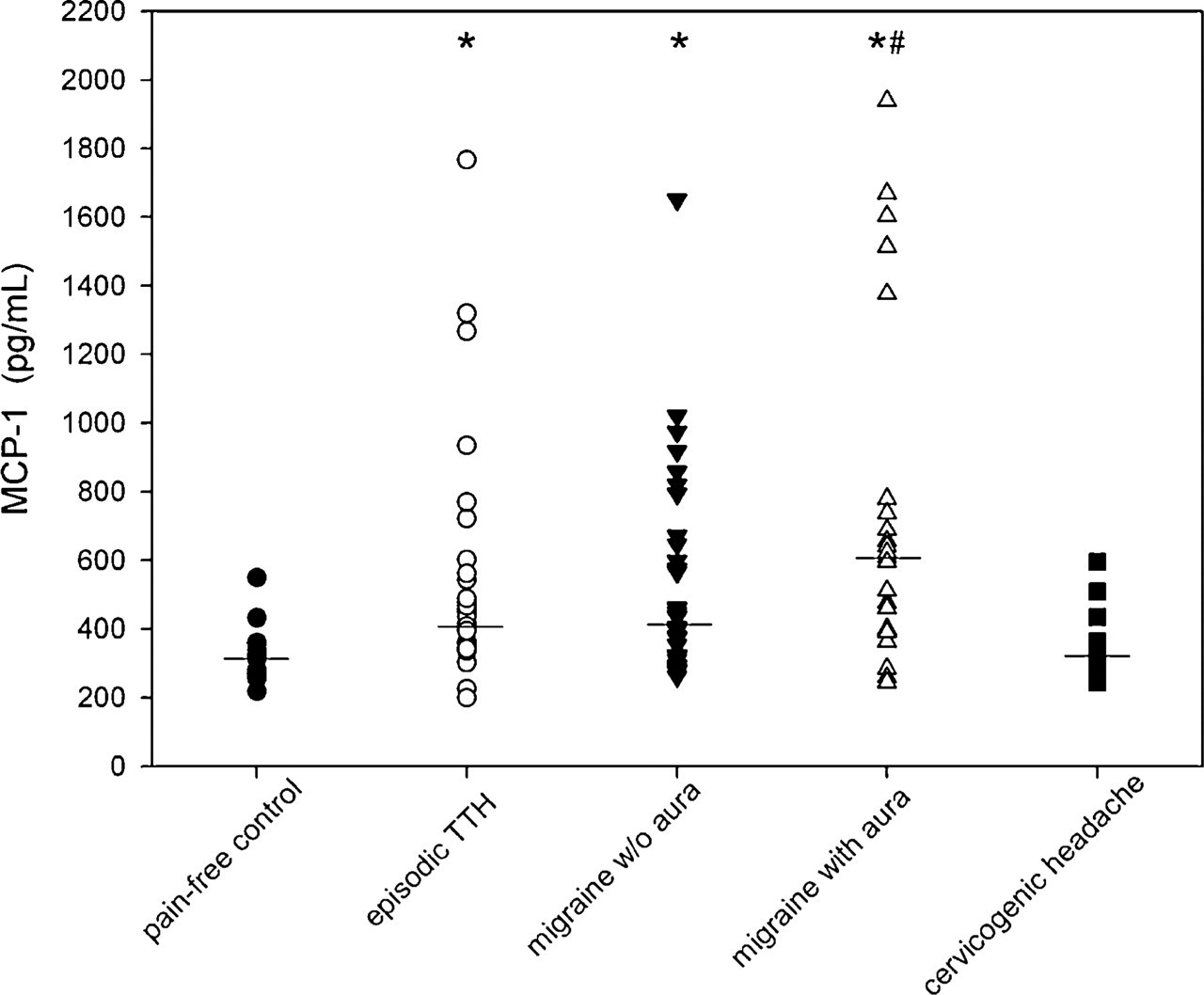
Distribution of monocyte chemoattractant protein (MCP)-1 levels in cerebrospinal fluid from patients without headache (n = 20), or with episodic tension-type headache (TTH, n = 39), migraine without aura (n = 34), migraine with aura (n = 24) or cervicogenic headache (n = 10). Lines indicate the median of the group. There are significant differences between the groups, P < 0.0005 (Kruskal–Wallis); Mann–Whitney indicates significant differences between the pain-free control group and groups of patients with episodic TTH (P < 0.0005), migraine without (P < 0.0005) or with (P < 0.0005) aura (∗), as well as between the cervicogenic headache and migraine with aura groups (#P = 0.007).

Distribution of total transforming growth factor (TGF)-β1 levels in cerebrospinal fluid from patients without headache (n = 20), or with episodic tension-type headache (TTH, n = 39), migraine without aura (n = 34), migraine with aura (n = 24) or cervicogenic headache (n = 10). Lines indicate the median of the group. There are significant differences between the groups, P = 0.003 (Kruskal–Wallis); Mann–Whitney indicates significant differences between the pain-free control group and episodic TTH (P = 0.002) and migraine without aura (P = 0.002) (∗).

Distribution of interleukin-10 (IL-10) levels in cerebrospinal fluid from patients without headache (n = 19), or with episodic tension-type headache (TTH, n = 33), migraine without aura (n = 27), migraine with aura (n = 18) or cervicogenic headache (n = 9). The number of samples with values below the level of detection is designated for each group by ‘x’. Lines indicate the median of the group. There is only a trend towards a difference between the groups, P = 0.063 (Kruskal–Wallis).
There were small, but significant increases in total protein in the episodic TTH and both migraine groups compared with the control group (Table 1). No control individuals had a CSF protein level outside the physiological range, whereas 10% cervicogenic headache, 36% episodic TTH, 38% migraine without aura and 13% migraine with aura patients had elevated CSF protein. Correlations are shown in Table 2. The clearest pattern was found in episodic TTH, where MCP-1 correlated significantly with IL-1ra, TGF-β1 and IL-10. Furthermore, there was a significant correlation between total protein and TGF-β1, as well as a trend between IL-1ra and TGF-β1 in the episodic TTH group.
Comparison of correlations between proteins

Significance P < 0.01, trend P < 0.05.
Kendall's τ.
NS, not significant; IL-1ra, interleukin-1 receptor antagonist; MCP-1, monocyte chemoattractant protein-1; TGF-β1, transforming growth factor-β1; TTH, tension-type headache.
In the migraine without aura group there were significant correlations between total protein and IL-1ra, or TGF-β1, as well as a trend between IL-1ra and TGF-β1. For the migraine with aura group, MCP-1 correlated significantly with IL-10, and there were trends for correlations between MCP-1 and IL-1ra, and TGF-β1 and IL-10. In the pain-free control and cervicogenic headache groups no significant correlations were found (Table 2). For all the results with these cytokines, no pattern emerged when specific analysis of the outliers was carried out.
IL-1β was detected only rarely in CSF in this study: five of 37 patients with episodic TTH (range 0.18–0.57 pg/ml), six of 33 patients with migraine without aura (range 0.40–6.71 pg/ml) and four of 23 patients with migraine with aura (range 0.14–0.35 pg/ml). It was not detected in any sample in the pain-free control or cervicogenic headache groups. TNF-α was detected only in low levels in a few samples: one of 20 controls (0.52 pg/ml), two of 10 patients with cervicogenic headache (0.49 and 0.51 pg/ml), four of 30 patients with episodic TTH (range 0.32–0.46 pg/ml), three of 25 patients with migraine without aura (range 0.35–0.50 pg/ml) and seven of 20 patients with migraine with aura (0.36–0.84 pg/ml). Apart from the one patient in the migraine without aura group with comparatively high IL-1β (and who was unremarkable for all other cytokines, including TNF-α), none of these values can be considered high. Measurable amounts of IL-4 were not found in any CSF sample in any group.
Discussion
It is often difficult to distinguish between migraine and episodic TTH, perhaps more so when the onset is acute and the headache severe. Some authors contend that the two disorders represent escalating steps in a common pathophysiological process, and point out that they share many clinical features as well as trigger factors (3, 6). They may even benefit from the same treatment. In this light, investigating CSF from both migraine and episodic TTH patients is highly relevant, and the opportunity was unique as all patients in these particular groups were lumbar punctured in order to rule out SAH. The patients with episodic TTH or migraine were all diagnosed following clinical elimination of secondary causes of headache. For most of the patients this was their first severe headache attack. According to the International Headache Society criteria, episodic TTH pain should be mild to moderate, although as only two pain criteria have to be met, this is not an absolute requisite. Indeed, severe pain during some attacks has been reported by 13% of patients diagnosed with episodic TTH in a large epidemiological study (30). Data from a more recent treatment study also suggest that episodic TTH patients can sometimes have particularly severe attacks (31).
The results indicated that there are significant differences in the CSF levels of certain cytokines (IL-1ra, MCP-1 and TGF-β1) in migraine and episodic TTH compared with pain-free controls, and these changes were not seen in cervicogenic headache. However, in the case of cervicogenic headache, CSF samples were drawn in connection with a project and the pain was not necessarily particularly severe at the time. Samples from the other headache patients were taken during an acute phase, the patients having their first or worst headache episode. It cannot be excluded that the cytokine changes found are as much a response to pain intensity as to headache type, or relate to acute vs. chronic headache, and may therefore explain the cytokine levels in cervicogenic headache patients being more similar to the controls, rather than constituting a real difference in pathophysiology between cervicogenic headache and episodic TTH or migraine. However, we found no pattern among the outliers to suggest that those with the highest cytokine levels also had the greatest pain.
In fact, no significant differences in cytokine levels were found between the migraine groups (with or without aura) and episodic TTH. However, it is interesting that correlation of the cytokines themselves showed that MCP-1 (proinflammatory) correlated significantly with IL-1ra, TGF-β1 and IL-10 (all anti-inflammatory) in episodic TTH. This pattern was not found in migraine or cervicogenic headache. Other correlations found were more random and may be accidental, or reflect the limited size of some of the groups. IL-1ra, TGF-β1 and IL-10 are largely immunosuppressive and potent deactivators of macrophages (32), whereas MCP-1 is a potent monocyte chemoattractant (33). The fact that changes to both pro- and anti-inflammatory cytokines were found suggests that these cytokines assist in restoring homeostasis (34). In this context, it is interesting that the positive correlation coefficients found for the relationship between proinflammatory MCP-1, and the anti-inflammatory IL-1ra, TGF-β1 and IL-10 in episodic TTH are consistent with a balance between these biologically opposing capacities, and MCP-1 may have particular relevance for episodic TTH. That such a pattern was not found for migraine or cervicogenic headache may simply be the result of such balances in these headaches being more dependent on other cytokines not measured in this study.
The increased levels of IL-1ra, MCP-1 and TGF-β1 in the migraine and episodic TTH groups compared with the control group were statistically significant, although modest compared with many cytokine levels induced by more serious neurological conditions. Moreover, the cytokines most often associated with inflammatory conditions, IL-1β and TNF-α were not increased in any condition in this study. By comparison, in a group of six patients with lymphocytic meningitis we found maximum levels of intrathecal IL-1ra, MCP-1, TGF-β1 and IL-10 to be respectively 4732, 3227, 180 and 97 pg/ml. IL-1β, TNF-α and IL-4 were respectively 4.07, 6.65 and 0.32 pg/ml (unpublished results). Several of these levels are substantially greater than those found in headache patients in the present study, despite the pain of the episodic and migraine attacks being severe. The increases in CSF protein concentration found in some headache patients suggests that the blood–brain barrier could be slightly compromised, and cytokine increases could be the result of leakage over the barrier from plasma. However, there was no clear general pattern of cytokine levels correlating with the protein concentration in the headache groups, so it seems likely that the response of these cytokines to these benign primary headaches is modest, even when attacks are severe. Such modest increases in these cytokines during severe attacks suggest that changes would be slight during attacks of normal intensity. The cytokine level changes found in the present study may even lie within the physiological range (which as far as we know has yet to be defined), at least for many patients.
Indeed, although the physiological function of cytokines in brain is neuromodulatory as well as immunological, for the preservation or restoration of homeostasis (35), it is known that even small changes in their level in brain (which might not be measurable in CSF) can sometimes result in profound systemic reactions and peripheral cytokine synthesis (34). This may account for the rather larger changes observed for some cytokines in serum previously reported in a number of studies, mainly in connection with migraine (see reviews (12, 20)), including IL-1β and TNFα which were not detected in CSF in the present study. They may be associated with symptoms of migraine other than headache. It is also possible that factors like cytokines could trigger the trigeminovascular system, considered to be part of the brain's defence (9, 10). In triggering the release of vasoactive and nociceptive neuropeptides, cytokines could contribute to headache development. Equally likely is the possibility that they may be released as a result of trigeminovascular activation. However, if the changes presented here are related to neurogenic inflammation, the reaction clearly does not lead to marked intrathecal increases of proinflammatory cytokines (although of course local levels in the dura may be marked).
Although there were significant increases in IL-1ra, MCP-1 and TGF-β1 in CSF of patients with episodic TTH and migraine with or without aura, compared with pain-free individuals, the increases were not sufficiently different between these headache types to distinguish them from each other. Rather, the results indicate that these changes seem to be mutual for these headaches, thereby supporting the theory that they have certain (patho)physiological processes in common. As far as we are aware, this is the first systematic attempt to measure the levels of several pro- and anti-inflammatory cytokines in CSF from patients with episodic TTH, migraine or cervicogenic headache, compared with levels in pain-free individuals.
Footnotes
Acknowledgements
The authors thank Sylvia Nome Kvam and Gøril Gravdahl for valuable technical assistance.
