Abstract
This cross-sectional clinical study was conducted in order to explore the relationship between atopic disorders and migraine. We evaluated 186 consecutive patients with migraine. Patients with a history of atopic disorders were compared with the others during headache-free intervals, for their headache characteristics, pulmonary test (PFT) performances and immunological screenings, through appropriate statistical methods. Of the patients with migraine, 77 (41.4%) reported at least one atopic disorder. PFT screening showed a general decreased pulmonary capacity and an important correlation between a positive history of atopic disorders and both increased eosinophil and IgE levels in headache-free periods. It should be discussed whether screening with PFT or immunological tests helps in early detection of progressive lung disease which might develop in these patients.
Introduction
Migraine is a common disorder characterized by severe headache accompanied by autonomic and neurological symptoms. A wide range of underlying mechanisms for migraine have been postulated so far, including ‘sterile neurogenic inflammation’, defects in arachidonic acid or serotonin metabolism, cyclical changes in ovarian steroid concentrations, food allergy and atopy (1–7). Multiple threads of research over the last 15 years have led to the concept that migraine is generated from a hyperexitable brain (8, 9).
Asthma has been described as ‘pulmonary migraine’ or ‘acephalic migraine’ (10, 11). Case reports and a few population-based studies have pointed out an association between migraine and asthma (12, 13). The relationship between migraine and atopy has also been the focus of historical interest (14) but reports about this issue have been conflicting. Comorbidity of migraine and atopic diseases such as eczema and asthma is an important argument for a suspected immune system dysfunction in migraineurs. In 1985, Nelson reviewed this issue and found evidence of a close association between migraine and atopic diseases (7). Recent studies have confirmed this association. One study, examining more than 1000 children, found a significantly increased prevalence of migraine in children with atopic diseases (15). Another study reported that the prevalence of asthma was about twice as high in children of mothers with migraine but without asthma or allergies (13). Both migraine and allergy appear in a paroxysmal and recurrent fashion and a hypersensitive response would fit well with both the duration of the migraine attack and the fact that selective foods can precipitate migraine. Based on these similarities, it is understandable that migraine is already linked to a hypersensitive immune system (16).
Although there exists some controversial evidence (17), asthma has generally been found to be associated with migraine (18). European surveys indicate an apparent rise in the prevalence of headache (especially childhood), prominently migraine subtype, asthma and wheezing, rhinitis and eczema. The reason for the apparent increase in the prevalence of headache and atopy remains unclear (15). Basically originating from some suggestive observations and subsequent clinical studies, an important report supported that montelukast, an antagonist of the cysteinyl leukotriene receptor, used in the treatment of asthma, showed important beneficial effect in the prophylaxis of migraine attacks and these results suggested pathophysiological links between migraine and asthma in some respects (19).
The results of some studies suggest a link between migraine (vascular reactivity) and asthma (bronchial reactivity) that is independent of allergic mechanisms. A shared functional abnormality of smooth muscle in blood vessels and airways offers a plausible explanation for this relationship (1) in the latest research. However, the pulmonary function test (PFT) results and immunological abnormalities in patients with migraine during headache-free intervals, which might be an indicator of pulmonary prognosis of migraineurs, are not clear. In order to determine relationships between the history of atopic disorder (including asthma) and migraine, we performed this study using a questionnaire for family history of these disorders, and administering PFTs and immunological tests.
Patients and methods
Among the patients followed by the Headache Out-patient Department of Mersin University Hospital, Mersin, Turkey, 186 consecutive patients (average age 37.7 ± 8.7 years, 9.6% male and 90.4% female) with migraine were included in this cross-sectional clinical study. Thirty age- and sex-matched patients with chronic tension-type headache (CTTH) were included as the control group. All subjects gave written informed consent to the study protocol and detailed explanations were made according to the Helsinki declaration. Diagnoses of migraine and CTTH were made by revised International Headache Society (IHS) criteria (20) and details were obtained by headache diary, used as a routine procedure of our headache out-patient department. In this diary, we asked the patients to note if they had headache or not each day. If they had headache, they were asked to write the time that the headache had started, its duration and intensity and the accompanying signs. The patients diagnosed as chronic migraine were not included in this study. Additionally, all patients were evaluated with a detailed structured questionnaire for allergy and asthma profile, including the presence of doctor-diagnosed allergic diseases and migraine in family members. Among atopic disorders, asthma, rhinitis, conjunctivitis, seasonal allergy, food allergy and drug allergy were taken into consideration. The asthma diagnosis was made clinically as described in the following section. Diagnosis of atopic disorders was made by a structured questionnaire. Allergic rhinitis was described as the presence of such symptoms as itching in the nose, nasal congestion, rhinorrhoea, which are characterized by an episodic course. The diagnosis of allergic conjunctivitis was based on episodic symptoms of ocular redness, itching and watering that were not explained otherwise. Signs of rhinoconjunctivitis limited to pollen season and remitting spontaneously were considered in favour of seasonal allergic rhinoconjunctivitis. Finally, a diagnosis of food and drug allergy was made when there was a history of itching, shortness of breath and rash upon exposure to identified agents.
All patients were examined by the same chest disease specialist and required investigations were carried out. The hyper-responsiveness, defined as bronchoconstriction following exercise and acute decrease in airway irritability after administration of a β agonist [forced expiratory volume in 1 s (FEV1) increased by >12% in the reversibility test], was accepted as an objective indicator of asthma. Chest X-ray and pulmonary function tests were performed in all subjects. Standardized pulmonary function tests were performed using a dry spirometer device (Sensor Medics MPM, Yorba Linda, CA, USA). We calculated the FEV1, the forced vital capacity (FVC), and FEV1/FVC ratio from maximal expiratory manoeuvres. Data were expressed as a percentage of the predicted standardized values according to European Respiratory Society (ERS) criteria (21). Acceptable recordings were obtained from each manoeuvre and the highest values were used for further analysis (22, 23).
Blood samples(3 ml) were drawn from a cubital fossa vein in headache-free periods and at least 48 h relief from pain during which no special prophylactic management was provided in 92 migraine sufferers [80 (86.9%) were female and 12 (13.1%) were male]. Patients were fasting except for taking their medication 12 h before sampling. Serum was separated by centrifugation at 4000
In statistical analysis data were first evaluated using descriptive statistics. Patients with a history of atopic disorders were compared with others with the PFT and immunological screen abnormalities, through appropriate statistical methods. Factors related to having atopic disorders were investigated by regression analysis. Differences of clinical, spirometric and immunological variables in patients with or without any atopic disorders were assessed by parametric and non-parametric statistics, as appropriate. Pearson and Spearman correlation analysis was made between clinical, spirometric and immunological parameters in the patient group, as appropriate. In order to investigate the effect of clinical, spirometric and immunological variables on having an atopic disorder, binary logistic regression analysis was also performed. Differences lower than 0.05% were accepted as significant.
Results
Of 186 patients with migraine only 18 (9.6%) were male and the rest were female (90.4%). The mean age was 37.7 ± 8.7 years (range 18–46 years) and the mean time from the headache diagnosis was 123.2 ± 98.0 months (range 8–260 months). Patients described a history of travel sickness (21.5%), abdominal pain attacks (11.2%), various operations (i.e. appendectomy, gynaecological operations and orthopaedic operations) and smoking (31.1% past smokers and 26.3% current smokers) in their past medical histories. Seventy-seven (41.4%) patients showed a history of atopic disorders including asthma (see Table 1) not fully dependent on gender. As shown in Table 2, detailed headache details of the patients did not show any distinctive headache characteristics.
The reported frequency of atopic disorders in patients with migraine
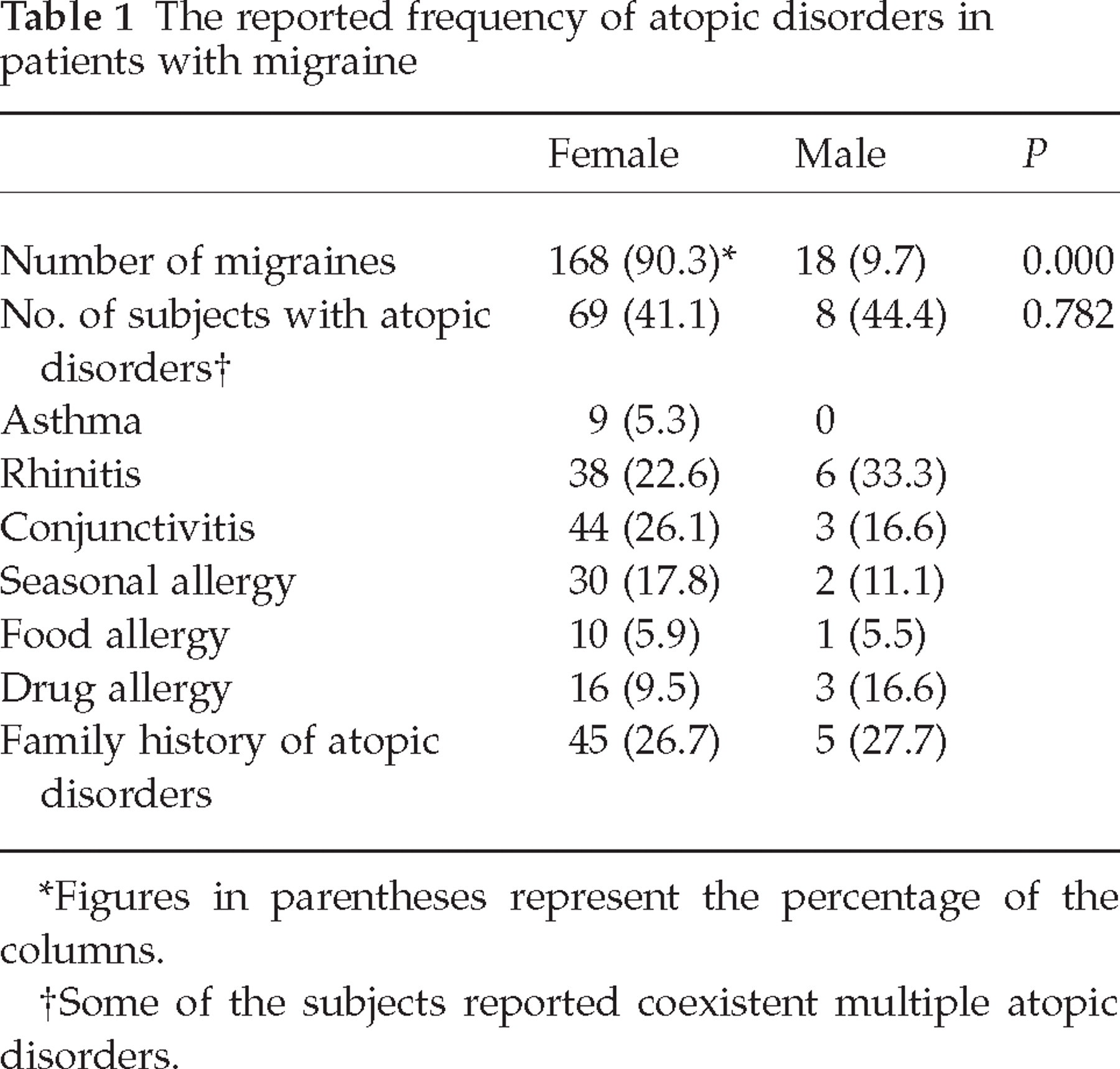
Figures in parentheses represent the percentage of the columns.
Some of the subjects reported coexistent multiple atopic disorders.
Demographic and headache characteristics of migraine patients with and without atopic disorders including asthma

Average frequency of attacks per month in past years.
VAS, Visual analogue scale.
The patients with CTTH were composed of 30 patients (13.3% male and 86.7% female). Their mean age was 42.0 ± 16.2 years (range 18–60 years) and the mean time from headache diagnosis was 85.2 ± 80.2 months (range 6–480 months). The patients described hypertension (eight patients, 26.6%) and smoking (16.6% past smokers and 6.6% current smokers) in their past medical histories. Only one patient described a history of penicillin allergy and none described other atopic disorders, including asthma. The general headache characteristics of the patients with CTTH were as follows: average duration of headache attacks was 6.5 ± 3.2 h, average frequency of attacks was 18 ± 3.8 months, average severity of attacks was 5.2 ± 1.2 on a visual analogue scale score. Headache attacks were commonly generalized but some of the patients reported a suboccipital location of headache. Most patients described their attacks as of a pressing or dull quality, with evenings commonly being the presentation time. As associated features nausea (26.6%), vomiting (16.6%), photophobia (20%) and phonophobia (20%) were reported. Among the aggravating factors of headache, 17 patients (56.6%) reported emotional stress and only three (10%) reported seasonal characteristics of attacks. Neurological examination of the patients showed depressive (13.3%) and anxious symptomatology (36.6%) but no localization signs.
Except for decreased deep tendon reflexes (4.8%) and positive sensory findings of the lower extremities (2.6%), possibly relating to their diabetes mellitus, none of the patients showed any neurological abnormality. All patients underwent cranial computed tomography investigation and none of them showed any significant abnormality, except non-specific ischaemic changes in periventricular regions in some. After the neurological evaluations the IHS revised diagnoses of the patients were as follows; 144 patients with migraine without aura (77.4%), four patients with possible migraine without aura (2.1%), 15 patients migraine with aura (8.0%), three patients possible migraine with aura (1.6%) and 20 patients (10.7%) migraine with aura coexistent with episodic tension-type headache.
Comparison of PFT variables between the two migraine groups and the control group showed some important limitations of the airway, especially in patients with migraine with atopic disorders. Additionally, patients with migraine with atopic disorders showed significantly lower PFT levels than migraine without atopic disorder, especially FEV1%, FVC%, force expiratory flow (FEF)25−75 (predicted) and FEF25−75% measurements. These results suggested a generally decreased pulmonary capacity independent of smoking and age in addition to a possible role of small airway obstruction (decreased FEF25−75 levels) in patients with migraine. Significantly lower FEF25−75% levels in the migraine with atopic disorders group compared with both the migraine without aura and the CTTH groups pointed to this possibility. On the other hand, independent of atopic disorders, FVC%, FEV1/FVC and FEF25−75 (l/s) measurements were lower in the patients with migraine than the CTTH patients and it was accepted as a predictor of small and large airway disorders (see Table 3).
Comparison of pulmonary function test results between the patients with and without atopic disorders including asthma

All figures represent mean ± SD.
MWA, Migraine with atopic disorders; MWOA, migraine without atopic disorders; P1, comparison between migraine with and without atopic disorders; P2, comparison between migraine with atopic disorders and chronic tension-type headache (CTTH) patients; P3, comparison between migraine without atopic disorders and CTTH patients; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; FEF, forced expiratory flow; PEF, peak expiratory flow.
Immunological screening of the patients with CTTH showed IgE abnormality in only two patients (6.6%) and IgA abnormality in only one patient. On the other hand, the patients with atopic disorders showed a significantly higher ratio of IgE abnormality and eosinophil count when compared with the patients with migraine without atopic disorders (see Table 4). Logistic regression analysis revealed that duration of headache (ExpB: 0.004, P = 0.025), age of patients (ExpB: 11.6, P = 0.001), FEV1% (ExpB: 13.46, P = 0.000), FVC% (ExpB: 10.35, P = 0.001) and FEF25−75% (ExpB: 6.42, P = 0.011) variables had important effects on having migraine with atopic disorders (binary logistic regression analysis, forward stepwise likelihood ratio). In this model the most important PFT parameter was FEV1% (ExpB: 0.96, P = 0.000). Likewise, correlation analysis showed that the atopic migraine history of patients was highly correlated with FEV1% (CC: 0.269, P = 0.000), FVC% (CC: 0.236, P = 0.001) and FEF25−75% (CC: 0.186, P = 0.011) parameters of PFT (Pearson correlation analysis).
Comparison of the number of abnormalities determined by immunological parameters in patients with and without atopic disorders
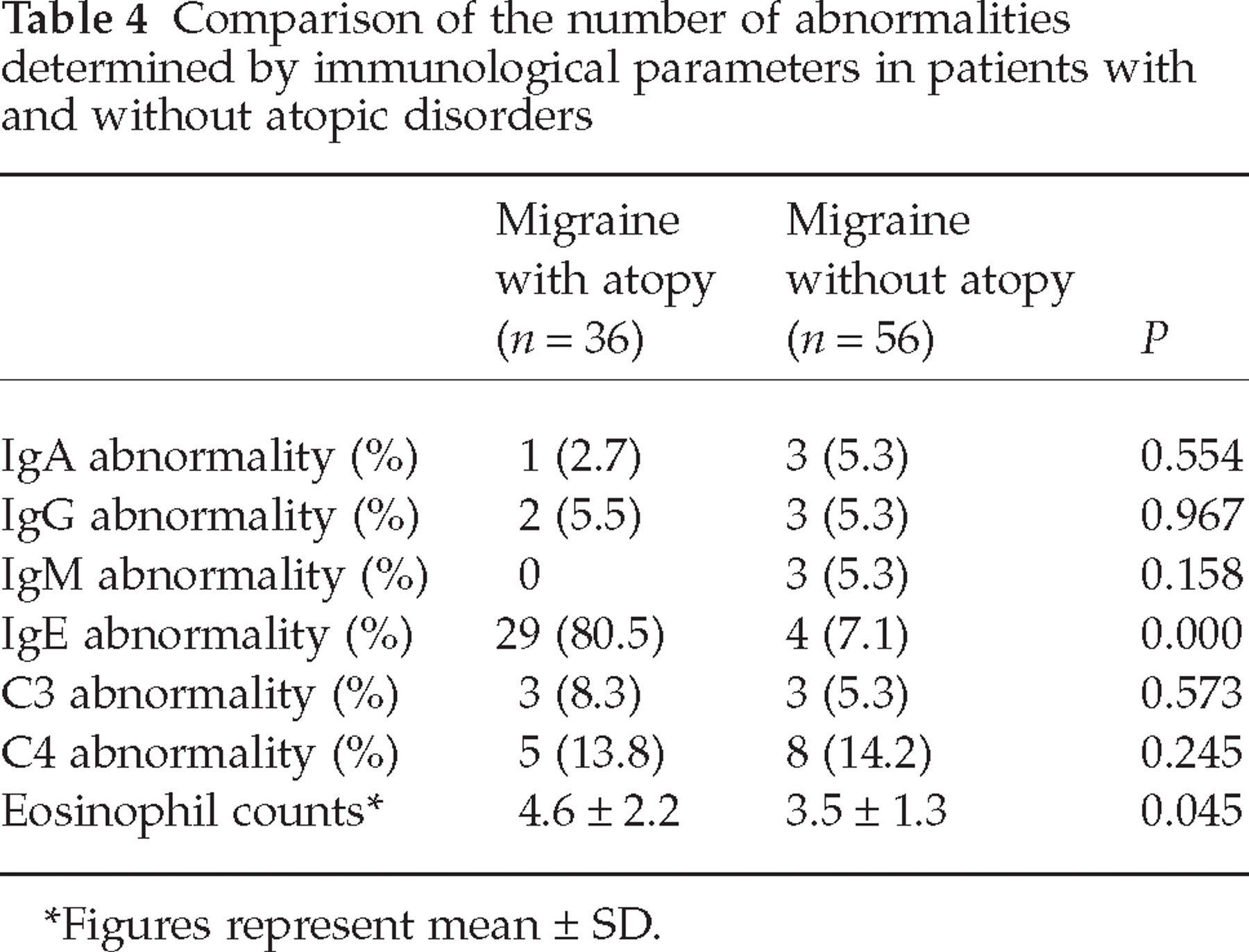
Figures represent mean ± SD.
For immunological parameters, regression analysis showed an important effect of increased IgE levels on having migraine with atopic disorders (ExpB: 9.33, P = 0.001). Except for a history of asthma, other clinical and laboratory variables showed no significant effect on IgE abnormalities (ExpB: 2.6, P = 0.001). However, when PFT variables and immunological parameters were evaluated together in this model, IgG abnormality (ExpB: 1.21, P = 0.023), IgA abnormality (ExpB: 0.5, P = 0.014) and IgE abnormality (ExpB: 0.99, P = 0.009) showed an important effect on having atopic disorders in patients with migraine. The overall percentage of this model was 88.2% by forward selection model of binary logistic regression analysis (step 1).
Discussion
Despite many studies on the association of migraine and atopy, methodological problems limit a clear comparison and evaluation of the results. Our study showed an important relationship between atopic disorder and migraine based on historical, functional and laboratory data.
In a detailed epidemiological study, Mortimer et al. (15) reported a high frequency of asthma (67%) among atopic children who complained of headache. They reported atopic illnesses in migraineurs as follows (overall figures in parentheses): asthma 30.2% (19.6%), eczema 32.1% (20.2%), and rhinitis 18.9% (7.6%). Their statistical analysis revealed that headache was significantly associated with atopic disorders. They suggested that a history of atopy should support the diagnosis of migraine in young children with paroxysmal headaches. As supportive data, Hoyos (24) reported that headaches in adults were found to be more prevalent among those whose family members were reported to have allergy, asthma and migraine. Likewise, a parental history of migraine was found to be more common among asthmatic children than among age- and sex-matched controls in a comprehensive hospital-based study in Turkey (odds ratio 5.5, 95% confidence interval 1.3, 25.0) (12). Our study groups showed a considerably increased prevalence of atopic disorders in patients with migraine, including asthma. The ratio of atopic disorders found in our study (41.4%) was higher than the prevalence reported in a Turkish study which found a rate of 26.1% for atopic disorders in subjects without asthma, but closer to the rate of atopy prevalence (42%) in patients with asthma (25). However, it should be kept in mind that our study was not population-based. History of atopic disorders did not show any significant difference for gender.
How can this possible relationship be explained? Is this a coincidence or a pathophysiological association? There are numerous similarities between migraine and asthma. Both have an inherited component, show alterations in vasoactive mediators and may in some situations be precipitated by certain foods, exercise and emotional stress. Platelet aggregation, known to occur in migraine, for example, is mediated by platelet activating factor (PAF) which also causes bronchoconstriction. Substance P, which is released on electrical stimulation of the trigeminal nerve, plays an important role in migraine pathogenesis by causing neurogenic inflammation and affects plasma extravasations when applied to the duramater. Moreover, substance P is also localized to unmyelinated sensory fibres (C-fibres) in airways and may take part in the inflammatory response of asthma. In addition to contraction of airway smooth muscle, substance P also potentially stimulates airway mucus secretion and increases airway microvascular permeability and exudation of plasma into the lumen (15, 26–31).
There are numerous reports on the frequency and links between other atopic disorders and migraine. Perennial allergic rhinitis (PAR) has higher level of comorbidities than seasonal allergic rhinitis (SAR). Our study has shown an association between migraine and atopic disorders, in both the positive family history and PFT abnormalities, partially confirming findings reported by others (1, 7, 15, 32). In their large case–control study, Davey et al. (1) have provided evidence for an association between migraine and asthma. They pointed out that migraine was more strongly associated with non-atopic asthma, which might also account for the increased risk of chronic obstructive pulmonary disease (COPD) in migraine patients, and they recommended that more detailed clinical and epidemiological studies be conducted in order to identify the association between migraine and asthma. For this reason, we included PFT and a detailed neurological examination in the study design. Our results drew attention to the importance of FEF25−75 measurements which are considered a marker of small airway resistance, in subjects at risk, especially in those with a positive family history for migraine or atopic disorders.
A basic limitation of this study was that the design lacked a longitudinal follow-up of patients for the development of COPD and other pulmonary disorders, and other allergy screening tests were not included because of ethical procedures in our hospital. On the other hand, to the best of our knowledge this study is the first concerning the clinical relationship between migraine and asthma based on a broad spectrum of asymptomatic atopic patients.
We have found no evidence in the available literature of a major role of immunoglobulin and/or complement-mediated type II or III hypersensitivity in migraine. In 1977, Lord reported decreased complement 4 (C4) and C5 in the early headache phase of nine migraine patients without aura (33). In another study, 20 classic migraineurs had increased levels of IgA ictally and interictally, and 35 non-prodromal (common) migraineurs showed increased IgA and IgG levels during both periods (34). IgA, IgG and IgM were reported to be increased during the headache-free phase (35), but the interictal IgA levels were decreased in another study (36). However, most clinical studies found no change in IgA, IgG and IgM levels (37–39) or complement factors (38, 40) in migraineurs, during either the headache or the interictal phase (38, 40, 41). Therefore, the conclusion seems justified that there is no change of serum immunoglobulin or complement levels in migraineurs and that these factors are not involved in migraine precipitation. This aspect our study showed only a positive correlation between migraine and increased levels of IgE, supported with increased eosinophil counts, except co-evaluation of PFT variables and immunological screenings. Also, this evaluation showed an important effect of IgG, IgA and IgE levels on the atopic disorders of migraineurs. We did not show any other immunological abnormality during headache-free intervals, supporting some above-mentioned reports.
The combined effect of genetic and environmental factors raises the possibility of an inflammation-induced trigeminal hyperalgesia as a mechanism of increased migraine vulnerability in genetically predisposed individuals. Thus, inflammation-induced hyperalgesia could explain why migraineurs report the highest headache intensity after an infection and why certain precipitants generate migraine in some conditions but not in others. This study has added some supportive evidence to above-mentioned issue.
We thought that PFT abnormalities found in patients with migraine were not the result of mere coincidence, supporting some previous epidemiological studies, although we were unable to make a causal link. At this point, the prognostic role of PFT screening of these patients in order to determine possible progressive lung disorders of migraine disorders, especially in those having important risk factors, should be open for discussion.
Footnotes
This study has been presented at the 8th Congress of the European Neurology Federation of Neurological Societies, Paris, 4–7 September 2004, as poster presentation (P1243–P1244).
