Abstract
Migraine is a common neurological disorder that is associated with an increase in plasma calcitonin gene-related peptide (CGRP) levels. CGRP, a potent vasodilator released from the activated trigeminal sensory nerves, dilates intracranial blood vessels and transmits vascular nociception. Hence, inhibition of trigeminal CGRP release may prevent neurotransmission and, thereby, ameliorate migraine headache. Therefore, the present study in anaesthetized pigs investigates the effects of a selective adenosine A1 receptor agonist, GR79236 (3, 10 and 30 μg/kg, i.v.) on capsaicin-induced carotid haemodynamic changes and on plasma CGRP release. Intracarotid (i.c.) infusion of capsaicin (10 μg/kg/min, i.c.) increased the total carotid blood flow and conductance as well as carotid pulsations, but decreased the difference between arterial and jugular venous oxygen saturations. These responses to capsaicin were dose-dependently attenuated by GR79236. However, the increases in the plasma CGRP concentrations by capsaicin remained essentially unmodified after GR79236 treatment. The above results suggest that GR79236 may have an antimigraine potential due to its postjunctional effects (carotid vasoconstriction) rather than to prejunctional inhibition of trigeminal CGRP release.
Keywords
Introduction
Migraine is a neurovascular disorder associated with cranial vasodilation and activation of perivascular trigeminal sensory nerves. Thus, activation of trigeminal sensory nerves releases calcitonin-gene related peptide (CGRP), which dilates cranial blood vessels and stimulates sensory nerve transmission (1, 2). In line with the above finding, it has been shown that during the headache phase of migraine, plasma concentrations of CGRP, but not of other neuropeptides, are elevated and these levels are normalized by triptans in parallel with amelioration of headache (3). Hence, trigeminal CGRP release is considered as a marker for trigeminal activation that can be measured in a jugular venous blood sample; accordingly, the decrease in this marker seems to be highly predictive of antimigraine activity in humans (1, 3). Therefore, a compound capable of inhibiting trigeminal CGRP release may be an effective agent in treating migraine (2–5).
Several experimental studies have suggested that adenosine, a purine analogue, may play an important role in blocking nociception (6) via adenosine A1 receptors (7). Moreover, adenosine A1 receptor agonists are effective in preclinical antinociceptive tests (8) and against experimental inflammatory and neuropathic pain (9). In addition, activation of adenosine A1 receptors inhibits the synthesis and release of CGRP from capsaicin-sensitive sensory neurons in the spinal cord (10, 11) and in the periphery (12). In the light of the above findings, it has been demonstrated that GR79236, a highly selective adenosine A1 receptor agonist (13), inhibited: (i) neurogenic vasodilation in anaesthetized rats (14), (ii) electrical stimulation of superior saggital sinus-induced trigeminovascular nociceptive transmission and CGRP release in anaesthetized cats (15) and (iii) trigeminal nociception in humans (16). However, to the best of our knowledge, there is no comprehensive in vivo evidence to show that capsaicin-induced trigeminal activation (trigeminal CGRP release) can be modulated by A1 receptor agonists. Therefore, the main objectives of the present study in anaesthetized vagosympathectomized pigs were to investigate the effects of GR79236 on capsaicin-induced carotid haemodynamic changes and plasma CGRP release.
Materials and methods
General
After an overnight fast, 15 domestic pigs (Yorkshire × landrace, female, 10–14 kg), divided into two groups (n = 8 and n = 7 for vehicle and GR79236, respectively), were sedated with azaperone (120 mg, i.m.), midazolam hydrochloride (10 mg, i.m.) and then anaesthetized with sodium pentobarbital (600 mg, i.v.). After tracheal intubation, the animals were connected to a respirator (BEAR 2E; BeMeds AG, Baar, Switzerland) for intermittent positive pressure ventilation with a mixture of room air and oxygen. Respiratory rate, tidal volume and oxygen supply were adjusted to keep arterial blood gas values within physiological limits (pH 7.35–7.48; pCO2 35–48 mmHg; pO2 100–120 mmHg). Anaesthesia was maintained with a continuous i.v. infusion of sodium pentobarbital (12–20 mg/kg/h). This anaesthetic regimen, together with bilateral vagosympathectomy (see below), increases the heart rate and markedly dilates carotid arterioles and arteriovenous anastomoses. Consequently, the carotid blood flow, particularly its arteriovenous anastomotic (AVA) fraction, was considerably higher in these pigs than in conscious or thiopental-anaesthetized pigs (17).
Heart rate was measured with a tachograph (CRW; Erasmus University, Rotterdam, the Netherlands) triggered by electrocardiogram signals. Both common carotid arteries were dissected free and the accompanying vagosympathetic trunks were cut between two ligatures to prevent any possible influence by baroreceptor reflexes on the carotid vascular responses produced by capsaicin. Pulsatile and mean carotid blood flows were measured in the right common carotid artery with a flow probe (internal diameter 2.5 mm) connected to a sine-wave electromagnetic flow meter (Transflow 601-system; Skalar, Delft, the Netherlands). The amplitude of carotid blood flow signals provided an index of the carotid flow pulse. Subsequently, two hub-less needles, connected to a polyethylene tube, were inserted into the right common carotid artery for the administration of capsaicin and the α1-adrenoceptor agonist phenylephrine. The use of phenylephrine was necessitated by the fact that the carotid arterioles and arteriovenous anastomoses were in a dilated state under the anaesthetic regimen (17); therefore, to study the effects of vasodilator agents (in the present case, capsaicin), these shunt vessels had first to be constricted. As described earlier (18), phenylephrine decreases total carotid conductance (TCC) exclusively by constricting carotid arteriovenous anastomoses, which results in an increase in the difference between arterial and jugular venous oxygen saturations (A–V SO2 difference) (19).
Last, catheters were placed in: (i) the right external jugular vein for the withdrawal of venous blood samples to measure blood gases (ABL-510; Radiometer, Copenhagen, Denmark) and plasma concentrations of CGRP (see below); (ii) the inferior vena cava (through the left femoral vein) for the administration of vehicle or GR79236; and (iii) the aortic arch (through the left femoral artery) for the measurement of arterial blood pressure (Combitrans disposable pressure transducer; Braun, Melsungen, Germany) as well as for the withdrawal of arterial blood samples to measure blood gases.
Heart rate and systolic, diastolic and mean arterial blood pressures as well as mean and pulsatile carotid artery blood flows were continuously monitored on a polygraph (CRW; Erasmus University). Vascular conductances were calculated by dividing the respective blood flows (ml/min) by mean arterial blood pressure (mmHg), multiplied by 100 and expressed as 10−2 ml min−1 mmHg−1. During the experiment, body temperature was maintained at 37 ± 1 °C by a heating pad and each animal was infused with physiological saline to compensate for fluid losses.
Determination of plasma levels of CGRP
Jugular venous blood samples were obtained from the 15 pigs, receiving either vehicle (n = 8) or GR79236 (n = 7). Blood was transferred immediately into a polypropylene tube containing ethylene dinitrotetraacetic acid (1 mg/ml of blood) and aprotinin (500 kIU/ml of blood). Aprotinin was used to inhibit endogenous plasma proteases, since we found that CGRP was not detectable in biological samples without aprotinin (unpublished). After centrifugation at 1600 g for 15 min, plasma samples were coded and stored at − 80°C until CGRP measurements were performed. The person measuring CGRP concentrations remained blind to the treatments until all data had been collated. CGRP was extracted from plasma using a C18 SEP-COLUMN, dried by lypholization and measured by radioimmunoassay (20), as per protocol of the Peninsula Laboratories, Inc. (Belmont, CA, USA).
Experimental protocol
The experiments (n = 15 animals) were started after a stabilization period of 15–20 min. Subsequently, baseline haemodynamic variables were measured (heart rate 118 ± 7 beats/min, mean arterial blood pressure 105 ± 4 mmHg, mean carotid blood flow 139 ± 11 ml/min, mean carotid conductance 114 ± 16 10−2 ml min−1 mmHg−1, and A–V SO2 difference 12 ± 3%) and phenylephrine was infused into the right common carotid artery at a rate of 10 µg/kg/min for 10 min, followed by 3–6 µg/kg/min throughout the rest of the experiment to maintain carotid blood flow at a constant low level. After a period during which the haemodynamic variables remained constant for at least 60 min (heart rate 169 ± 6 beats/min, mean arterial blood pressure 115 ± 7 mmHg, mean carotid blood flow 115 ± 16 ml/min, mean carotid conductance 81 ± 5 10−2 ml min−1 mmHg−1, and A–V SO2 difference 24 ± 5%; n = 15), the animals received consecutive infusions (0.15, 1.5 and 4.5 ml, i.c. for 3 min each) of the capsaicin vehicle (see Compounds and kits).
Five to 10 min after the last infusion of the capsaicin vehicle, blood samples were obtained for the measurements of blood gases and CGRP concentrations and the values of heart rate, mean arterial blood pressure and total carotid blood flow (TCBF) and TCC as well as carotid pulse were collated (baseline values). The animals then received consecutive infusions of capsaicin (1, 3 and 10 µg/kg per minute, i.c. for 3 min each) and heart rate, arterial blood pressure, TCBF and TCC and plasma CGRP concentrations were determined at the end of each capsaicin infusion. In addition, after the last infusion of capsaicin (10 µg/kg per minute), blood gases were measured as described above (control values). Subsequently, a recovery period of 20 min was allowed until all haemodynamic parameters had returned to baseline levels. At this point, the animals were divided into two groups, which were treated with i.v. infusions (rate 1 ml/min for 3 min) of either vehicle (three times, 3 ml of physiological saline; n = 8) or GR79236 (3, 10, and 30 µg/kg; n = 7). Seven minutes after each infusion, capsaicin was given and the haemodynamic and biochemical variables were measured again, as described above.
Data presentation and statistical analysis
All data are presented as mean ± SEM. The statistical analysis was performed using the SPSS package for windows (version 11.0; SPSS Inc., Chicago, IL, USA). The significance of changes within one group (vehicle or GR79236) was analysed with repeated-measures
Ethical approval
The Ethics Committee of the Erasmus MC, Rotterdam dealing with the use of animals in scientific experiments approved the protocols for this investigation.
Compounds and kits
The following compounds were used: aprotinin (5850 kIU/mg; Roth, Karlsruhe, Germany); azaperone (Stresnil®; Janssen Pharmaceuticals, Beerse, Belgium); GR79236 (gift from Dr H. E. Connor, Glaxo Group Research, Stevenage, UK); capsaicin, Tween 80, ethanol and phenylephrine hydrochloride (all from Sigma-Aldrich Chemie b.v., Zwijndrecht, the Netherlands); ethylene dinitro-tetraacetic acid (Merck, Darmstadt, Germany); heparin sodium (to prevent blood clotting in catheters; Leo Pharmaceutical Products, Weesp, the Netherlands); midazolam hydrochloride (Dormicum®; Hoffmann La Roche b.v., Mijdrecht, the Netherlands); and sodium pentobarbital (Sanofi Sante b.v., Maasluis, the Netherlands). The radioimmunoassay kit for CGRP was purchased from Peninsula Laboratories, Inc.
Capsaicin was initially dissolved in Tween 80, ethanol and physiological saline at a ratio of 0.5 : 1 : 8.5 ml, respectively. Phenylephrine was dissolved in distilled water, while GR79236 was dissolved in sterile physiological saline.
Results
Baseline values
Baseline values (i.e. after capsaicin vehicle infusion) in the 15 pigs used were: heart rate 149 ± 8 beats/min; mean arterial blood pressure 113 ± 4 mmHg; TCBF 83 ± 11 ml/min; TCC 68 ± 8 10−2 ml min−1 mmHg−1; A–V SO2 difference 31 ± 5%; and plasma CGRP concentration 10 ± 1 pg/ml. No significant differences in the baseline values were found between the two groups that subsequently received vehicle (n = 8) or GR79236 (n = 7).
Capsaicin-induced systemic haemodynamic changes
Figure 1 depicts the heart rate and mean arterial blood pressure values at baseline and following different doses of capsaicin (1, 3 and 10 µg/kg/min, i.c.) given before (control response) and after treatment with GR79236 (3, 10 and 30 µg/kg, i.v.) or the corresponding volumes of vehicle (3 ml, three times, i.v.). In both groups, the higher infusions of capsaicin (3 and 10 µg/kg per minute, i.c.) significantly increased the mean arterial blood pressure (per cent change from baseline 19 ± 4) and heart rate (only the highest dose; per cent change from baseline 15 ± 3). These changes to capsaicin remained essentially unmodified following vehicle or GR79236 treatment.

Heart rate (HR) and mean blood pressure (MAP) at baseline (0 µg/kg per minute capsaicin) and following infusions of capsaicin (1, 3 and 10 µg/kg er minute, i.c.) in anaesthetized pigs before (Control) and after i.v. administrations of vehicle (V, 3 ml, three times; n = 8) or GR79236 (3, 10 and 30 µg/kg; n = 7). ∗P < 0.05 vs. baseline; ♯P < 0.05 vs. response after the corresponding volume of vehicle. ○, Control; •, V/3; □, V/10; ▪, V/30.
It is noteworthy that the last two doses of GR79236 (10 and 30 µg/kg, i.v.) significantly decreased the baseline heart rate without altering blood pressure (see Fig. 1 and Table 1; ♯P < 0.05 vs. corresponding values in the vehicle-treated group).
Effects of vehicle or GR79236 on heart rate, mean blood pressure, arterial jugular venous oxygen saturation difference (A–V SO2), plasma calcitonin gene-related peptide (CGRP) concentrations and total arotid conductance in anaesthetized pigs

All values are expressed as mean ± SEM.
P < 0.05 vs. baseline;
P < 0.05 vs. corresponding values in vehicle-treated animals.
Capsaicin-induced carotid haemodynamic changes
The effects of capsaicin on carotid haemodynamic changes were investigated in detail in the animals receiving vehicle or GR79236.
Total carotid blood flow and conductance
Figure 2 shows the effects of capsaicin (10 µg/kg/min, i.c.) on TCBF and TCC, given before (control response) and after treatment with GR79236 (3, 10 and 30 µg/kg per minute, i.v.) or the corresponding dose of vehicle (3 ml, three times; i.v.). Of note, we also investigated lower doses of capsaicin (1 and 3 µg/kg/min, i.c.) on these variables. However, these lower doses of capsaicin produced no significant effects on TCBF and TCC (data not shown). Capsaicin markedly and significantly increased the TCBF and the corresponding conductance and these increases remained unaffected after treatment with the three doses of the vehicle. Interestingly, GR79236 (particularly in doses of 10 and 30 µg/kg per minute, i.v.) produced a dose-dependent and significant attenuation of the above-mentioned responses to capsaicin (Fig. 2). In vehicle-treated animals, repeated administration of capsaicin produced a small but non-significant attenuation of TCBF and TCC and increase in A–V SO2 difference (Figs 2 and 3).

Changes in total carotid blood flow (TCBF) and total carotid conductance (TCC) at baseline and after infusions of capsaicin (10 µg/kg/min, i.c.) given in anaesthetized pigs before (Control) and after i.v. administrations of vehicle (V, 3 ml, three times; n = 8) or GR79236 (3, 10 and 30 µg/kg; n = 7). ∗P < 0.05 vs. baseline; ♯P < 0.05 vs. response after the corresponding volume of vehicle.
In both groups, the infusion of capsaicin (10 µg/kg per minute, i.c.) also increased the carotid pulsations, an effect that was not affected by either vehicle or GR79236 treatment (data not shown).
Difference between arterial and jugular venous oxygen saturations (A–V SO2 difference)
Figure 3 depicts the A–V SO2 difference at baseline and following an infusion of capsaicin (10 µg/kg/min, i.c.) given before (control response) and after treatment with GR79236 (3, 10 and 30 µg/kg, i.v.) or the corresponding volumes of vehicle (3 ml, three times; i.v.). The above infusion of capsaicin significantly reduced the A–V SO2 difference in both groups (compare baseline and control values). This response to capsaicin remained essentially unaffected after treatments with vehicle, but was dose-dependently and significantly attenuated by all doses of GR79236. The highest dose of GR79236 (30 µg/kg, i.v.) per se significantly increased the A–V SO2 difference (see Table 1).

Arterial and jugular venous oxygen saturation difference (A–V SO2 difference) measured at baseline and after the infusion of capsaicin (10 µg/kg/min, i.c.) given in anaesthetized pigs before (Control) and after i.v. administrations of vehicle (V, 3 ml, three times; n = 8) or GR79236 (3, 10 and 30 µg/kg; n = 7). ∗P < 0.05 vs. baseline; ♯P < 0.05 vs. response after the corresponding volume of vehicle.
Capsaicin-induced increase in the jugular venous plasma concentrations of CGRP
Figure 4 depicts plasma CGRP concentrations at baseline and following the infusion of capsaicin (10 µg/kg/min, i.c.), given before (control response) and after treatment with GR79236 (3, 10 and 30 µg/kg/min, i.v.) or the corresponding dose of vehicle (3 ml, three times; i.v.). Lower doses of capsaicin (1 and 3 µg/kg/min, i.c.) were without significant effects (data not shown). In both treatment groups, the above dose of capsaicin significantly increased the plasma CGRP concentrations (maximal per cent change from baseline > 100 in both groups). This response to capsaicin remained unaffected after treatment with vehicle or GR79236 (Fig. 4). Moreover, the resting plasma CGRP levels remained unchanged after treatment with GR79236 (Table 1). As has been published previously, it is notable that sumatriptan that affects CGRP release did not modify capsaicin-induced CGRP release in anaesthetized pigs (25).
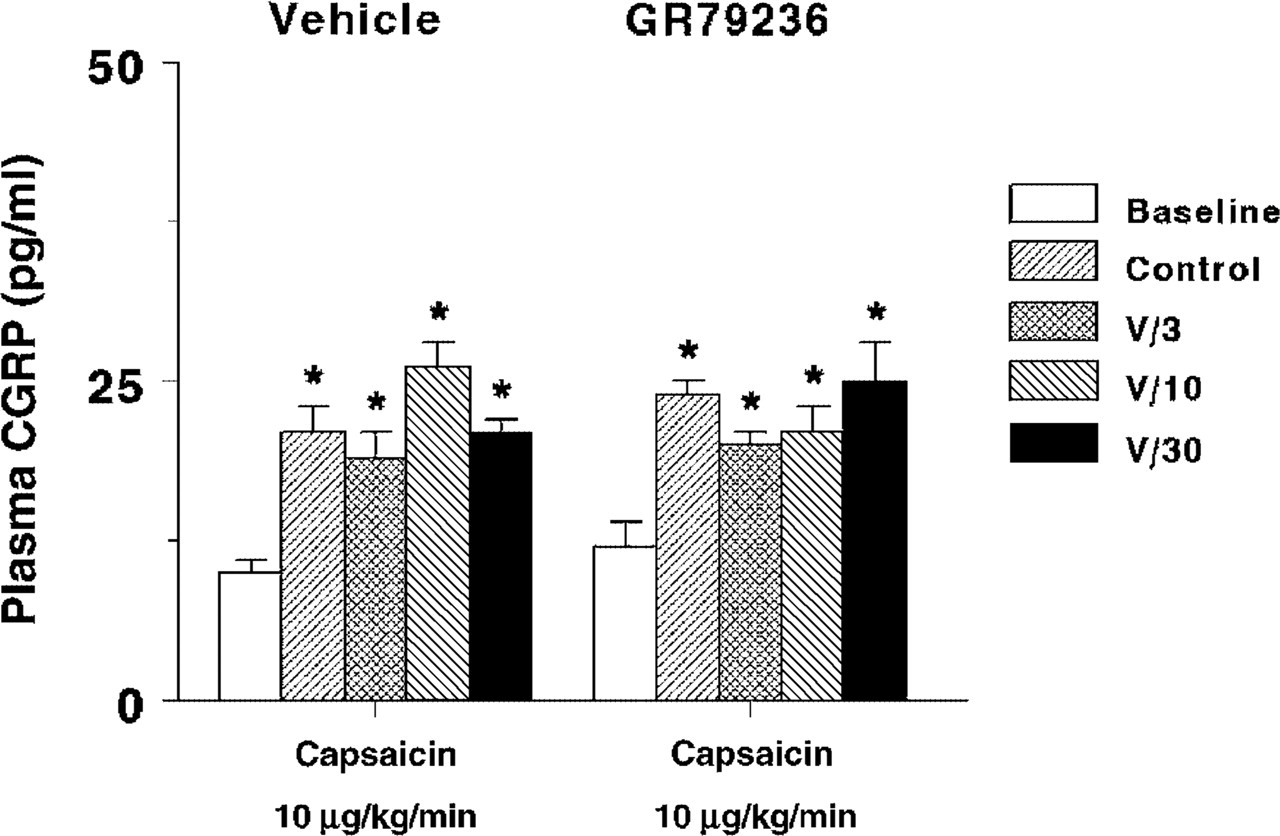
Jugular venous plasma calcitonin gene-related peptide (CGRP) concentrations measured at baseline and after the infusion of capsaicin (10 µg/kg/min, i.c.) given in anaesthetized pigs before (Control) and after i.v. administrations of vehicle (V, 3 ml, three times; n = 8) or GR79236 (3, 10 and 30 µg/kg; n = 7). All values are expressed as mean ± SEM. ∗P < 0.05 vs. baseline.
Discussion
The present investigation in anaesthetized pigs shows that: (i) capsaicin increased the TCBF and TCC as well as jugular venous plasma CGRP concentrations, but narrowed the A–V SO2 difference, and (ii) GR79236 dose-dependently attenuated the capsaicin-induced carotid haemodynamic changes, but not the CGRP release.
Effects of GR79236 on capsaicin-induced systemic haemodynamic changes
The systemic haemodynamic effects of capsaicin have been investigated extensively (25–27). Thus, the significant decrease in heart rate produced by GR79236 in our study is a consequence of its cardiac depressive effect that is mediated through adenosine A1 receptors (6, 15, 28, 29). Nevertheless, GR79236 did not modify the capsaicin-induced increases in heart rate and blood pressure, which are mediated by the release of vasopressor agents, such as neurokinin A and catecholamines (26).
Effects of GR79236 on capsaicin-induced carotid haemodynamic changes
Carotid haemodynamics
Several experimental studies have suggested that cranial blood vessels are innervated by CGRP–containing trigeminal sensory nerves, which may be involved in the vascular responses of cerebral arteries (1). In this respect, as previously reported (27), the capsaicin–induced porcine carotid vasodilation in our study are mediated by the release of CGRP. Admittedly, as reported earlier (30), vasodilator responses to capsaicin tended to wear off in vehicle–treated animals (non-significant), suggestive of tachyphylaxis. This tachyphylaxis was not due to complete depletion of CGRP from the nerve terminals, but rather limited, possibly due to a neuronal reuptake of released CGRP into capsaicin–sensitive perivascular nerves (31). It is clearly evidenced in our study that the plasma concentrations of CGRP did not vary significantly after repeated infusion of capsaicin (Fig. 4). In contrast to vehicle-treated animals, the slight (though significant) attenuation of capsaicin–induced carotid vasodilation by GR79236 could be attributed to two possible mechanisms: (i) prejunctional inhibition of CGRP release by A1 receptors; and/or (ii) postjunctional counteraction of the CGRP-induced response. The former possibility can be excluded, as our results clearly show that GR79236 had no effect on capsaicin-induced CGRP release (see Fig. 4). Moreover, since GR79236 has no effect on CGRP-induced carotid vasodilation (14), it is most likely that this compound produces a physiological antagonism, similar to that of triptans (25), which involves carotid vasoconstriction.
It has been suggested that GR79236 inhibits neurogenic vasodilation in anaesthetized rats by prejunctional inhibition of trigeminal CGRP release (14). Admittedly, this effect of GR79236 was not supported with plasma CGRP measurements (14). Therefore, no categorical conclusion can be drawn whether attenuation of neurogenic vasodilation by GR79236 is due to prejunctional modulation of CGRP release. Furthermore, GR79236 per se had no effect on either the baseline total carotid conductance (Table 1) or the resting meningeal artery diameter (14). Hence, as discussed below, the attenuation of capsaicin-induced carotid vasodilation by GR79236 in our study could be due predominantly to vasoconstriction of carotid arteriovenous anastomoses, since the baseline A–V SO2 difference was increased by GR79236 (Table 1).
A–V SO2 difference
As previously described, the capsaicin-induced reduction in A–V SO2 difference in anaesthetized pigs is mediated by CGRP release (27). This view gains additional weight within the context of antimigraine mechanisms when considering that during migraine there is a dilation of carotid arteriovenous anastomoses, which results in a narrowing of the A–V SO2 difference (19, 32). Moreover, the reduction in the carotid arteriovenous anastomotic blood flow, with a consequent normalization of the A–V SO2 difference, reconfirms our porcine vascular model as highly predictive of antimigraine activity (33–35). As GR79236 normalized the capsaicin-induced reduction in A–V SO2 difference and attenuated the capsaicin-induced increase in TCC, it is quite likely that this compound also constricts carotid arteriovenous anastomoses (not measured in the present study).
Admittedly, several lines of evidence have suggested that the direct effects of adenosine on blood vessels through adenosine A1 receptors are rare and, if so, they could be due to the modulation of presynaptic neurotransmitter release (6). Nevertheless, adenosine A1 receptors have been shown to mediate contractions in isolated porcine coronary arteries (36), guinea-pig aorta (37), guinea-pig pulmonary artery (38) and rat isolated spleen (39).
Effects of GR79236 on capsaicin-induced plasma CGRP levels
Stimulation of capsaicin-sensitive trigeminal nerve fibres with electrical or chemical (capsaicin) stimuli results in the release of endogenously stored CGRP (25, 27, 40, 41). Moreover, GR79236 has been shown to inhibit the electrically induced CGRP release without concomitant vasoconstriction (15); this suggests that GR79236 inhibits trigeminal CGRP release through prejunctional adenosine A1 receptors (15). Interestingly, it has been shown that the inhibitory effects of adenosine-related agonists are limited to electrical activation of sensory-motor nerves, but not to that of capsaicin stimulation (42, 43). However, CGRP concentrations were not measured in parallel and, consequently, no conclusion can be drawn whether adenosine modulates capsaicin-induced CGRP release. To the best of our knowledge, our results constitute the first in vivo evidence that GR79236 failed to modify capsaicin-induced trigeminal CGRP release; moreover, the attenuation of capsaicin-induced carotid haemodynamic changes by GR79236 is not associated with a corresponding change in CGRP levels.
Admittedly, there is no clear-cut explanation for GR79236 attenuating the responses to electrical stimulation, but not to capsaicin. However, capsaicin and electrical stimuli activate the efferent function of the sensory nerves by two independent mechanisms (43). In this context, Ω-conotoxin, a peptide modulator of neuronal calcium channels, has been shown to block the electrically induced release of neuropeptides (43–45), but not the excitatory effect of capsaicin on the same nerves (43). Furthermore, adenosine has been shown to inhibit neuronal calcium channels, which are responsible for calcium influx that triggers neuropeptide release (42). Therefore, the inhibitory effect of adenosine-related agonists on neurotransmitter release may be due to blockade of neuronal calcium channels that are sensitive to Ω-conotoxin (42). In contrast, capsaicin directly stimulates the transmitter release from the sensory nerve endings and these responses are mediated viaΩ-conotoxin-insensitive receptors (43). Moreover, we have recently reported that the antimigraine agent, sumatriptan (a selective 5-HT1B/1D receptor agonist that produces constriction of carotid arteriovenous anastomoses), did not have any effect on capsaicin-induced CGRP release (25).
Possible clinical implications
Finally, it is important to consider the possible clinical implications of our results with GR79236 in the context of antimigraine therapy. The attenuation of capsaicin-induced carotid vasodilation with a concomitant increase in the A–V SO2 difference suggests vasoconstriction of carotid arteriovenous anastomoses. Therefore, inhibition of cranial vasodilation, including arteriovenous anastomoses, may form a better treatment strategy to abort migraine attacks than inhibition of trigeminal CGRP release. Indeed, to date, all acutely acting antimigraine agents, such as triptans and ergot alkaloids, produce vasoconstriction of carotid arteriovenous anastomoses (27, 33, 35). Moreover, BIBN4096BS (a CGRP receptor antagonist with antimigraine properties) inhibits capsaicin-induced vasodilation of carotid arteriovenous anastomoses (27, 33, 35).
The pathophysiology of migraine headache involves dilation of carotid arteriovenous anastomoses, which narrows the A–V SO2 difference (32, 34). Thus, normalization of the A–V SO2 difference by GR79236 leads us to suggest that GR79236 may have antimigraine potential. Admittedly, the bradycardia observed in our study may limit the use of GR79236 in clinical practice. However, a recent clinical investigation with GR79236 (10 µg/kg, i.v.) showed no significant side-effects (16). Obviously, clinical trials with GR79236 would be crucial in determining whether adenosine A1 receptor agonists can be developed as novel antimigraine agents.
In conclusion, our study shows that the adenosine A1 receptor agonist, GR79236, (i) dose-dependently attenuated capsaicin-induced carotid vasodilation, and (ii) did not modify capsaicin-induced increase in trigeminal CGRP release. Therefore, the antimigraine potential of GR79236 may be mediated through postjunctional effects rather than prejunctional inhibition of CGRP release.
Footnotes
Acknowledgements
The authors thank Dr H. E. Connor (GlaxoSmithKline Research, Stevenage, UK) for providing GR79236.
