Abstract
We investigated the effects of donitriptan, which possesses a uniquely high affinity and efficacy at 5-HT1B/1D receptors, on carotid and systemic haemodynamics in anaesthetized pigs. Donitriptan (0.16-100 μg kg-1, i.v.) dose-dependently decreased total carotid blood flow and vascular conductance (maximum response: -25 ± 3%). This effect was entirely due to a selective reduction in the cephalic arteriovenous anastomotic fraction (maximum response: -63 ± 3%; ED50%: 92 ± 31 nmol/kg); the nutrient vascular conductance increased. Donitriptan did not decrease vascular conductances in or blood flow to a number of organs, including the heart and kidneys; in fact, vascular conductances in the skin, brain and skeletal muscles increased. Cardiac output was slightly decreased by donitriptan, but this effect was confined to peripheral arteriovenous anastomoses. The haemodynamic effects of donitriptan were substantially reduced by the 5-HT1B/1D receptor antagonist GR127935. These results show that donitriptan selectively constricts arteriovenous anastomoses via 5-HT1B receptor activation. The drug should be able to abort migraine headaches and it is unlikely to compromize blood flow to vital organs.
Introduction
The last decade has seen tremendous progress in migraine therapy, with sumatriptan, belonging to a new class of drugs designated as 5-HT1B/1D/1F receptor agonists, providing the lead (1–3). Several studies have clearly established the therapeutic value of sumatriptan as well as of other triptans (4–7). Pharmacologically, triptans inhibit dural plasma protein extravasation, suppress action potentials in trigeminal nucleus caudalis, constrict isolated cranial blood vessels and decrease carotid arteriovenous anastomotic blood flow in anaesthetized animals (6, 8, 9). Although the trigeminal neural effects of triptans may be involved to some extent in their anti-migraine action (7, 10, 11), the efficacy of triptans is primarily attributed to cranial vasoconstriction (6, 10). Using compounds selective at 5-HT1B, 5-HT1D or 5-HT1F receptors, it is now well established that the vasoconstrictor effect of triptans is mediated via 5-HT1B receptors (12–18).
Despite its success in the treatment of migraine headaches, sumatriptan has several shortcomings, such as low bioavailability, headache recurrence and propensity to elicit chest symptoms (6). Several newer triptans display a better pharmacokinetic profile, but this has not been translated into any substantial improvement in either the efficacy or side-effect potential compared with sumatriptan (for reviews, see 6, 7). Recently, another triptan, donitriptan (F11356 or F12640), has been synthesized by the Centre de Recherche, Pierre Fabre, Castres, France (Fig. 1). Donitriptan displays a very high affinity at recombinant human as well as porcine 5-HT1B/1D receptors with little affinity for the 5-HT1F receptor and, more importantly, it has uniquely high intrinsic efficacy (inhibition of cAMP accumulation and enhancement of [35S]GTPγS binding) at human 5-HT1B/1D receptors (Tables 1, 19–23). John et al. (19, 24) have hypothesized that the relatively low intrinsic activity of the currently available triptans at 5-HT1B/1D receptors may explain this ceiling effect in therapeutic response and therefore donitriptan may show a high therapeutic efficacy.
Affinity and efficacy of donitriptan, sumatriptan and 5-HT at human (h) and porcine (p) 5-HT receptors
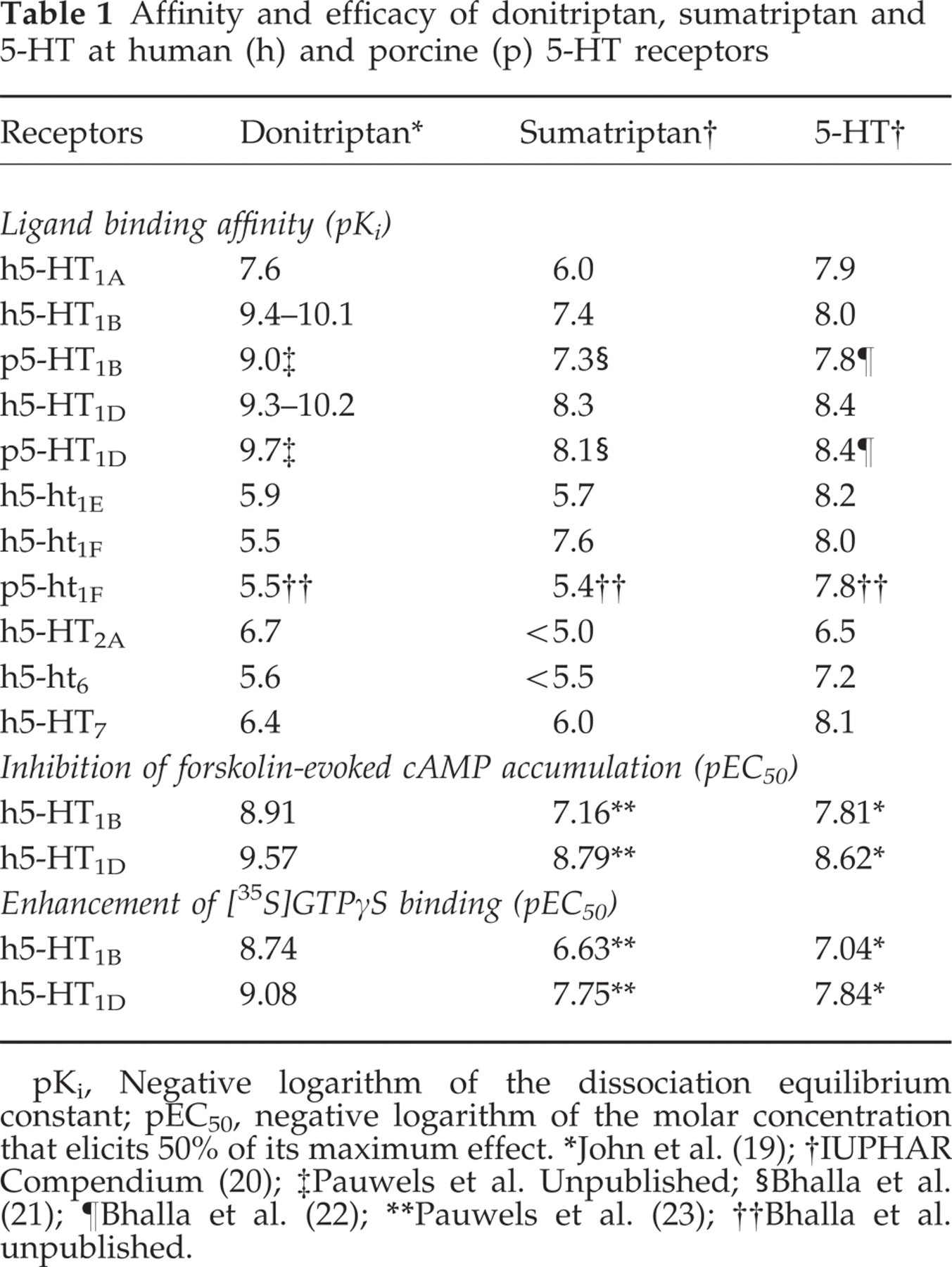
pKi, Negative logarithm of the dissociation equilibrium constant; pEC50, negative logarithm of the molar concentration that elicits 50% of its maximum effect.
∗John et al. (19);
†IUPHAR Compendium (20);
‡Pauwels et al. Unpublished;
§Bhalla et al. (21);
¶Bhalla et al. (22);
∗∗Pauwels et al. (23);
††Bhalla et al. unpublished.

Chemical structures of donitriptan and sumatriptan.
In the present study, we report on the effects of donitriptan on the distribution of carotid blood flow into nutrient (capillary) and non-nutrient (arteriovenous anastomotic) fractions in the anaesthetized pig. Previous investigations from our laboratory have established that constriction of carotid arteriovenous anastomoses in the anaesthetized pig serves as a predictive model for the anti-migraine efficacy of 5-HT-based drugs (10, 25). Moreover, to establish the cardiovascular safety, we studied the effects of donitriptan on cardiac output and its regional distribution to vital organs.
Materials and methods
General
After an overnight fast, 36 pigs (Yorkshire × landrace; 10–15 kg) were anaesthetized with azaperone (140 mg, i.m.), midazolam hydrochloride (7.5 mg, i.m.) and pentobarbitone sodium (600 mg, i.v.). The animals were intubated and connected to a respirator (BEAR 2E, BeMeds AG, Baar, Switzerland) for intermittent positive pressure ventilation with a mixture of room air and oxygen. Respiratory rate, tidal volume and oxygen supply were adjusted to keep arterial blood gas values within physiological limits (pH: 7.35–7.48; pCO2: 35–48 mmHg; pO2: 100–120 mmHg). Anaesthesia was maintained with a continuous i.v. infusion of pentobarbitone sodium at 12–20 mg kg−1 h−1. It may be noted that with this anaesthetic regimen, arteriovenous anastomotic blood flow is considerably higher than that in pigs in a conscious state or under thiopentone anaesthesia (26), thereby producing one of the putative features of migraine, i.e. dilatation of carotid arteriovenous anastomoses (25, 27).
Heart rate was measured with a tachograph (CRW, Erasmus University, Rotterdam, The Netherlands) triggered by electrocardiographic signals. Catheters were placed in the inferior vena cava via the left femoral vein for the administration of drugs and fluids, and in the aortic arch via the left femoral artery for the measurement of arterial blood pressure (Combitrans disposable pressure transducer; Braun, Melsungen, Germany) and the withdrawal of arterial blood for determining blood gases (ABL-510, Radiometer, Copenhagen, Denmark). Body temperature was kept at about 37°C and the animals were continuously infused with saline to compensate for fluid losses during the experiment.
In the case of the carotid artery experiments, both common carotid arteries, external jugular veins and vagosympathetic trunks were identified. The vagosympathetic trunks were cut between two ligatures in order to avoid reflex-mediated cardiovascular changes. Subsequently, the right common carotid artery was dissected free and a needle was inserted against the direction of blood flow for the administration and uniform mixing of radioactive microspheres. Another catheter was placed in the right external jugular vein for the withdrawal of venous blood samples. Blood flow was measured in the right common carotid artery with a flow probe (internal diameter: 2.5 mm) connected to a sine-wave electromagnetic flow meter (Transflow 601-system, Skalar, Delft, The Netherlands).
In the case of the cardiac output experiments, a 6F Swan-Ganz thermodilution catheter (Braun Melsungen AG, Melsungen, Germany) was introduced into the pulmonary artery via the right femoral vein to measure cardiac output. Another catheter, connected to a pressure transducer (Combitrans disposable pressure transducer; Braun, Melsungen, Germany), was guided through the left carotid artery into the left ventricle for the injection of radioactive microspheres. The presence of the tip of the catheter in the left ventricle was confirmed by the observation of the sudden switch from an arterial to a ventricular pressure profile. Lastly, a catheter was placed into the right femoral artery and connected to a Harvard pump for the withdrawal of reference blood samples during the injection of radioactive microspheres.
Heart rate, systolic, diastolic and mean arterial blood pressure as well as carotid blood flow were continuously monitored on a polygraph (CRW, Erasmus University, Rotterdam, The Netherlands). Vascular conductance was calculated by dividing blood flow or cardiac output (ml/min) by mean arterial blood pressure (mmHg), multiplied by hundred and expressed as 10−2 ml/min mmHg−1. Stroke volume (cardiac output dividing by heart rate) and nutrient cardiac output (cardiac output minus lung blood flow, i.e. mainly total arteriovenous shunting) were calculated.
Distribution of carotid blood flow
The distribution of common carotid blood flow was determined with 15.5 ± 0.1 (SD) µm diameter microspheres labelled with either 141Ce, 113Sn, 103Ru, 95Nb or 46Sc (NEN Dupont, Boston, USA). For each measurement, a suspension of about 200 000 microspheres, labelled with one of the isotopes, was mixed and injected into the carotid artery. At the end of the experiment, the animal was killed, using an overdose of pentobarbital, and the heart, kidneys, lungs and the different cranial tissues were dissected out, weighed and put in vials. The radioactivity in these vials was counted for 5 min in a γ-scintillation counter (Minaxi autogamma 5000, Packard Instruments, Downers Grove, IL, USA), using suitable windows for discriminating the different isotopes (141Ce: 120–167, KeV, 113Sn: 355–435 KeV, 103Ru: 450–548 KeV, 95Nb: 706–829 KeV and 46Sc: 830–965 KeV). All data were processed by a set of specially designed programs (28), using a personal computer.
The fraction of carotid blood flow distributed to different tissues was calculated by multiplying the ratio of tissue and total radioactivities by the total common carotid blood flow at the time of the injection of microspheres. As little or no radioactivity is detected in the heart or kidneys, all microspheres trapped in lungs reach lungs from the venous side after escaping via carotid arteriovenous anastomoses. Therefore, the amount of radioactivity in the lungs can be used as an index of the arteriovenous anastomotic fraction of carotid blood flow (28, 29).
Distribution of cardiac output
The distribution of cardiac output was also determined with radioactive microspheres (see above). For each measurement, about 1000 000 microspheres, labelled with one of the isotopes, were injected into the left ventricle. Starting 15 s before microsphere injection and lasting 70 s, a reference arterial blood sample was drawn at the rate of 6 ml/min. Blood loss during this procedure was compensated for by infusing the corresponding volume of Haemaccel. At the end of the experiments, the animals were killed as described above and a number of tissues (lungs, kidneys, heart, stomach, small intestine, spleen, liver, adrenals, brain, skin and skeletal muscles) were dissected out, weighed, put into vials for counting radioactivity (see above). As described by Saxena et al. (28), tissue blood flow was calculated by multiplying the ratio of tissue and reference blood sample radioactivity by the blood withdrawal rate (6 ml/min). Radioactivity in the lungs mainly represents peripheral arteriovenous anastomotic blood flow (non-nutrient part of the cardiac output), although a small part (1–1.5% of cardiac output) is derived from the bronchial arteries (30).
Experimental protocol
After a stabilization period of about 1 h, the animals were divided into two groups (n = 18 each) used for either carotid blood flow or cardiac output experiments. The two experimental groups, where an identical experimental protocol was followed, were subdivided into three subgroups (n = 6 each). The first and second subgroups received physiological saline, whereas the third subgroup was pre-treated with 0.5 mg kg−1 of the 5-HT1B/1D receptor antagonist GR127935 (31–33); both were given intravenously over a period of 10 min at a rate of 0.5 ml/min. Ten minutes after the end of these infusions, baseline values of heart rate, mean arterial blood pressure, carotid blood flow or cardiac output and their distributions, as well as arterial and jugular venous blood gases were measured. Then, the first group received four consecutive infusions of vehicle (5 ml of distilled water, containing 40% polyethylene glycol, v/v), whereas the second and third groups received consecutive doses of donitriptan (0.16, 2.5, 40 and 100 µg kg−1). Both vehicle and donitriptan were administered intravenously over a period of 10 min at a rate of 0.5 ml/min, given every 20 min. Ten minutes after the end of each infused dose of vehicle or donitriptan, all haemodynamic variables were again collected.
The Ethics Committee of the Erasmus University Rotterdam dealing with the use of animals in scientific experiments approved the protocol for this investigation.
Data presentation and statistical analysis
All data have been expressed as the mean± SE mean. The significance of changes within one group was evaluated with Duncan's new multiple range test, once an analysis of variance (randomized block design) had revealed that the samples represented different populations (34). The percentage changes from baseline values caused by donitriptan in the GR127935 pre-treated group were compared with those with the corresponding doses of donitriptan in the saline-pre-treated group by using Student's unpaired t-test. Statistical significance was accepted at P < 0.05 (two-tailed).
In the saline pre-treated group, the dose of donitriptan needed to decrease baseline values of carotid arteriovenous anastomotic blood flow or vascular conductance by 50% (ED50%) was calculated using linear regression analysis.
Drugs
Apart from the anaesthetics, azaperone (Janssen Pharmaceutica, Beerse, Belgium), midazolam hydrochloride (Hoffmann La Roche b.v., Mijdrecht, The Netherlands) and pentobarbitone sodium (Apharma, Arnhem, The Netherlands), the compounds used in this study were: donitriptan (4-[4-[2-[3-(2-aminoethyl)-1H-indol-5-yloxy]-acetyl]-piperazin-1-yl] benzonitrile mesylate; F12640 and GR127935 (N-[methoxy-3-(4-methyl-1-piperazinyl) phenyl]-2'-methyl-4′-(5-methyl-1,2,4-oxadiazol-3-yl) [1,1-biphenyl]-4-carboxamide hydrochloride (both from Centre de Recherche Pierre Fabre, Castres, France), haemaccel® (Hoechst Marion Roussel b.v., Hoevelaken, The Netherlands) and heparin sodium (Leo Pharmaceutical Products, Weesp, The Netherlands) for preventing clotting of the catheters. Donitriptan was dissolved in distilled water, containing 40% polyethylene glycol (v/v).
Results
Carotid blood flow experiments
Systemic haemodynamics and arterio-jugular venous oxygen saturation difference
As shown in Table 2, donitriptan as well as its vehicle did not change heart rate. There was a slight decrease in mean arterial blood pressure after the second and third infusions of vehicle. In the saline pre-treated group donitriptan did not change mean arterial blood pressure, but there was a moderate hypotensive response (− 7.4 ± 1.9%) following the highest dose of the drug in the GR127935 pre-treated animals. Donitriptan produced a dose-dependent increase in arterio-jugular venous oxygen saturation difference in both saline and GR127935 pre-treated animals; vehicle was devoid of this effect.
Values of heart rate (HR), mean arterial blood pressure (MAP) and arterio-jugular venous oxygen saturation difference (A−V SO2) at baseline and after 10 min intravenous infusions (0.5 ml/min) of either donitriptan or the corresponding volumes of vehicle (n = 6). The effects of donitriptan were analysed in animals pre-treated with saline (n = 6) or the 5-HT1B/1D receptor antagonist GR127935 (0.5 mg kg−1; n = 6)

All values have been presented as the mean±SE mean.
Carotid haemodynamics
As shown in Figure 2 (absolute values) and Figure 3 (percentage changes from baseline), except for an increase in total carotid vascular conductance after the third infusion and a decrease in arteriovenous anastomotic conductance after the last infusion, vehicle did not produce any changes in carotid haemodynamics. On the other hand, donitriptan dose-dependently decreased total carotid and arteriovenous anastomotic blood flow and concomitant conductances; nutrient blood flow and conductance increased. The dose of donitriptan that was needed to decrease baseline values of arteriovenous anastomotic blood flow and conductance by 50% (ED50%) was found to be 58 ± 27 µg kg−1 (113 ± 52 nmol/kg) and 47 ± 16 µg kg−1 (92 ± 31 nmol/kg), respectively. The maximum changes observed in total carotid, arteriovenous anastomotic and nutrient vascular conductances with the highest dose of donitriptan were −25 ± 3%, −63 ± 3% and + 87 ± 21%, respectively. After treatment with GR127935, donitriptan-induced decreases in the total carotid and arteriovenous anastomotic blood flows and vascular conductances were significantly less; the maximum decreases in total carotid and arteriovenous anastomotic vascular conductances were −12 ± 2% and −35 ± 5%, respectively, (see Figs 2 and 3). Compared with saline-treated animals, GR127935 appeared to reduce donitriptan-induced increases in nutrient blood flow and vascular conductance, but statistical significance (based on percentage changes from the baseline in the two groups) was achieved only in the case of blood flow increase observed with 40 µg kg−1 of donitriptan.

Values of total carotid, arteriovenous anastomotic (AVA) and nutrient (a) blood flow (ml.min−1) and (b) conductance (10−2 ml.min−1.mmHg) at baseline (□) and after 10 min intravenous infusions (0.5 ml/min) of either donitriptan (0.16, 2.5, 40 and 100 ▪ µg kg−1) or the corresponding volumes of vehicle (5 ml; n = 6). The effects of donitriptan were analysed in animals pre-treated with either saline (n = 6) or GR127935 (0.5 mg kg−1; n = 6). All values are presented as the mean ± SE mean. ∗P < 0.05 vs. baseline. + P < 0.05 vs. response (% change from baseline) by corresponding dose of donitriptan in animals pre-treated with saline.

Percentage changes from baseline values in total carotid, arteriovenous anastomotic (AVA) and nutrient blood flow (upper panels) and conductance (lower panels) after 10 min intravenous infusions (0.5 ml/min) of either donitriptan (0.16, 2.5, 40 and 100 µg kg−1) or the corresponding volumes of vehicle (5 ml; n = 6). The effects of donitriptan were analysed in animals pre-treated with either saline (•n = 6) or GR127935 (□ 0.5 mg kg−1; n = 6). ○ Vehicle. ▪ All values are presented as the mean±SE mean. ∗P < 0.05 vs. baseline. + P < 0.05 vs. response (% change from baseline) by corresponding dose of donitriptan in animals pre-treated with saline.
The distribution of carotid blood flow to the head tissues in the three groups of animals is shown in Fig. 4. Donitriptan produced significant increases in vascular conductance to the skin, ear, skeletal muscle, tongue, bone, fat, salivary gland, dura mater and brain; no change was observed in the eyes. In animals treated with GR127935, these increases were attenuated, but statistical significance (based on percentage changes from the baseline in the two groups) was reached only in skeletal muscle and fat. The corresponding volumes of vehicle did not produce changes in vascular conductance values.

Values of carotid vascular conductance in different cranial tissues at baseline (□) and after 10 min intravenous infusions (0.5 ml/min) of either donitriptan (0.16, 2.5, 40 and ▪ 100 µg kg−1) or the corresponding volumes of vehicle (5 ml; n = 6). The effects of donitriptan were analysed in animals pre-treated with either saline (n = 6) or GR127935 (0.5 mg kg−1; n = 6). All values are presented as the mean± SE mean. ∗P < 0.05 vs. baseline. + P < 0.05 vs. response (% change from baseline) by corresponding dose of donitriptan in animals pre-treated with saline.
Cardiac output experiments
Systemic haemodynamics
The effects of vehicle and donitriptan on systemic haemodynamic variables are presented in Fig. 5. No changes were observed with vehicle. In animals pre-treated with saline, donitriptan did not change heart rate, mean arterial blood pressure, nutrient cardiac output or systemic vascular conductance, but it moderately decreased cardiac output (maximum change: −10 ± 6%) and stroke volume (maximum change: −12 ± 6%). These changes in cardiac output and stroke volume were absent in animals pre-treated with GR127935, where donitriptan did decrease blood pressure by up to 18 ± 3%.

Systemic haemodynamic values of heart rate (HR), mean arterial blood pressure (MAP), cardiac output (CO), nutrient cardiac output (NCO), stroke volume (SV) and systemic vascular conductance (SVC) at baseline □ and after 10 min intravenous infusions (0.5 ml/min) of either donitriptan (0.16, 2.5, 40 and ▪ 100 µg kg−1) or the corresponding volumes of vehicle (5 ml; n = 6). The effects of donitriptan were analysed in animals pre-treated with either saline (n = 6) or GR127935 (0.5 mg kg−1; n = 6). All values are presented as the mean±SE mean. ∗P < 0.05 vs. baseline; + P < 0.05 vs. response (% change from baseline) by corresponding dose of donitriptan in animals pre-treated with saline.
Regional haemodynamics
Apart from a slight decrease in skeletal muscle vascular conductance, vehicle did not change tissue vascular conductances (Table 3). Donitriptan decreased vascular conductance in lungs (maximum change: −75 ± 6%), which was significantly less in GR127935 pre-treated animals (maximum change: −32 ± 13%). Whereas no changes were observed in vascular conductances in the kidneys, heart and portal tissues (liver, spleen, stomach, adrenals and small intestines), donitriptan slightly increased brain, skin and skeletal muscle vascular conductances.
Effects of 10 min intravenous infusions (0.5 ml/min) of either donitriptan or the corresponding volumes of vehicle (n = 6) on regional vascular conductance in anaesthetized pigs. The effects of donitriptan were analysed in animals pre-treated with either saline (n = 6) or the 5-HT1B/1D receptor antagonist GR127935 (0.5 mg kg−1; n = 6)

All values have been presented as the mean±SE mean.
∗ P < 0.05 vs. baseline.
+ P < 0.05 vs. response by corresponding dose of donitriptan in animals pre-treated with saline.
Discussion
John et al. (19, 24) have reported that donitriptan equals 5-HT in potency and efficacy in contracting rabbit isolated saphenous vein and in increasing outward K+-current in guinea-pig trigeminal ganglion cells. Donitriptan also decreases carotid blood flow in anaesthetized pigs and conscious dogs. In both in vitro and in vivo experiments, the responses to donitriptan can be antagonized by GR127935 and, thus, donitriptan shows a high affinity as well as efficacy at 5-HT1B/1D receptors (Table 1).
Heart rate and blood pressure
In contrast to the clinical experience (for references, see 6), sumatriptan as well as other triptans exert small bradycardic and hypotensive responses in anaesthetized pigs (17, 35). Donitriptan was devoid of these systemic effects in saline-treated animals. The exact reason for the difference between donitriptan and other triptans is not clear. However, this difference may be partly related to the slightly higher affinity of donitriptan at the 5-HT2A receptor (Table 1), which, by virtue of its capacity to mediate hypertension, could annul a potential hypotensive effect of donitriptan. This view seems to be supported by the appearance of hypotension with donitriptan in animals treated with GR127935 (see Table 2; Fig. 5), which can moderately attenuate 5-HT2A receptor-mediated hypertension in rats (33).
Carotid haemodynamics
In agreement with earlier findings (19, 24), donitriptan dose-dependently decreased total porcine carotid blood flow and conductance in the present experiments. In addition, we observed that this vasoconstrictor effect of donitriptan, as is the case with other triptans, including sumatriptan, avitriptan and eletriptan (see 36), was exclusively confined to cephalic arteriovenous anastomoses; the total nutrient fraction distributed to the head tissues increased. In this respect donitriptan exhibited the highest potency amongst several triptans that we have examined in this porcine model; the mean±SE mean ED50% (nmol/kg) of donitriptan in decreasing arteriovenous anastomotic vascular conductance (92 ± 31) was lower than that of avitriptan (150 ± 37), sumatriptan (156 ± 54), eletriptan (400 ± 91) or GMC2021 (2317 ± 734) (36). The increase in the arterio-jugular venous oxygen saturation difference with donitriptan confirmed its constriction action on arteriovenous anastomoses.
It is now well recognized that the vasoconstrictor effect of triptans is mediated via 5-HT1B receptors (12, 13, 15–18). Accordingly, in the present experiments, GR127935 (0.5 mg kg−1) antagonized the constriction of cephalic arteriovenous anastomoses elicited by donitriptan. However, unlike sumatriptan, but as observed with some other triptans, for example eletriptan and GMC2021 (36), this dose of GR127935 did not completely abolish the donitriptan-induced arteriovenous constriction, suggesting the involvement of another, yet uncharacterized, receptor to some extent. Indeed, this is also the case for the arteriovenous anastomotic constriction elicited by ergotamine, dihydroergotamine and 5-HT (36, 37). On the other hand, it is possible that higher doses of GR127935 may be needed for a complete blockade of the effects of donitriptan. Unfortunately, the partial agonist property of GR127935 precludes the use of much higher doses of this antagonist in our experiments (33).
Similar to other 5-HT1B/1D agonists, donitriptan produced a dilatation of carotid arterioles (nutrient vascular bed) and, as a result, blood flow and vascular conductance in many cranial tissues increased (see Fig. 4). In view of the partial antagonism by GR127935, this dilatation seems to be at least partly due to the activation of the 5-HT1B receptor, probably mediating endothelium-dependent relaxation in different vascular preparations. Indeed, the 5-HT1B receptor has been located on vascular endothelium (12) and there is some evidence that 5-HT1B/D receptors may mediate endothelium-dependent vasodilatation (38–40). Alternatively or in addition, it may be due to an indirect consequence of the closure of arteriovenous anastomoses.
Cardiac output and regional haemodynamics
In previous studies in anaesthetized pigs, it has been shown that sumatriptan reduces total cardiac output and systemic vascular conductance, including decreases in vascular conductances in the kidneys and spleen (41). A decrease in renal vascular conductance by sumatriptan has also been reported in anaesthetized dogs (42). In our study, no decrease in blood flows to or vascular conductances in vital organs (brain, heart, kidneys and portal tissues) were observed with donitriptan; in fact there was a small increase in vascular conductance in the skin, skeletal muscle and brain. Donitriptan was also devoid of any effect on the total systemic vascular conductance, but cardiac output decreased. However, as was the case with carotid blood flow, the decrease in cardiac output was also entirely in the non-nutrient part, i.e. lung blood flow, which mainly reflects peripheral arteriovenous shunting (29). Indeed, the nutrient fraction of cardiac output (blood distributed to peripheral tissues) remained unchanged (Fig. 5).
Therapeutic implications
Our study shows that donitriptan selectively constricts porcine carotid arteriovenous anastomoses, with a higher potency compared with several other triptans. As constriction of cephalic arteriovenous anastomoses is of high predictive value for anti-migraine activity (6), donitriptan should be effective in the treatment of migraine. The drug does not decrease blood flows to vital organs, including the heart, brain and kidneys; sumatriptan can decrease renal blood flow in dogs (42). Lastly, it may be mentioned here that donitriptan has little affinity at the 5-HT1F receptor, which, in view of the effects of the 5-HT1F receptor agonist LY334370 (43, 44), may be responsible for side-effects, such as somnolence, asthenia, numbness and paresthesia.
Footnotes
Acknowledgements
This study was supported by Erasmus Pharma B.V., Rotterdam, The Netherlands, on the basis of an agreement with Centre de Recherche, Pierre Fabre S.A., 17 Avenue Jean Moulin, F 81106 Castres Cédex, France.
