Abstract
It has recently been shown that the α-adrenoceptors mediating vasoconstriction of porcine carotid arteriovenous anastomoses resemble both α1- and α2-adrenoceptors, but no attempt was made to identify the specific subtypes (α1A, α1B and α1D) involved. Therefore, the present study was designed to elucidate the specific subtype(s) of α1-adrenoceptors involved in the above response, using the α1-adrenoceptor agonist phenylephrine and α1-adrenoceptor antagonists 5-methylurapidil (α1A), L-765 314 (α1B) and BMY 7378 (α1D). Ten-minute intracarotid infusions of phenylephrine (1, 3 and 10 μgkg−1.min−1) induced a dose-dependent decrease in total carotid and arteriovenous anastomotic conductance, accompanied by a small tachycardia. These carotid vascular effects were abolished by L-765 314 (1000 μgkg−1; i.v.), while these responses were only attenuated by 5-methylurapidil (1000 μgkg−1; i.v.), and BMY 7378 (1000 μgkg−1; i.v.). Furthermore, intravenous bolus injections of phenylephrine (3 and 10 μgkg−1) produced a dose-dependent vasopressor response, which was only affected by 1000 μgkg−1 of 5-methylurapidil, while the other antagonists were ineffective. These results, coupled to the binding affinities of the above antagonists at the different α1-adrenoceptors, suggest that both α1A- and α1B-adrenoceptors mediate constriction of carotid arteriovenous anastomoses in anaesthetized pigs. In view of the less ubiquitous nature of α1B- compared to α1A-adrenoceptors, the development of potent and selective α1B-adrenoceptor agonists may prove to be important for the treatment of migraine.
Introduction
Although the pathophysiology of migraine has not yet been completely unravelled, there is little doubt that dilatation of large cephalic arteries and, possibly, arteriovenous anastomoses is involved in the headache phase of migraine (1–4). Indeed, over the years we have shown that two important groups of drugs that are highly effective in the acute treatment of migraine, i.e. the triptans and ergot alkaloids, potently constrict carotid arteriovenous anastomoses (4–6). While the carotid vasoconstrictor effect of sumatriptan, as well as some other triptans, is mediated by the 5-HT1B receptor (4, 6, 7), the ergot-induced vasoconstriction involves, in addition to 5-HT1B receptors (8), also α-adrenoceptors (9).
There are several reasons to believe that α-adrenoceptors may regulate vascular tone of carotid arteriovenous anastomoses, providing a potential avenue for the development of new antimigraine drugs. It is well known that α-adrenoceptors play an important role in the regulation of vascular tone and blood pressure (10) and that stimulation of the receptors results in constriction of the isolated carotid artery (11–15). Furthermore, administration of α-adrenoceptor antagonists to conscious pigs as well as pigs under thiopentone anaesthesia results in an increase in arteriovenous anastomotic blood flow (16, 17). More recently, we have shown that both α1- and α2-adrenoceptors mediate the constriction of carotid arteriovenous anastomoses in anaesthetized pigs (18).
The objective of the present study was to elucidate the subtype(s) of α1-adrenoceptors — α1A-, α1B- and α1D-adrenoceptor subtypes (19–22) — involved in the constriction of carotid arteriovenous anastomoses in anaesthetized pigs. For this purpose, we investigated the effects of 5-methylurapidil, L-765 314 and BMY 7378, which are preferential antagonists, respectively, at α1A-, α1B- and α1D-adrenoceptors (see Table 1 for affinity constants) (23–25), on the carotid vasoconstriction induced by the α1-adrenoceptor agonist phenylephrine in a well-defined in vivo animal model predictive for antimigraine activity (5, 26). Unfortunately, selective agonists at the α1-adrenoceptor subtypes are not available.
Binding affinity constants (pKi values) for cloned human α1-adrenoceptor subtypes

Data from
Weinberg et al. (23)
Patane et al. (24)
Goetz et al. (25).
Materials and methods
General
After an overnight fast, 41 domestic pigs (Yorkshire × landrace; female; 10–14 kg) were anaesthetized with azaperone (120 mg, intramuscular, i.m.), midazolam hydrochloride (5 mg, i.m.) and sodium pentobarbital (600 mg, intravenous, i.v.). After tracheal intubation, the animals were connected to a respirator (BEAR 2E, BeMeds AG, Baar, Switzerland) for intermittent positive pressure ventilation with a mixture of room air and oxygen. Respiratory rate, tidal volume and oxygen supply were adjusted to keep arterial blood gas values within physiological limits (pH: 7.35–7.48; pCO2: 35–48 mmHg; pO2: 100–120 mmHg). Anaesthesia was maintained with a continuous i.v. infusion of sodium pentobarbital (20 mgkg−1.h−1). It may be pointed out that this anaesthetic regimen, together with bilateral vagosympathectomy (see below), leads to an increase in heart rate and dilatation of arteriovenous anastomoses due to a loss of parasympathetic and sympathetic tone, respectively. Indeed, basal arteriovenous anastomotic blood flow is considerably higher in sodium pentobarbital-anaesthetized pigs (70–80% of carotid blood flow; present results) than in conscious (< 5% of carotid blood flow; 16) or fentanyl/thiopental anaesthetized (∼19% of carotid blood flow; 17) pigs. A high basal carotid arteriovenous anastomotic blood flow is particularly useful for investigating the effects of drugs that constrict these ‘shunt’ vessels.
A catheter was placed in the inferior vena cava via the left femoral vein for the administration of sodium pentobarbital, vehicle (distilled water) or the antagonists. Another catheter was placed in the aortic arch via the left femoral artery for the measurement of arterial blood pressure (Combitrans disposable pressure transducer; Braun, Melsungen, Germany) and arterial blood withdrawal for the measurement of blood gases (ABL-510; Radiometer, Copenhagen, Denmark). Subsequently, bilateral vagosympathectomy was performed in order to prevent the possible influence via baroreceptor reflexes on phenylephrine-induced carotid vascular responses. Two hub-less needles, each connected to a polyethylene tube, used for the administration of radioactive microspheres and phenylephrine, respectively, were inserted into the right common carotid artery against the direction of blood flow for uniform mixing.
Total common carotid blood flow was measured with a flow probe (internal diameter: 2.5 mm) connected to a sine-wave electromagnetic flow meter (Transflow 601-system, Skalar, Delft, The Netherlands), as described elsewhere (5, 27). Heart rate was measured with a tachograph (CRW, Erasmus University, Rotterdam, The Netherlands) triggered by electrocardiogram signals. Arterial blood pressure, heart rate and total carotid blood flow were continuously monitored on a polygraph (CRW, Erasmus University). During the experiment, body temperature was kept at ∼37 °C and the animal was continuously infused with physiological saline to compensate for fluid losses.
The ethical committee of the Erasmus University Rotterdam, dealing with the use of animals in scientific experiments, approved the protocols followed in this investigation.
Distribution of carotid blood flow
As described in detail previously (27–29), the
distribution of carotid blood flow was determined with 15.5 ± 0.1
μm (

where Itis and Itot denote the tissue and total (i.e. the sum of all tissue) radioactivity, respectively, of each radioisotope and CaBF represents the common carotid artery blood flow at the time of microsphere injection. Since little or no radioactivity was detected in the heart and kidneys, all microspheres trapped in the lungs reached this tissue from the venous side after escaping via carotid arteriovenous anastomoses. Therefore, the amount of radioactivity in the lungs was used as an index of the arteriovenous anastomotic fraction of the total carotid blood flow (5). Vascular conductance (ml/min mmHg−1) was calculated by dividing blood flow (ml/min) by mean arterial blood pressure (mmHg).
Experimental protocol
After a stabilization period of at least 60 min, baseline values of heart rate, mean arterial blood pressure, total carotid blood flow and its distribution into arteriovenous anastomotic and capillary fractions, as well as arterial blood gases, were measured. Thereafter, the animals were divided into seven groups, receiving i.v. infusions (0.5 ml/min for 10 min) of either vehicle (distilled water; n = 8), 5-methylurapidil (300 or 1000 μgkg−1; n = 6 each dose), L-765 314 (300 or 1000 μgkg−1; n = 6 and 3, respectively) or BMY 7378 (300 or 1000 μgkg−1; n = 6 each dose). After a waiting period of 15 min, all variables were reassessed. Subsequently, the animals received cumulative doses of phenylephrine (1, 3 and 10 μgkg−1 min−1) infused into the right common carotid artery (0.1 ml/min for 10 min). All variables were collated again 10 min after the start of each agonist infusion.
At least 90 min after the last microsphere injection, the animals received i.v. bolus injections of phenylephrine (3 and 10 μgkg−1) and peak changes in mean arterial blood pressure were noted.
Data presentation and statistical analysis
All data have been expressed as mean±
Drugs
Apart from the anaesthetics azaperone (Stresnil®; Janssen
Pharmaceuticals, Beerse, Belgium), midazolam hydrochloride
(Dormicum®; Hoffmann La Roche b.v. Mijdrecht, The
Netherlands) and sodium pentobarbital (Apharmo, Arnhem, The Netherlands), the
compounds used in this study were:
All drugs were dissolved in distilled water. A short period of heating was needed
to dissolve 5-methylurapidil (acidified to pH = 6.8–7.0
with 0.1
Results
Baseline values and effect of antagonists per se
Baseline values of haemodynamic variables in anaesthetized pigs (n = 41) were: heart rate (100 ± 2 beats/min), mean arterial blood pressure (93 ± 2 mmHg), total carotid blood flow (123 ± 6 ml/min) and conductance (132 ± 6 10−2 ml/min mmHg−1), arteriovenous anastomotic blood flow (95 ± 6 ml/min) and conductance (102 ± 6 10−2 ml/min mmHg−1) and capillary blood flow (28 ± 2 ml/min) and conductance (30 ± 2 10−2 ml/min mmHg−1). There were no major differences between the baseline values in the different groups of animals (data not shown).
No haemodynamic changes were observed with vehicle or BMY 7378 (data not shown). 5-Methylurapidil (1000 μgkg−1) slightly decreased mean arterial blood pressure (−8 ± 4%) and increased heart rate (9 ± 1%) as well as capillary blood flow (35 ± 4%) and conductance (49 ± 9%). L-765 314 (1000 μgkg−1) decreased mean arterial blood pressure (−9 ± 4%) and increased capillary conductance (27 ± 11%).
Systemic haemodynamic responses to intracarotid infusions of phenylephrine
As shown in Table 2, after treatment with vehicle intracarotid infusions of phenylephrine (1, 3 and 10 μgkg−1.min−1) caused a dose-dependent increase in heart rate by up to 28 ± 5%, without affecting mean arterial blood pressure. This phenylephrine-induced tachycardia was slightly less (maximal increase: 11 ± 3%) after 300 μgkg−1 of 5-methylurapidil (probably due to higher initial value), but was not different after the highest dose of 5-methylurapidil (1000 μgkg−1). In animals treated with either 1000 μgkg−1 of L-765314 or BMY7378, a small decrease in mean arterial blood pressure (9 ± 1 or 8 ± 4%, respectively) was observed with phenylephrine; however, when compared with the corresponding blood pressure change in vehicle-treated animals, statistical significance was not reached.
Changes in heart rate and mean arterial blood pressure induced by 10-min intracarotid infusions of phenylephrine in anaesthetized pigs treated i.v. with either vehicle, 5-methylurapidil, L-765 314 or BMY 7378

Values after treatment with respective antagonist or vehicle. a, P < 0.05 vs. baseline. b, P < 0.05 vs. response (% response from respective baseline) to the corresponding phenylephrine dose in animals treated with vehicle.
Carotid haemodynamic responses to intracarotid infusions of phenylephrine
Absolute values of total carotid, arteriovenous anastomotic and capillary vascular conductances in the different groups of animals before and after intracarotid infusions of phenylephrine are shown in Fig. 1. In animals treated with vehicle, phenylephrine produced a dose-dependent decrease in total carotid conductance by up to 75 ± 4%. Since phenylephrine did not change the vascular conductance in the capillary fraction, the decrease in total carotid conductance was exclusively caused by a decrease in the arteriovenous anastomotic fraction (maximal response: 92 ± 3%).

Effects of 10-min intracarotid infusions of phenylephrine on total
carotid, arteriovenous anastomotic (AVA) and capillary vascular
conductances in anaesthetized pigs treated i.v. with either vehicle
(Veh; n = 8), 5-methylurapidil (5-MU; 300
or 1000 μgkg−1; n
= 6 each dose), L-765314 (L; 300 or 1000
μgkg−1; n
= 6 and 3, respectively) or BMY 7378 (BMY; 300 or 1000
μgkg−1; n
= 6 each dose).  Baseline;
Phenylephrine:
Baseline;
Phenylephrine:  1
μg.kg−1.min−1,
1
μg.kg−1.min−1,
 3
μg.kg−1.min−1,
▪ 10
μg.kg−1.min−1.
∗P < 0.05 vs. baseline (B;
values after treatment); †P <
0.05 vs. response (% response from respective baseline) of the
corresponding phenylephrine dose in vehicle-treated animals.
3
μg.kg−1.min−1,
▪ 10
μg.kg−1.min−1.
∗P < 0.05 vs. baseline (B;
values after treatment); †P <
0.05 vs. response (% response from respective baseline) of the
corresponding phenylephrine dose in vehicle-treated animals.
As shown in Figs 1 and 2, the constrictor effect of phenylephrine on carotid arteriovenous anastomoses was not affected by 300 μgkg−1 of either 5-methylurapidil or BMY 7378, but was significantly attenuated by 300 μgkg−1 of L-765314 and 1000 μgkg−1 of 5-methylurapidil and BMY 7378. Furthermore, after the highest dose of L-765 314, the responses to phenylephrine were clearly abolished and the values did not significantly differ from those before phenylephrine infusion.
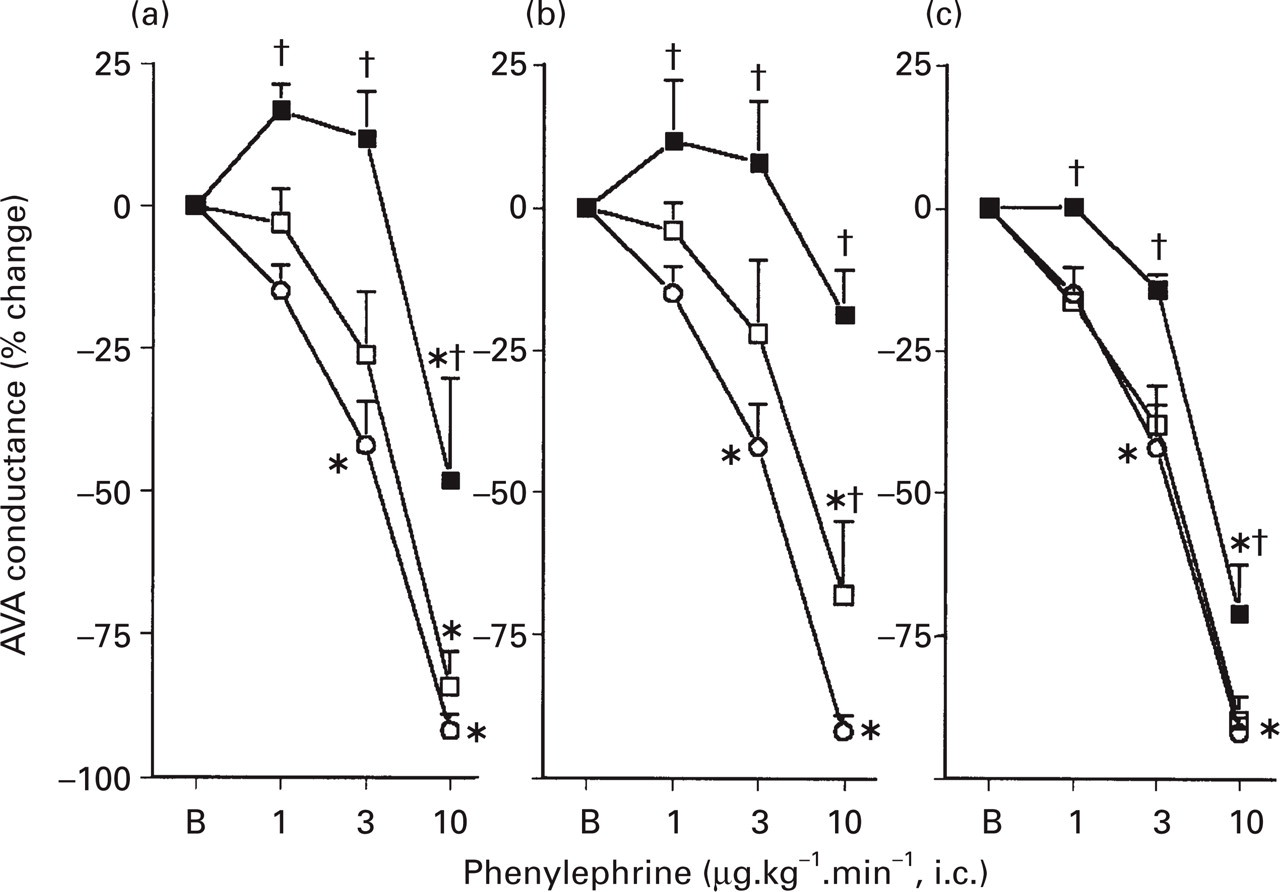
Percentage changes in arteriovenous anastomotic (AVA) conductance induced by 10-min intracarotid infusions of phenylephrine in anaesthetized pigs treated i.v. with either vehicle (○n = 8), (a) 5-methylurapidil (5-MU; 300 □ or 1000 ▪ μg.kg−1; n = 6 each dose); (b) L-765314 (L; 300 □ or 1000 ▪μg.kg−1; n = 6 and 3, respectively); (c) BMY 7378 (BMY; 300 □ or 1000 ▪μg.kg−1; n = 6 each dose; right graph). ∗P < 0.05 vs. baseline (B; values after treatments); †P < 0.05 vs. response (% response from respective baseline) of the corresponding phenylephrine dose in vehicle-treated animals. Note that the control curves are identical in each graph.
Since mean arterial blood pressure was little affected by the intracarotid infusions of phenylephrine, the responses of vascular conductances were qualitatively and quantitatively similar to those of blood flow (data not shown).
Changes in mean arterial blood pressure by i.v. bolus administration of phenylephrine
In vehicle-treated animals, bolus injections of phenylephrine (3 and 10 μgkg−1, i.v) produced a dose-dependent increase in mean arterial blood pressure, yielding peak responses of 28 ± 3 and 51 ± 3%, respectively (Fig. 3). These phenylephrine-induced vasopressor responses were significantly attenuated by 1000 μgkg−1 of 5-methylurapidil (peak responses: 17 ± 3 and 31 ± 4%, respectively), but were not affected by L-765314 (300 or 1000 μgkg−1), BMY 7378 (300 or 1000 μgkg−1) or 300 μgkg−1 of 5-methylurapidil.

Peak changes in mean arterial blood pressure (MAP) induced by bolus injection of phenylephrine (□ 3 or ▪ 10 μg.kg−1, i.v.) in animals treated i.v. with either vehicle (Veh; n = 8), 5-methylurapidil (5-MU; 300 or 1000 μg.kg−1; n = 6 each dose), L-765 314 (L; 300 or 1000 μg.kg−1; n = 6 and 3, respectively) or BMY 7378 (BMY; 300 or 1000 μg.kg−1; n = 6 each dose). ∗P < 0.05 vs. percentage response of the corresponding phenylephrine dose in vehicle-treated animals.
Discussion
General
We have recently shown that both α1- and α2-adrenoceptors mediate constriction of arteriovenous anastomoses within the carotid vascular bed in anaesthetized pigs (18). This conclusion was based on the findings that (i) intracarotid administration of phenylephrine (α1-adrenoceptor agonist) and BHT933 (α2-adrenoceptor agonist) decreased total carotid blood flow exclusively confined to the arteriovenous anastomotic fraction, without affecting mean arterial blood pressure; and (ii) these effects of phenylephrine and BHT933 were selectively antagonized by the α1- and α2-adrenoceptor antagonists, prazosin and rauwolscine, respectively (18). This is also in agreement with recent findings demonstrated in the external carotid vascular bed of anaesthetized dogs (31).
Based on radioligand binding, molecular biology and isolated tissue experiments, it is known that α1-adrenoceptors are a heterogeneous group of receptors, currently subdivided into α1A, α1B and α1D subtypes (21, 32). As reviewed by Vargas and Gorman (10), the α1A-adrenoceptor subtype, which is widely distributed throughout the body and is the major subtype regulating systemic vascular resistance and blood pressure; α1D-adrenoceptors seem to play only a minor role in blood pressure regulation (33). Whereas there is limited information concerning the vascular effects mediated by α1B-adrenoceptors, it has been shown that constriction of the isolated carotid artery of the dog and rabbit mainly resembles the cloned α1B-adrenoceptor (10). However, no information is available on the subtype mediating vasoconstrictor effects within the carotid arterial bed in vivo. Therefore, the objective of the present study was to identify the α1-adrenoceptor subtype(s) that mediate constriction in the carotid vasculature in anaesthetized pigs, with particular emphasis on the arteriovenous anastomotic fraction, which may be of relevance to migraine therapy (1, 5–7)
In recent years, several selective antagonists at α-adrenoceptor subtypes have been developed (Table 1). In the present study, we made use of 5-methylurapidil and BMY 7378, which have frequently been used to characterize α1A- and α1D-adrenoceptors, respectively (Table 1; 22, 25, 32, 34, 35–37). To block α1B-adrenoceptors, we employed L-765 314, which shows a moderate to high selectivity for cloned α1b-adrenoceptors over cloned α1a- or α1d-adrenoceptors (24). Many studies have used the clonidine derivative, chloroethylclonidine, for this purpose (32, 38). However, it is now evident that chloroethylclonidine alkylates several other receptors as well (32, 39, 40).
Systemic and carotid haemodynamic effects of different antagonists
Whereas the vehicle was devoid of any systemic and carotid haemodynamic effects, administration of the α1A-adrenoceptor antagonist 5-methylurapidil produced a small hypotension and tachycardia (Table 2). Similar hypotensive effect was observed earlier with 5-methylurapidil (41) and may be related to either blockade of vascular smooth muscle α1A-adrenoceptors, which play an important role in the maintenance of vascular tone (10) or to its agonist properties at central 5-HT1A receptors (42). Admittedly, we do not have a clear-cut explanation for the slight hypotension and increase in capillary conductance produced by L-765 314, since it has been shown by Piascik et al. (43) that α1B-adrenoceptors play only a minor role in the contraction of peripheral blood vessels in vitro.
Changes in heart rate and mean arterial blood pressure by phenylephrine
Intracarotid infusions of phenylephrine produced only minor systemic haemodynamic responses in vehicle-treated animals. The tachycardia (Table 2), which was also observed with i.v. phenylephrine (data not shown), most likely involves an interaction with β-adrenoceptors (18). Furthermore, i.v. administration of phenylephrine (3 and 10 μgkg−1) induced a dose-dependent increase in blood pressure (Fig. 3), which was antagonized by 100 μgkg−1 of prazosin, but not by 300 μgkg−1 of rauwolscine (Willems et al., unpublished observations). In view of the antagonism of this response by 5-methylurapidil, but not by L-765 314 (α1B-adrenoceptor antagonist) or BMY 7378 (α1D-adrenoceptor antagonist) (Fig. 3), the pressor response to phenylephrine is likely to be mediated by α1A-adrenoceptors. In keeping with the approximately 10-fold higher affinity at the cloned α1a-adrenoceptor displayed by prazosin compared to 5-methylurapidil (Table 1), a 10-fold higher dose of 5-methylurapidil (1000 μgkg−1) was needed to produce antagonism of the phenylephrine-induced vasopressor response. Thus, as previously discussed (10), these results support the role of α1A-adrenoceptors in the increase in peripheral vascular resistance and concomitant hypertensive effect upon activation.
Carotid haemodynamic responses to intracarotid infusions of phenylephrine
As previously reported (18), phenylephrine caused a pronounced and dose-dependent decrease in total carotid conductance, which was exclusively caused by constriction of carotid arteriovenous anastomoses; nutrient vascular conductance was not modified (Fig. 1). These carotid vasoconstrictor responses were not affected by treatment with 300 μgkg−1 of the α1D-adrenoceptor antagonist BMY 7378. This dose of BMY 7378 should be sufficient to block α1D-adrenoceptors in view of comparable affinities of prazosin and BMY 7378 at the cloned human α1d-adrenoceptor (Table 1) and the fact that 100 μgkg−1 of prazosin abolished this response (18). On this basis, the involvement of α1D-adrenoceptors in the cranial vasoconstriction induced by phenylephrine in anaesthetized pigs seems highly unlikely. Nevertheless, as shown in Figs 1 and 2, the higher dose of BMY 7378 (1000 μgkg−1) produced a slight, but significant, attenuation in the phenylephrine-induced total carotid and arteriovenous anastomotic vasoconstriction. Since BMY7378 displays a moderate affinity at α1A- and α1B-adrenoceptors (pKi: 6.6 and 7.2, respectively; Table 1), a non-selective blockade of these receptors (which can mediate carotid vasoconstriction; see below) is a likely explanation. Indeed, in two experiments a combination of 5-methylurapidil (1000 μgkg−1) and BMY 7378 (1000 μgkg−1) did not cause any more attenuation of the phenylephrine-induced arteriovenous constriction than 5-methylurapidil (1000 μgkg−1) alone (see below).
The α1B-adrenoceptor antagonist L-765314 abolished the constriction of carotid arteriovenous anastomoses by phenylephrine, a finding that supports the role of α1B-adrenoceptors in this effect. Interestingly, the α1A-adrenoceptor antagonist, 5-methylurapidil was also able to antagonize the cranial vasoconstrictor effects of phenylephrine, but in contrast to L-765 314, the highest dose of phenylephrine still elicited a 50% decrease in arteriovenous anastomotic conductance (Figs 2 and 3). Thus, at similar doses, L-765314 acted as a more potent antagonist when compared with 5-methylurapidil. The above findings lead us to conclude that both α1A- and α1B-adrenoceptors mediate the phenylephrine-induced constriction of carotid arteriovenous anastomoses, whereas the α1D-adrenoceptor plays a minor role, if any.
Admittedly, a critique of the above conclusion may be that, unlike in vitro studies, the exact concentration of α1-adrenoceptor antagonists at the receptor site can be influenced by pharmacokinetic differences in such an in vivo investigation. However, current techniques do not allow us to study carotid arteriovenous anastomoses in vitro and, to some extent, we have tried to ensure effective antagonist concentration at the receptor site by using as high dose of antagonists as possible. Moreover, we would like to emphasis the fact that selective agonists at the different α1-adrenoceptor subtypes are currently unavailable. Since porcine α1-adrenoceptor subtypes have not yet been cloned, the selectivity of the subtype-selective antagonists (see Table 1) is based on their affinity for human cloned receptors.
Possible clinical implications
Lastly, we would like to consider possible clinical implications of the present results showing that α1A- and α1B-adrenoceptors mediate the constriction of carotid arteriovenous anastomoses in anaesthetized pigs. To date all acutely acting antimigraine agents, such as the triptans and ergot alkaloids, potently constrict carotid arteriovenous anastomoses (44, 45). Moreover, dilatation of these ‘shunt’ vessels may be involved in the pathophysiology of the headache phase of migraine (1, 5, 6). Since a vasoconstrictor effect in this experimental model seems to be highly predictive for antimigraine efficacy, a α1A- (such as A61603, see 46) or α1B-adrenoceptor agonist (which is yet to be developed) should be able to abort migraine headaches. Of the two α1-adrenoceptor subtypes, the α1B-adrenoceptor is an interesting target for future antimigraine drugs, especially when considering that this receptor, unlike the α1A-adrenoceptor, does not seem to be much involved in the constriction of the peripheral blood vessels (10, 43). Indeed, our results suggest that the hypertensive effect produced by intravenous administration of phenylephrine is predominantly mediated via the α1A-, but not α1B-adrenoceptor (Fig. 3). An α1B-adrenoceptor agonist may have a major advantage over the currently available acute antimigraine drugs, which all constrict human isolated coronary artery (47), where, importantly, the α1B-adrenoceptor is not present (48).
In conclusion, the present study shows that both α1A- and α1B-adrenoceptors mediate constriction of porcine carotid arteriovenous anastomoses produced by phenylephrine. Since the α1B-adrenoceptor subtype is not much involved in constriction of the systemic vasculature, a cranioselective vasoconstriction may be achieved using selective α1B-adrenoceptor agonists, which may prove effective in migraine.
Footnotes
Acknowledgements
We would like to thank Dr M.A. Patane (Merck & Co. Inc., USA) for generously providing L-765 314.
