Abstract
Recent studies suggested that genetic factors play a role in cluster headache (CH). However, the type and the number of genes involved in the disease are still unclear. We performed an association study in a cohort of Italian CH patients to evaluate whether a particular allele or genotype of the Clock gene would modify the occurrence and the clinical features of the disease. One hundred and seven CH patients, diagnosed according to the International Classification of Headache Disorders, 2nd Edition, (ICHD-II) criteria, and 210 healthy age, sex and ethnicity-matched controls were genotyped for the 3092 T°C Clock gene polymorphism (also known as 3111 T°C). Phenotype and allele frequencies were similarly distributed in CH patients and controls. The clinical features of the disease were not significantly influenced by different genotypes. In conclusion, our study suggests that the 3092 T°C polymorphism of the Clock gene is unlikely to play an important role in cluster headache.
Introduction
Cluster headache (CH) is an idiopathic syndrome consisting of recurrent brief attacks of sudden, severe, unilateral periorbital pain associated with cranial autonomic symptoms and restlessness. The aetiology of CH is still not completely understood (1). Recently, several studies have suggested that genetic factors play an important role in the disease. In some families, the phenotype is inherited as an autosomal dominant trait (2). Several cases of monozygotic twin pairs concordant for the disease have been reported in the literature (3, 4). Finally, recent epidemiological surveys have demonstrated that, in comparison with the general population, both first- and second-degree relatives of CH patients have a significantly increased risk for the disease (5–7). To date, however, no molecular genetic clues have been identified for CH (8).
The signature feature of CH is its periodicity: the patients exhibit a striking unique circadian and circumannual rhythmicity in headache attacks. It is well known that the hypothalamus governs various body rhythms and several studies have suggested that this structure plays a pivotal role in the pathogenesis of the disease. Neuroendocrine studies have shown that the cyclic secretion of various hormones is altered in CH patients, both in the cluster period and in the remission phase of the illness (9, 10). Recent studies with positron emission tomography and voxel-based morphometry have clearly identified an area in the posterior hypothalamic grey as the primum movens in the pathophysiology of CH attacks (11, 12).
Circadian rhythmicity of biological processes is a fundamental property of all organisms. A number of genes responsible for the maintenance of these rhythms, the so-called ‘clock genes’, have been recently cloned (13). One of these is the Clock gene, which was first identified in mouse and subsequently in a large number of organisms, including humans (14, 15). The human Clock gene is located on 4q12 and is highly conserved in evolution. It encodes the CLOCK protein, an 855-amino acid polypeptide that is essential for the function of the circadian system. In-situ hybridization analysis has demonstrated intense expression of Clock in the suprachiasmatic nucleus of the hypothalamus and in the cerebellum (15). The human Clock gene has been proposed as a possible candidate for disorders affected by alterations of circadian rhythm, including bipolar disorder and schizophrenia (16). Recent studies have shown that a polymorphism (T to C nucleotide substitution) in the 3′ flanking region of the human Clock gene modifies the sleep cycles in healthy subjects and influences circadian mood fluctuations in patients with bipolar depression (17, 18). As regards the clinical characteristics of the disease, the Clock gene is thus an interesting candidate for conferring susceptibility for CH.
To evaluate whether a particular allele or genotype of the Clock gene would modify the occurrence and the clinical features of CH, we performed an association study in a cohort of Italian CH patients.
Methods
Patients
A total of 107 consecutive unrelated patients with CH (85 men, 22 women; mean age ± SD 43.9 ± 12.14 years) were involved in the study. The diagnosis of cluster headache was made according to the International Classification of Headache Disorders, 2nd Edition, (ICHD-II) criteria (19). Ninety-six patients (87%) fulfilled the diagnostic criteria for episodic CH and 14 (13%) for chronic CH. Age at onset and duration of the disease were 25.4 ± 11.7 and 18.8 ± 11.3 years, respectively. Duration of CH attacks was 68.8 ± 38.7 min. The patients underwent an extensive physical and neurological examination. A standardized record of all the clinical characteristics of headache, suitable for computer analysis, was obtained. A group of 210 age and geographically (Northern Italy) matched healthy subjects (159 men, 51 women, mean age ± SD 43.3 ± 11.9 years) was used as controls. The controls were blood donors and were screened by a neurologist specializing in headaches in order to exclude CH and migraine. Both patients and controls were of caucasian origin. Table 1 shows the epidemiological and clinical characteristics of CH patients and healthy controls. Written informed consent was obtained from all participants and the study was approved by the Hospital Ethics Committee.
Genotype distribution and allele frequencies (AF) of the Clock gene 3092 T→C polymorphism

CH, Cluster headache.
Genetic analysis
Genomic DNA was extracted from whole blood samples using the QIAamp® DNA Mini Kit (Qiagen S.p.A, Milan, Italy). Cases and controls were genotyped for the nucleotide substitution T→C in position 3092 of the DNA sequence (the same polymorphism is also known as 3111 T→C). The single nucleotide polymorphism (SNP rs 1801260) is located in the 3′ flanking region of the human Clock gene on chromosome 4 (NCBI RefSeq NM004898.2). Polymerase chain reaction (PCR) was performed with the following primers: F: 3′-TCCAGCAGTTTCATGAGATGC-5′; R: 3′-GAG GTCATTTCATAGCTGAGC-5′.
PCR reactions were performed in a final volume of 50 µl, with 90 ng of genomic DNA, 0.4 U of Taq Gold DNA polymerase (Applied Biosystems, Monza, Italy), 250 n
Statistics
The Hardy–Weinberg equilibrium was verified for all tested populations. Statistical analyses were performed using Genepop version 3.4 (http://wbiomed.curtin.edu.au/genepop) and SigmaStat version 1.0 (Jandel Corp., San Rafael, CA, USA). χ2 test and Fisher's exact test were used to compare allele (AF) and genotype frequencies (GF) between cases and controls. According to recent guidelines for genetic association studies in neurological disorders, the level of statistical significance was taken at P < 0.01 (20).
Results
Genotype (GF) and allele (AF) frequencies of the 3092 T→C polymorphisms of the Clock gene are shown in Table 1. AF and GF in our control group were remarkably similar to what has been reported in other European American populations (21). χ2 comparison of AF and GF showed no significant differences between cases and controls (χ2 = 0.03; P = 0.87 and χ2 = 0.43; P = 0.80, respectively). Multiple comparisons according to different genotypes (T/T + T/C vs. CC, T/T + C/C vs. T/C, C/C + C/T vs. T/T) showed no significant differences (data not shown).
Finally, no significant difference between episodic vs. chronic CH was found (GF: χ2 = 2.25; P = 0.32; AF: χ2 = 1.89; P = 0.17).
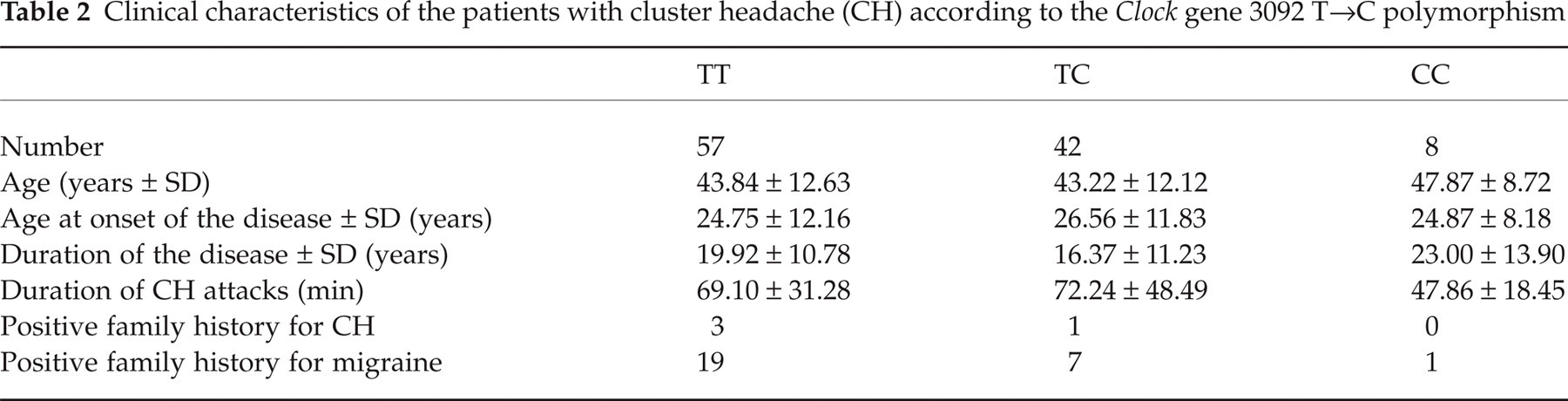
Table 2 compares the clinical characteristics of CH patients according to different 3092 T→C Clock gene genotypes. Multiple comparisons showed no significant difference between these subgroups.
Clinical characteristics of the patients with cluster headache (CH) according to the Clock gene 3092 T→C polymorphism
Discussion
The clock genes regulate the circadian and seasonal rhythms in lower organisms and in humans and are therefore interesting candidates in molecular genetic studies of CH (8, 21). In our study, we evaluated the association between the 3092 T→C polymorphism of the Clock gene and CH in a population of Italian patients and we found no significant association. Phenotype and allele frequencies were similarly distributed in patients and controls. No significant difference between episodic and chronic CH patients was found. Finally, the different genotypes of the Clock gene do not seem to modify significantly the main clinical features of the disease. Thus, our data do not support a significant role of this polymorphism in conferring genetic susceptibility to CH.
To our knowledge, this is the first study to evaluate the association between a polymorphism of the Clock gene and CH. Genetic association studies are becoming increasingly frequent in headache literature and they are considered particularly useful in deciphering the genetic basis of complex diseases such as CH. However, association studies are liable to several biases such as phenotypic definition of the disease, adequate sample size of cases and controls and population stratification. In our study there were several safeguards regarding these issues. The diagnosis of CH relies on the IHCD-II criteria that are unambiguous and precise. We increased the statistical power of our study, enlarging the number of controls and setting the level of statistical significance at P < 0.01. Finally, in our control group, allele and genotypes frequencies of the 3092 T→C Clock gene polymorphism were similar to those reported previously in an another Italian population (18). It is therefore likely that cases and controls arose from the same homogeneous population. However, considering the limits of genetic association studies, additional data from different populations are needed to exclude definitively a role for the Clock gene in CH.
Previous investigations of the molecular genetics of CH examined the possible association of the disease with the CACNA1A gene and with the NOS genes. Sjostrand et al. (22) performed an association analysis of two polymorphisms of the CACNA1A gene in 75 CH patients and 108 matched controls. Genotype and allele frequencies were similarly distributed in patients and controls. A haplotype study of a CH family, followed by mutation analysis, found no evidence of an involvement of the same gene in the disease (23). A Swedish association study investigated several polymorphisms in the genes regulating the synthesis of nitric oxide (NOS1, NOS2 and NOS3) and found no significant difference between cases and controls (24). So, at present, these genes are not considered genetic susceptibility factors for the disease.
Considering the paroxymal character and periodicity of CH, despite our first negative results, clock genes remain interesting candidates for conferring genetic susceptibility to the disease. The analysis of these genes may contribute to elucidating the molecular biology of cluster headache.
Footnotes
Acknowledgements
Supported by grants from the Ministry of University and Scientific and Technological Research (MURST) and Regione Piemonte (Italy).
