Abstract
Background
Cluster headache (CH) is well known to show a seasonal predilection; however, the impact of temperature and other meteorological factors on cluster periods (or bouts) has not been established.
Methods
This nationwide survey included 758 patients with episodic CH retrieved from the Taiwan National Health Insurance Research Database from 2005 to 2009. Corresponding meteorological recordings were obtained from the Central Weather Bureau. A case-crossover study design was used to investigate the association between cluster periods and meteorological factors.
Results
A total of 2452 episodes of cluster periods were recorded. The cluster periods were most frequent in the autumn and least frequent in the winter. Seasonal changes from winter to spring and from autumn to winter also increased the frequency of cluster periods. The risk of cluster periods increased when there was a higher mean temperature on event days (odds ratio (OR), 1.014, 95% confidence interval (CI), 1.005–1.023, p = 0.003) or within seven to 56 days. Either an increase or a decrease in temperature (0.05℃/day) following a warm period (mean temperature ≥26℃) was associated with the onset of cluster periods. In contrast, a greater increase in temperature (0.15℃/day) following a cold period (mean temperature < 21℃) was needed to evoke cluster periods. No such associations were found following moderate periods (21℃ ≤mean temperature <26℃).
Discussion
Our study shows that temperature is associated with precipitating or priming cluster periods. The influence depends on the temperature of the preceding periods.
Introduction
Cluster headache (CH) is a primary headache disorder characterized by a unique seasonal rhythmicity. The signature feature of its periodicity suggests involvement of the hypothalamus, the biological clock (1–3). Ekbom (4) first noted the seasonal predilection of CH in spring and autumn, but Kudrow (5) reported that CH attacks were more common in February and June than in August. This discrepancy may be due to variations in weather conditions in different geographic regions; however, the impact of differences in meteorological factors on the onset of cluster periods (or bouts) is not well established (5–7).
A French study of 316 patients with CH, which analyzed data from the Observatory of Migraine and Headaches Database, reported unpleasant weather as a possible additional trigger (6). However, the definition of unpleasant weather was not explored. In our previous hospital-based study, we demonstrated a possible relationship between sunshine duration and the incidence of cluster periods, but a population-based estimate is still lacking (8). Consequently, identifying effects of weather conditions on the occurrence of cluster periods would allow clinicians or patients to manage CH more effectively.
Taiwan is located in both subtropical and tropical regions with diverse climates, which provides an opportunity to investigate the influence of weather changes on patients with episodic CH. We conducted a nationwide, population-based, case-crossover analysis using meteorological data obtained from the Central Weather Bureau of Taiwan. The aims of the present study were to explore the association of weather changes and to identify if weather variables play a role in precipitating CH periods or if they have a priming effect, i.e. if weather conditions on preceding days determine the occurrence of cluster periods (9).
Methods
Data source
The study subjects were selected from Taiwan’s National Health Insurance Research Database (NHIRD). The Bureau of National Health Insurance (BNHI) of Taiwan reimburses necessary medical claims of all enrollees and the data began to be released in electronic form under the NHIRD project in 1995. Since then, various extracted datasets have been available to researchers, and hundreds of published studies have used the NHIRD as their basis. The NHIRD contains claims data on disease diagnoses coded by the International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) and treatment procedures. In this study, we excerpted data from a specially ordered dataset that included all claims information from patients who were diagnosed with migraine or CH (ICD-9-CM, 2001 edition, codes 346.x) from January 2005 to December 2009. We used the admission and outpatient visit database to obtain information on the patient characteristics of the sample cohort, including sex, date of birth, date of admission, date of discharge, dates of visits, and up to five discharge diagnoses or three outpatient visit diagnoses. The data files also contained information on patient prescriptions, including the names of prescribed drugs, dosage, duration, and total expenditure. This study was approved by the institutional review board of Taipei Veterans General Hospital (201302033BC).
Meteorological data
Located in the western portion of the Pacific Ocean, Taiwan is a small mountainous island that is 394 km long and 144 km wide at its broadest point. In Taiwan, summer is the warmest months of June, July, and August, and winter is the coldest months of December, January, and February. Two seasons, autumn (September to November) and spring (March to May) are the transition periods between summer and winter (10). The Tropic of Cancer passes through Chiayi and Hualien counties, dividing Taiwan into subtropical (northern Taiwan) and tropical (southern Taiwan) areas (11). Since the weather conditions differ between the subtropical and tropical areas, we collected temperature measurements made at corresponding times from all 14 weather stations of the Central Weather Bureau. These measurements included average daily temperatures, daily maximum temperatures, daily minimum temperatures, and temperature variations (slope of change in daily temperature). Since changes in temperature, rather than an absolute value, might also serve as a precipitating factor for CH, we calculated the regression slope of daily temperature, defined as “temperature variation” in this study, using different durations, i.e. seven days, 14 days, 28 days, and 56 days through a time series. Furthermore, other weather measurements, including average daily relative humidity, average daily barometric pressure, maximum wind speed, and total daily hours of sunshine were obtained from the same weather stations.
Priming effect of temperature variables on cluster periods
To explore the possible priming effects of temperature variables on cluster periods, the average temperature during the 28 days before the analyzed period was stratified by 23.5 (average temperature in Taiwan) ± 2.5℃ (approximately half of the standard deviation of the average temperature in Taiwan) and divided into three periods as follows: warm periods (mean temperature ≥ 26℃), moderate periods (21℃ ≤ mean temperature < 26℃), and cold periods (mean temperature < 21℃).
Study design and patient selection
We conducted a case-crossover study designed to examine the effect of meteorological factors on the occurrence of cluster periods. Each case serves as its own control, i.e. the study is self-matched. In our case-crossover study, the exposure (meteorological condition) at the time just prior to the event (the case period) is compared with the exposures during uneventful reference periods (the control periods). We used the ambidirectional approach to select the control periods (12). Each case had two control periods, i.e. three months before and three months after the date of the clinical visit for headache treatment. Symmetric reference periods were selected to decrease potential biases from linear time trends of temperature exposure (Figure 1) (12). In this study, the distribution of exposure was compared between the case periods and control periods.
Case-crossover study design of association between cluster periods and weather changes.
The CH cohort was identified from our special dataset. The CH cohort comprised all patients who were diagnosed with CH (ICD-9-CM code 346.2x). The CH diagnosis was further validated if it was coded by a neurology specialist and if the patient was prescribed standard prophylactic drugs for CH, e.g. verapamil (13). From January 1, 2005 to December 31, 2009, all outpatient or emergency department visits for headache complaints in the CH cohort were identified. The date of the first ambulatory visit was defined as the beginning of a cluster period. If the interval between two ambulatory visits for CH was less than one month, these two visits were defined as in the same period. The date of the last ambulatory visit was defined as the end of a cluster period. “Chronic CH” was defined as CH persisting for a period longer than one year based on the second edition of the International Classification of Headache Disorders (ICHD-II) (14). Patients with chronic CH were excluded from the analyses in our study.
Statistical analysis
Continuous data were expressed as means ± standard deviations. Pearson χ2 tests were carried out for categorical variables; the independent t test and Mann-Whitney U test were used for parametric and nonparametric continuous variables, respectively. For the case-crossover analyses, a conditional logistic regression model was used to estimate odds ratios (OR) and 95% confidence intervals (CIs). For sensitivity analyses, we also computed ORs using the following time periods: seven days, 14 days, 28 days, and 56 days (Figure 1). Applying the Dunn-Šidák correction for multiple comparisons to minimize compounding type I error, p < 0.0102 was considered to be statistically significant (15). All analyses were conducted using STATA statistical software (version 11.0, Stata Corp.).
Results
Characteristics of the study population
A total of 749,027 incident patients with headache were identified in our five-year study cohort dataset. After excluding patients who were diagnosed with migraine or other headaches (n = 728,919), those who were not diagnosed by neurology specialists or who did not receive standard CH prophylactic drugs (n = 728,142), and those who were diagnosed with chronic CH (n = 19), we enrolled 758 patients with episodic CH for analysis. During the study interval, a total of 2452 episodes of cluster period occurred in these patients. Cluster periods occurred predominantly in males (70.8%) with mean age of 39.3 ± 12.5 years. There were 1537 (62.7%) episodes that occurred in the subtropical areas and 915 (37.3%) that occurred in the tropical areas.
Weather characteristics of the subtropical and tropical areas
Supplement Table 1 summarizes the weather characteristics of Taiwan from 2005 to 2009. Generally, the average daily temperature was 23.5℃, with temperatures ranging from 3.5 to 33.3℃. In the subtropical and tropical areas, the average daily temperatures were 23.1 ± 5.2℃ (range: 3.5–32.4℃) and 24.0 ± 4.9℃ (range: 7.7–33.3℃), respectively. In comparison to the tropical area, the subtropical area is characterized by higher wind speeds and more rainfall days, but by a lower humidity, barometric pressure, and sunshine duration (Supplement Table 1).
Sensitivity analysis for weather variables and the risk of cluster periods
As shown in Figure 2, the incidence of cluster periods exhibited a seasonal pattern, with cluster periods being more common in autumn and less common in winter. Additionally, the incidence increased during the seasonal change from winter to spring. Table 1 summarizes the associations among the meteorological variables and the occurrence of cluster periods. We found positive correlations between average temperature and sunshine duration with the occurrence of cluster periods. In contrast, inverse correlation was observed between rainfall days and the occurrence of cluster periods, indicating that more rainfall days were associated with lower incidence rates of cluster periods. We performed further sensitivity analyses with seven-, 14-, 28-, and 56-day periods as the case and control periods using an ambidirectional approach. Except for sunshine duration, the results were generally consistent for the four different time periods. The temperature variables were the most important weather parameters, and they were highly correlated to other meteorological factors. The multivariable Cox proportional hazards analysis after adjusting the other weather variables showed that cluster periods were still more likely to occur with a higher mean temperature (Supplement Table 2). Thus, we performed further analyses focusing on the influences of temperature variables among different areas and different periods of temperatures.
Relationship between monthly temperature and the incidence of cluster periods in the subtropical (northern Taiwan) and tropical (southern Taiwan) areas from 2005 to 2009. The risk of weather variables on the cluster periods. Per one degree Celsius. OR: odds ratio; CI: confidence interval. bp < 0.0102 was considered to be statistically significant by Dunn-Šidák correction.

Sensitivity analysis for temperature variables and the risk of cluster periods in tropical and subtropical areas
The risk of temperature variable on cluster periods in subtropical and tropical areas.
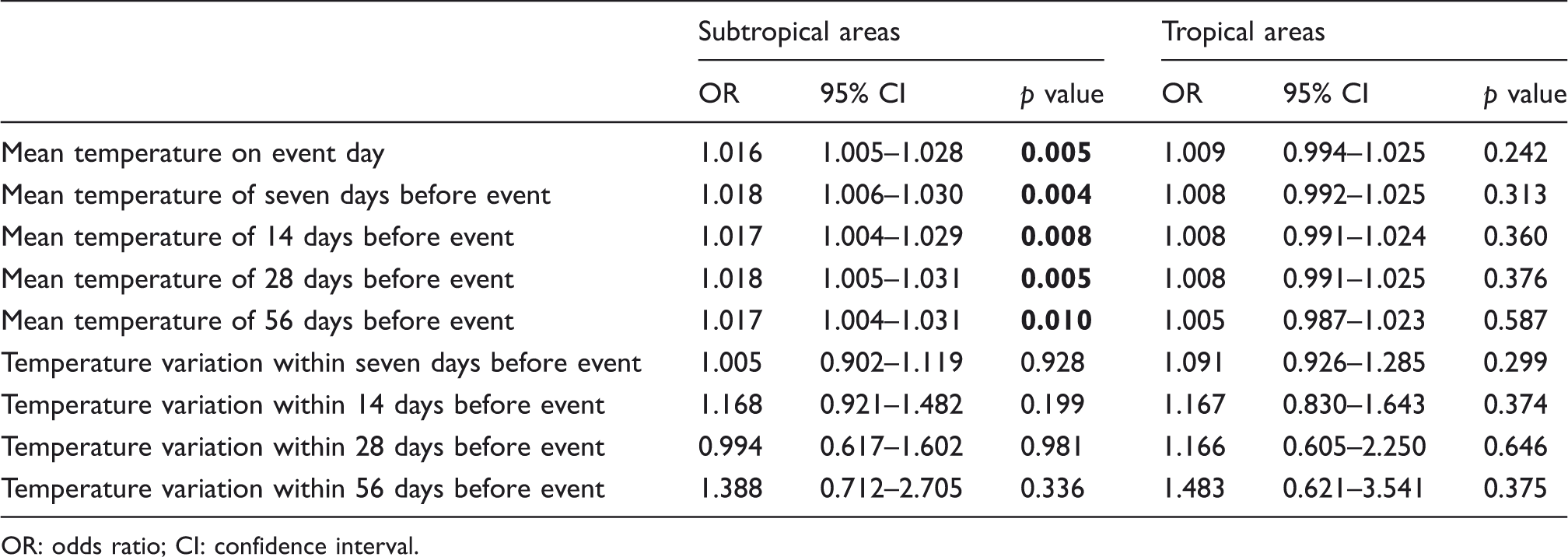
OR: odds ratio; CI: confidence interval.
The priming effects of temperature variables on the occurrence of cluster periods
To clarify the possible priming effects of temperature variables, the associations between three different periods (e.g. warm, moderate, and cold periods) and the risk of cluster periods are illustrated in Figure 3. Following a warm period, a temperature increase or decrease of 0.05℃/day was associated with an increased risk of cluster periods. Following a cold period, we observed a trend that an increase in temperature was associated with an increased risk of cluster periods. The increase reached statistical significance when the slope of the temperature variation increased to more than 0.15℃/day. Of note, no such association was observed following a moderate period.
Comparison of the occurrence of cluster periods during warm, moderate, and cold periods.
Discussion
Our study had three major findings: (1) In Taiwan, cluster periods most commonly occurred in autumn and least commonly occurred in winter. The incidence also increased during the transitions from winter to spring and from spring to summer. (2) Compared to tropical areas, the occurrence of cluster periods in subtropical areas demonstrated a more apparent seasonal variation. (3) Preceding cold or warm periods may have priming effects on the occurrence of cluster periods.
Inconsistent results of previous studies on the association between migraine and weather changes have been noted, possibly due to the variability of weather conditions in different geographic locations (16–19). A population-based study in Croatia reported that weather change was associated with migraine, especially in those with migraine with aura (16). Studies conducted in Germany supported an association between headache incidence and temperature and demonstrated that either a temperature increase or decrease of five degrees Celsius was associated with an increased risk of migraine (18). In contrast, another study in Canada showed no association between emergency visits for migraine and weather conditions (20). However, no previous studies focused on the occurrence of cluster periods and weather changes as in our study. We found that cluster periods occurred more commonly in the autumn than in the winter, a finding that is similar to previous reports (4,21). Notably, the seasonal changes from winter to spring and from spring to summer also triggered cluster periods. In addition, we demonstrate that either an increase or decrease in temperature following a preceding warm period could trigger cluster periods. A greater increase in temperature was needed after a preceding cold period than after a preceding warm period. In contrast, there was no such priming effect observed following the moderate periods, neither warm nor cold. Previously we examined the headache diary data from a sample of migraine subjects in a community-based cohort and found that cold fronts have either a precipitating or a priming effect on migraine occurrence (9). This current study further supports the notion that temperature changes after a cold or warm period, but not after a moderate period, may have priming effects on the occurrence of cluster periods.
The underlying mechanisms for how temperature variables influence or prime the occurrence of cluster periods are unknown. We postulate that the mechanism may involve thermoreceptors in the preoptic area of the hypothalamus (22–24). Warm-sensitive neurons, instead of cold-sensitive neurons, are considered to be the “dominant effector neurons” that control the main thermoregulatory response, such as heat loss, heat retention, and heat production. Moreover, the number of warm-sensitive neurons is much greater than the number of cold-sensitive neurons (25–27). Because of the dominance of warm-sensitive neurons, either a higher mean temperature on event days or changes in temperature following preceding warm periods may be more likely to evoke cluster periods. One study showed that increased body heat could provoke CH. In contrast, a preceding cold period may inhibit the firing rates of preoptic warm-sensitive neurons, which may suppress heat loss responses and then effectively elevate the hypothalamic set-point temperature. Therefore, a greater increase in temperature following cold periods might be needed to precipitate cluster periods.
Our study showed that weather patterns in subtropical and tropical regions affected the occurrence of cluster periods differently. This discrepancy may be partly explained by the influence of temperature variables. The subtropical areas in Taiwan are characterized by a cool winter and a hot summer, ranging from 15℃ in winter to 27℃ in summer (9). Larger temperature fluctuations, resulting in considerable drops or elevations in temperature, provide a chance to prime the hypothalamus for the occurrence of subsequent cluster periods.
Blau and Engel (28) first described a new CH precipitant—increased body heat, either from the environment, a hot bath, central heating, or from exertion. In addition to the hypothalamus, there is growing evidence that the pineal gland may also have a role in the biological regulation of circadian rhythms by producing melatonin (29). Animal models suggested a link of the pineal gland and melatonin with the regulation of core body temperature. In human beings, the nocturnal decline of core body temperature was inversely correlated with the rise of melatonin (30,31). Moreover, it was reported that nocturnal serum melatonin level was decreased during cluster periods (32). Our study found that sunshine duration was positively correlated with the occurrence of cluster periods. Sunshine has been reported to regulate specific hormonal and neurotransmitter levels, including melatonin (33).
Our study has several advantages. First, the primary strength of the case-crossover study design is that the effects of meteorological factors were not confounded by age, sex, smoking, body mass index, underlying chronic disease, medical care-seeking behaviors, drug compliance, or other individual-level characteristics. Second, this study included a large nationwide sample, and applied strict diagnostic criteria and unbiased subject selection. The NHI program offers comprehensive medical care and has a high coverage rate of up to 98.29% (34). The diagnosis of CH required comprehensive review of supporting medical records, examination reports, medicines used, and image studies by neurology specialists. Last, the data collected from all of the weather stations in Taiwan allowed us to determine the real weather conditions to which the CH patients were exposed. The potential limitations of this study should also be addressed. First, an innate limitation of the NHIRD database is that some demographic and clinical data were not available. Second, despite the well-described clinical patterns, diagnoses of CH are often missed or delayed, and the mean diagnostic latency is 8.1 years in Taiwan (8). This diagnostic latency is probably due to the fact that non-neurology specialists have limited knowledge of CH. Furthermore, we excluded CH patients who were diagnosed by non-neurology specialists to ensure the diagnosis were valid. Thus, the occurrence of CH in our study is underestimated (35,36). Additionally, the diagnosis of CH was further validated by the prescription of verapamil. After enrollment, every cluster period during follow-up period was counted even if the patient did not receive verapamil at that time. Therefore, CH patients who never received a prescription of verapamil could not be enrolled in this study. Third, CH patients who experienced cluster periods might not always seek medical help immediately. Therefore, there may be a delay between the onset of symptoms and the recording of the cluster period in our study. Finally, if the patients frequently travelled from subtropical to tropical zones, or vice versa, such information could not be gathered in this NHIRD dataset.
In conclusion, seasonal variation in the occurrence of cluster periods may be related to seasonal changes in meteorological factors. Changes in temperature following preceding warm or cold periods were associated with the occurrence of cluster periods. Warm or cold periods may have a priming effect on cluster periods. Since weather conditions vary dramatically in different areas of the world, further research is needed to investigate whether our findings are generalizable to other localities.
Clinical implications
Cluster periods most commonly occurred in autumn and least commonly occurred in winter. The incidence also increased during the transitions from winter to spring and from spring to summer. Compared to tropical areas, the occurrence of cluster periods in subtropical areas demonstrated a more apparent seasonal variation. Preceding cold or warm periods may have priming effects on the occurrence of cluster periods.
Footnotes
Funding
This work was supported in part by grants from
Conflict of interest
None declared.
Contributorship statement
Lee YJ and Chen YT designed the study, ran the data, performed the statistical analysis, analyzed the data, and helped write the manuscript. Ou SM contributed to the study design and ran the data. Li SY and Yang Albert C. performed analysis and data interpretation. Tang CH contributed to the study design. Wang SJ designed the study, helped write the manuscript and is the guarantor.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
