Abstract
It is known that mutations of CACNA1A, which encodes a neuronal P/Q Ca2+ channel, are present in patients with familial hemiplegic migraine, and possibly in other types of migraine as well. This calcium channel is also involved in neuromuscular transmission. To assess if the single-fibre EMG (SFEMG) method can demonstrate a neuromuscular transmission deficit in migraine, a group of 26 patients with different types of migraine and 20 healthy control subjects were studied. The migraine patients were divided into three groups: 8 patients with migraine without aura (MoA), 12 with migraine with aura excluding visual aura (MA) and 6 with visual aura (VA). A SFEMG of the voluntarily activated extensor digitorum communis muscle was performed. The SFEMG results were normal in the healthy controls and the MoA group (migraine without aura). Slight neuromuscular transmission disturbances were present in 6/12 (50%) of patients with MA and in 1/6 (17%) of patients with VA. We suggest that abnormal neuromuscular transmission detectable by SFEMG may reflect a genetically determined dysfunction of the P/Q Ca2+ channels in a subgroup of migraineurs with aura.
Introduction
Mutations of the CACNA1A gene on chromosome 19p13 were first described in patients with episodic ataxia type 2 and spino-cerebellar ataxia type 6 (1). It has been demonstrated that CACNA1A mutations are present in about 50% of families with hemiplegic migraine (1–3). The T666M mutation is the most frequent CACNA1A mutation in FHM (4–6). Mutations in the CACNA1A gene were also found in patients with sporadic hemiplegic migraine, associated with cerebellar ataxia (4, 6, 7) or cerebral atrophy with mental retardation (8). Recently, Terwindt et al. (7) described a patient with sporadic hemiplegic migraine and a CACNA1A mutation, without cerebellar symptoms. Linkage analyses provided evidence that CACNA1A may also be involved in other types of migraine (9), especially migraine with prolonged aura (10, 11). CACNA1A encodes neuronal voltage-gated P/Q Ca2+ channels, which are located in motor nerve terminals and are responsible for acetylcholine release at the presynaptic membrane. Therefore, their dysfunction may result in impairment of neuromuscular transmission. Antibodies against voltage-gated P/Q calcium channels cause the neuromuscular transmission defect observed in Lambert–Eaton myasthenic syndrome (12–15). A subclinical defect of neuromuscular transmission was demonstrated by single-fibre EMG (SFEMG) in patients suffering from migraine with aura (16) and episodic ataxia type 2 (17, 18). We aimed to look for subclinical impairment of neuromuscular transmission, as a possible indicator of underlying chanellopathy, in different groups of migraineurs, including migraine patients without aura, to confirm previous results.
Material
We studied a group of 26 migraine patients, and 20 healthy age-matched controls. Migraine was diagnosed according to the International Headache Society criteria, 2nd edition (19). The migraine patients were divided into three groups: 8 patients with migraine without aura (MoA), 12 with migraine with aura excluding visual aura (MA) and 6 patients with visual aura (VA).
The MoA group (IHS code 1.1) consisted of 7 women and 1 man with a mean age of 41 years. The MA group (without visual aura) consisted of 8 patients with the hemiplegic form (4 with familial hemiplegic migraine – IHS code 1.2.4; and 4 with sporadic hemiplegic migraine – IHS code 1.2.5), 1 female patient with basilar-type migraine (IHS code 1.2.6), 2 patients with migraine with persistent aura without infarction (IHS code 1.5.3), and 1 female patient with migraine aura without headache – sensory and speech symptoms during aura (IHS code 1.2.3). The mean age in the MA group was 34 years. The VA group consisted of 6 female patients, with a mean age of 40 years. None of the migraine patients suffered from other coexisting headaches or other disorders. Demographic characteristics of the patients are summarized in Table 1.
Demographic characteristics of the migraine patients in the study

n, number of patients; F, female; M, male; IHS, the International Headache Society.
The study was conducted after approval by our university ethics committee and with the informed prior consent of all subjects.
Methods
The SFEMG examination was conducted by an investigator blinded to the clinical diagnosis of the patient (A K-P). SFEMG of the voluntarily activated extensor digitorum communis muscle was conducted as described by Stalberg et al. (20, 21), using a Keypoint electromyograph (Medtronic, Skovlunde, Denmark). In all tested subjects 20 potential pairs were collected, and the mean consecutive difference (MCD), jitter, value was subsequently calculated for each pair. The MCD is the mean of the differences between the intervals between consecutive potential pairs, calculated by the software, and is a standard parameter for assessment of neuromuscular transmission, reflecting the safety factor of the neuromuscular junction (22). The mean MCD value and the number of potential pairs with an MCD exceeding 55 µs or blocking were noted (23).
The results were compared with our laboratory reference values and considered abnormal when the mean MCD value exceeded 33.8 µs, or two or more pairs had an MCD value above 55 µs with or without blocking.
All migraine patients were examined interictally, at least three days after the last episode of headache. The patients were not receiving any drugs for a period of 3 days before the SFEMG examination.
The results were expressed as means and SD for quantitative variables and as proportions for binary data. Proportions were compared by Duncan test with P < 0.05 taken as a cut-off value. Mean values were compared by one-way analysis of covariance. Statistical calculations were carried out using the SAS program.
Results
The SFEMG results are presented in Table 2. The results were within the normal range in the reference group and in all of the patients with migraine without aura. Mild neuromuscular transmission impairment was found in half (6/12) of the patients with hemiplegic migraine, paresthesias and speech disturbances during aura, or persistent aura without infarction. In the group with visual aura, the SFEMG result fell outside the normal range in only one case. All patients showing neuromuscular transmission impairment had only slight SFEMG abnormalities, with mean MCD values just above the upper limit of normal. Although pairs of potentials with an MCD above 55 µs were recorded in 5 patients with MA and 4 patients with VA, no blocking was observed. The individual MCD values were shifted towards the upper limit of normal for a single potential pair in each case (Figs 1 and 2). We did not find statistically significant differences in jitter values between the studied groups, but 50% of SFEMG results of patients with MA were considered abnormal by a blinded examiner (Table 2).

Histogram of MCD (mean values of consecutive differences) of individual potemtial pairs (a) in patients with visual aura and (b) in hemiplegic migraine. Note a shift of MCD values towards the uper limit of normal (the vertical line represents 55 µs.

Single-fibre EMG results. ‘Boxes’ values falling between the 25th and 75th centiles and the ‘whiskers’ extend to the highest and lowest values, excluding outliers. The line across the box indicates the median value.
Results of SFEMG studies in the migraine patients in the study
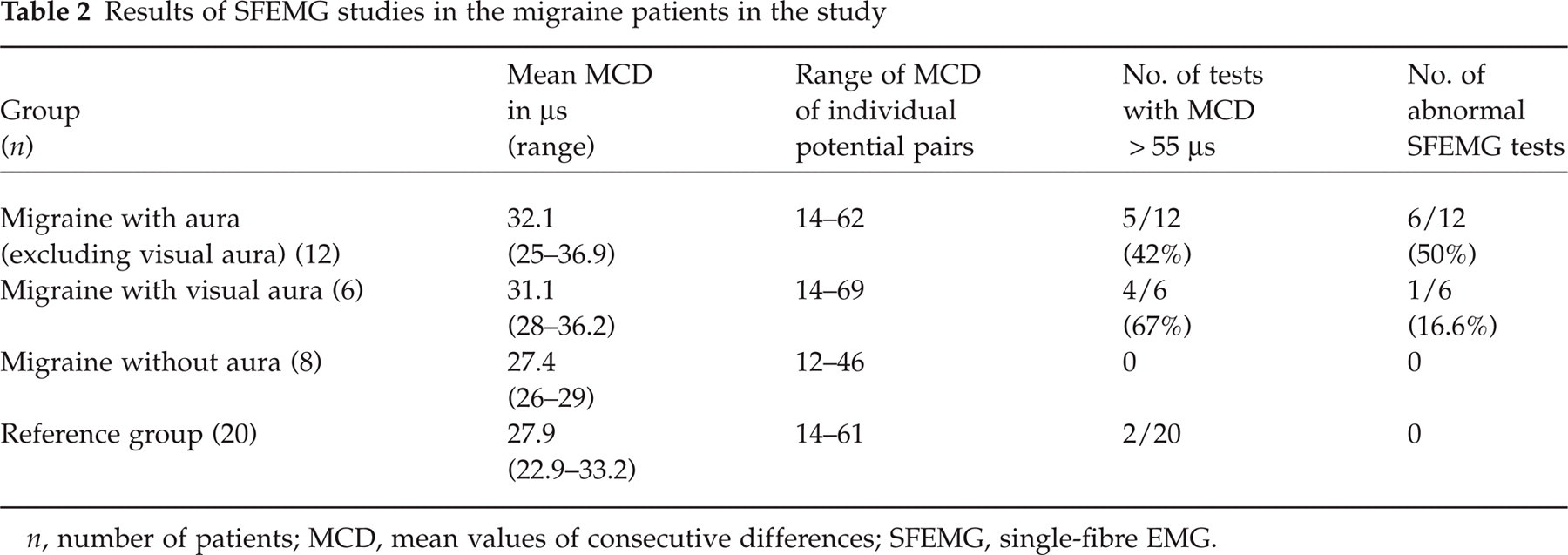
n, number of patients; MCD, mean values of consecutive differences; SFEMG, single-fibre EMG.
Discussion
Our study disclosed a subclinical impairment of neuromuscular transmission in a subgroup of patients with migraine with aura, as observed previously by other authors (10, 16, 18). Interestingly, although the majority of our patients in whom neuromuscular transmission was impaired had hemiplegic migraine, we also found abnormalities in one of the patients with visual aura.
The SFEMG abnormalities present in all of our migraineurs were mild. They were manifested as a shift of individual MCD values towards the upper limit of normal. Although in a few potential pairs the MCD values exceeded 55 µs, there was no blocking. Such findings could be interpreted as a mild, yet generalized reduction in the neuromuscular junction's safety factor. Neuromuscular transmission abnormalities were reported by Ambrosini et al. (16) who studied neuromuscular transmission in patients with migraine with stimulated SFEMG, observing prolonged jitter and blocking. Recently, three patients were described with episodic ataxia type 2, in whom the mean MCD values fell between 40 and 59 µs, with maximal jitter values up to 214 µs, reaching the degree of abnormalities observed in myasthenic patients (17). In the SFEMG study of 12 patients with familial hemiplegic migraine (9 with mutation of CACN1A gene), SFEMG results were abnormal in 2/12 patients and 1/10 controls, and no significant difference in jitter values was found between the patients and the controls, leading to a conclusion that neuromuscular transmission is normal in FHM (24). Our study suggests that neuromuscular transmission abnormalities may be milder then reported previously (16, 25). Therefore, the blinded setting of the SFEMG is recommended for studying migraine patients, as it decreases the possible observation bias. SFEMG with voluntary activation, as employed in our study, excludes the risk of subthreshold stimulation, as the cause of increased jitter or axonal blocking when the SFEMG is studied with axonal stimulation (24). It is possible that the patients with episodic ataxia type 2, hemiplegic migraine and possibly some of the patients with migraine with prolonged aura represent a clinical continuum of CACNA1A mutations, with the FHM patients having milder neuromuscular junction impairment. The SFEMG abnormalities in all of these patients could be attributed to the malfunctioning P/Q Ca2+ channels on presynaptic motor nerve terminals, as these channels are involved in acetylcholine release at the neuromuscular junction (26). Therefore, neuromuscular transmission impairment detected by SFEMG could be helpful in characterizing the phenotype of migraine patients, who are candidates for further genetic testing. The fact that SFEMG abnormalities were limited to patients with migraine with aura may be an additional argument supporting the opinion that migraine with aura and migraine without aura are distinct disorders (27). It can also influence the choice of treatment. Drugs modifying P/Q Ca2+ channels can be useful in the treatment of migraine. Calcium channel blockade with verapamil is beneficial in sporadic and familial hemiplegic migraine (28, 29). Clinical improvement was demonstrated in two sisters with FHM (30) and five nonhemiplegic migraineurs, as well as episodic ataxia type 2 patients treated with acetazolamide (25, 30). Although DNA analysis is necessary to confirm the presence of CACNA1A mutations, SFEMG can be considered a valuable diagnostic tool in patients with migraine with aura.
