Abstract
The aim was to investigate neuromuscular transmission (NMT) by single-fibre EMG (SFEMG) in a large series of patients having migraine with aura (MA) or cluster headache (CH). Recent studies using SFEMG have shown subclinical dysfunction of NMT in MA and CH. Forty-three patients having MA, 51 with CH and 38 healthy control subjects underwent nerve conduction studies, EMG and SFEMG during voluntary contraction of the extensor digitorum communis muscle. Twenty different potential pairs were recorded and individual, mean and total abnormal individual jitter values were calculated. The results obtained from MA patients were compared with those from CH patients. In MA patients, 32 of 860 jitters were abnormally high, whereas 73 of 1020 of the jitters showed this abnormality in CH patients. None of the control subjects, five MA patients (11.6%) and 11 CH patients (21.6%) were designated as having subclinical NMT abnormality. Thus, patients having junction dysfunction were significantly more common in the CH group. The subclinical NMT abnormality shown by SFEMG is more common in CH than in MA. These two primary headache syndromes may have some shared functional abnormality of NMT constituents which is more evident in CH.
Introduction
Impairment of neuromuscular transmission (NMT) has been detected by stimulation single-fibre electromyography (SFEMG) in migraine with aura (MA) (1–4). We have recently reported pronounced abnormalities in NMT by SFEMG in a small group of six patients with episodic cluster headache (ECH), suggesting that CH may share the same abnormality of NMT observed in MA (5).
In this study, we aimed to examine a large group of patients with MA or CH to compare these disorders for NMT abnormality.
Methods
Subjects
Ninety-four patients fulfilling the International Headache Society (IHS) criteria (6) for MA (IHS code 1.2) or CH (IHS code 3.1) (45 women, 49 men; age 17–68 years, mean 35.4 ± 10.3 years) and 38 healthy control subjects (26 women, 12 men; age 16–68 years, mean 46.8 ± 12.2 years) were recruited after their written consent. The patients were chosen from those admitted to the headache out-patient clinic between 2000 and 2004. Patients with sporadic hemiplegic migraine, familial hemiplegic migraine, basilar-type migraine and migraine without aura (MoA) were excluded, to have uniform groups. The local Ethics Committee of Istanbul University approved the study.
Patients' demographic characteristics are summarized in Table 1. There were 43 patients with MA and 51 with CH. Four MA patients had prolonged aura. Among the latter group, 46 had episodic CH and the remaining five had chronic CH. In the group with ECH, the mean frequency of attacks was 1.02 ± 0.43 per year and mean duration of cluster episode was 41 ± 20 per day. Thirty-eight healthy volunteers without a headache disorder comprised the control group. All patients and healthy control subjects underwent detailed neurological examination.
Demographic characteristics of the patients
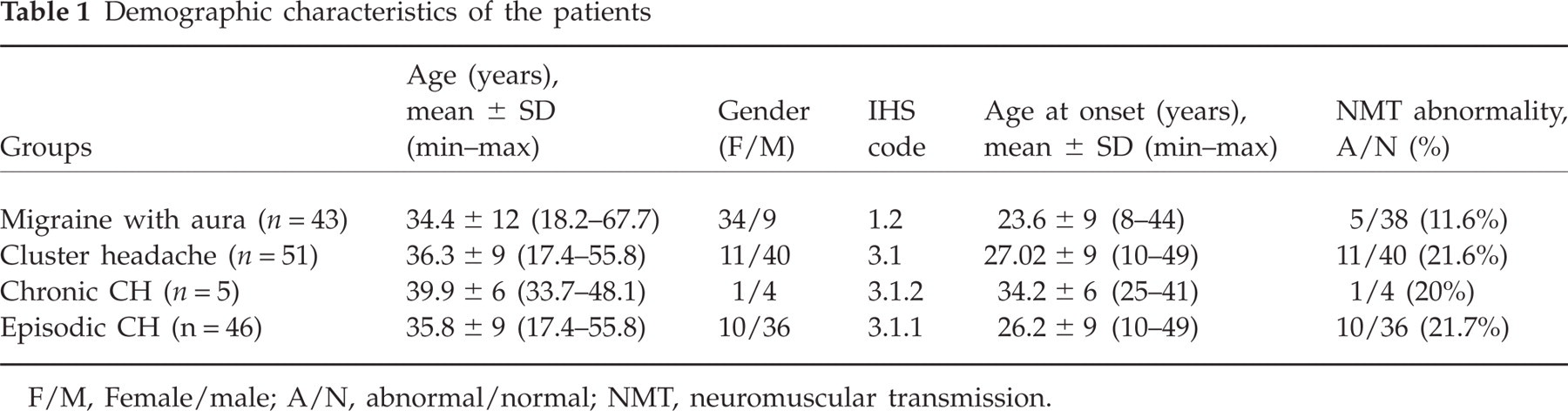
F/M, Female/male; A/N, abnormal/normal; NMT, neuromuscular transmission.
Thirty-one of 46 patients with ECH were in the cluster period at the time of SFEMG investigation. None of the patients was under prophylactic treatment during the SFEMG investigation and they were asked not to use their acute attack treatments 4 h before the investigation. Patients did not use any other drugs which could have a potential effect on the NMT. Of the CH patients, 51% (26/51 patients) used tobacco (in ECH 47.8%) and 47% (24/51 patients) used alcohol within the limits of social drinking (in ECH 43.4%), whereas only 9% of patients with MA used tobacco and 7% used alcohol.
Electrophysiological study
Motor nerve conduction studies included median, ulnar, peroneal and tibial nerves; sensory nerve conduction studies included median, ulnar and sural nerves; and both concentric needle electromyography (EMG) and SFEMG were performed on the extensor digitorum communis (EDC) muscle. During the examination, skin temperature was maintained above 32°C in the upper extremity and above 31°C in the lower extremity. SFEMG was performed during voluntary contraction of the EDC muscle and a single-fibre electrode (22596; Medelec, Old Woking, UK) was used for the SFEMG recording. A Keypoint electromyograph (version 5.3; Physiomed, Denmark) was used for recording, saving data and analysis. The low-cut filter was 500 Hz and the high-cut filter was 10 kHz for the SFEMG recording. Only potentials with a stable shape, a rise time of <0.3 ms and amplitude of >200 µV were accepted for jitter analysis. For each jitter analysis, 50–100 consecutive traces were recorded (7). Twenty different single-fibre potential pairs were recorded in patient groups. In control subjects, 10 (20 subjects), 15 (one subject) or 20 (17 subjects) different single-fibre potential pairs were recorded. The mean consecutive difference (MCD) and mean sorted difference (MSD) were calculated. The calculated MCD was accepted as ‘jitter’. If the MCD/MSD ratio was greater than 1.25, MSD was used instead of MCD as the jitter value. The mean value of 20 jitters and number of individual abnormal jitters exceeding the upper limit of normal (55 µs) were calculated for each subject. Mean jitter and the number of abnormal jitter values were taken into account for the interpretation of the SFEMG data. The range of normal values used for individual and mean jitter results was derived from a multicentre study (8). If a subject had >10% individual abnormally high jitter values out of all individual jitter values (e.g. >2 out of 20), NMT was labelled as ‘abnormal’.
Statistical analysis
Mean value of individual jitter values and the number of abnormally high individual jitter values were calculated for each subject and for each of the three groups (MA, CH and normal groups). The mean values of individual jitter values were compared by one-way
Results
All of the 94 patients and normal subjects had normal neurological examinations and no history of fatigue or weakness suggesting any disorder of NMT. Likewise, all patients and control subjects had normal nerve conduction studies and concentric needle EMG studies of the EDC muscle except three patients with carpal tunnel syndrome in the MA group.
Thirty-two (3.7%) of 860 individual jitter values obtained from MA patients and 73 (7.2%) of 1020 values from CH patients were abnormally high, whereas only six (1%) out of 555 jitter values from normal subjects were abnormal. SFEMG investigations disclosed ‘abnormal’ neuromuscular transmission in 16 (17%) patients (5/43 MA patients, 11.6%, and 11/51 CH patients, 21.6%) (Table 1), whereas none of the control subjects had abnormal NMT. The number of patients with abnormal NMT was significantly more in the CH group than in the MA and normal groups. The MA group had also significantly more patients with NMT than the normal group.
Mean value of individual jitters was 26.5 ± 16.9 µs in MA patients and 29.8 ± 21.3 µs in CH patients, while it was 25.6 ± 10.5 µs in the normal group (Fig. 1). The individual jitter values were significantly higher in CH than in MA patients and normal control subjects, while there was no statistically significant difference between individual jitter values of MA patients and control subjects. Detailed SFEMG results of the patients are summarized in Table 2. None of the four patients with prolonged aura had SFEMG abnormality.

Box plots showing individual jitter values of patients with cluster headache and migraine with aura.
Summary of SFEMG results and statistical test results

A/N, Counts of patients with abnormal/normal NMT; (%), percentage of patients with abnormal NMT; MW, Mann–Whitney unpaired test; UT, unpaired t-test; OW, one-Way
P < 0.05 is accepted for statistical significance.
There were significantly more smokers in the CH group when compared with the MA group. However, the NMT was entirely normal in 22 (84.6%) out of 26 CH patients who were smokers and abnormal in seven of 25 CH patients who did not smoke. Thus, there was no obvious effect of smoking on SFEMG.
Discussion
Although clinically there is no reason to suppose that NMT might be affected in migraine or CH patients, recent studies have shown that some subgroups of migraineurs as well as CH patients have mild subclinical abnormalities of NMT (1, 5, 9). SFEMG is the most sensitive neurophysiological method of assessing the neuromuscular junction in humans. In our preliminary study, three patients with CH showed prominent NMT abnormality and one further patient had borderline dysfunction of NMT on SFEMG (5). Our SFEMG study compared NMT abnormality in MA and CH patients and found that it was more prominent in CH.
Ambrosini et al. described NMT abnormalities by using stimulation SFEMG in 17 patients with MA in a group of 62 migraineurs, measuring variation in the MCD, abnormal jitter values and impulse blocking of single muscle fibre potentials (1). Their study revealed impairment of NMT especially in migraine with prolonged aura (1–3). Interestingly, however, none of four patients with prolonged aura in our present study had NMT abnormality. Domitrz et al. (4) recently studied a small group of 26 patients with different types of migraine (eight patients with MoA, 12 with MA excluding visual aura and six with visual aura) and 20 healthy control subjects. They used SFEMG in the EDC muscle during voluntary contraction and reported normal SFEMG results in healthy controls and the MoA group, but slight NMT disturbances in six of 12 (50%) patients with MA. Although in a few potential pairs the MCD values exceeded 55 µs, values were clustered towards the upper normal limit and there was no blocking. They interpreted these findings as a mild, yet generalized reduction in the neuromuscular junction's safety factor. On the other hand, Ambrosini et al. (1–3) used another technique, stimulation SFEMG, and observed prolonged jitter and blocking. SFEMG during voluntary contraction, as employed in our study, excludes the risk of threshold stimulation as the cause of artificially increased jitter or axonal blocking.
To explain this unexpected subclinical NMT abnormality in migraineurs, it was initially thought that some migraineurs might share similar genetic abnormalities to familial hemiplegic migraine (FHM) patients and have abnormalities of Ca channels or of energy metabolism involving ATP (10, 11). However, in the SFEMG study of 12 patients with FHM (six with mutation of the CACNA1A gene), SFEMG results were abnormal in only two of 12 patients and one of 10 controls, and no significant difference in jitter values was found between patients and controls, leading to a conclusion that neuromuscular transmission is normal in FHM (12). This finding, although in accord with previous observations, casts some doubts on the biological substrate of the neuromuscular junction abnormality found in some migraineurs, suggesting that this is not likely to be due to CACNA1A abnormalities (13). ATP1A2, the second mutated gene in FHM (14–17), encodes the catalytic subunit of Na+/K+ ATPase. Sodium pumping provides the steep Na+ gradient essential for the transport of amino acids (e.g. glutamate) and calcium. Dysfunction of the neuronal sodium channel SCN1A can also cause FHM type 3 (18). Other proteins (e.g. transporters or channels, clock genes, stress response pathways implied in some paroxysmal movement disorders., etc.), but not necessarily CACNA1A or Na+, K+ pump or SCN1A, may also render the nervous system susceptible to MA or CH with differing effects on phenotype in relation to this subclinical NMT abnormality, in the presence of some unknown modifying factors.
The effects of nicotine on the NMT have been discussed in previous studies (19, 20). Nicotine in small doses stimulates the neuromuscular junction directly and facilitates transmission of impulses; in very large doses, it tends to inhibit the neuromuscular junction because of persistent depolarization (acute effect) or desensitization of the receptor site (chronic effect) (21). There is a clear difference between the smoker rates of the MA and CH groups. However, closer examination of the groups showed that there was no predominant effect of smoking on our data. Only four of 11 (36.4%) ofour patients with CH with abnormal NMT were smokers, whereas 7/11 did not smoke.
There may be a few biological explanations for patients who have normal or pathological results of SFEMG with the same diagnosis. The underlying genetic defect may have variable expressivity, and some patients may demonstrate alternative clinical phenotypes with regard to subclinical NMT dysfunction, or they may have a different genetic basis. It seems possible that during cell stress, a normally compensated but marginally operative system (glutamate cycle, channel functions or mitochondrial activity) may fail, causing paroxysmal onset of headache. There are preliminary data indicating that the Drosophila nerve terminal cannot fully compensate physiologically in the regulation of synaptic transmission during larval life for a reduced amount of evoked calcium entry. These findings are relevant, since many deficits in synaptic transmission in various systems are compensated for either physiologically or structurally in the course of development, but not in this case for reduced calcium entry during evoked transmission (22).
Our results might be interpreted as a shared subclinical functional abnormality of NMT constituents of these two primary headache disorders. Some clinical and pathophysiological overlap between these two primary headache disorders has been delineated, and the importance of specific environmental triggers for both of them is well established (23).
In conclusion, NMT defect as a feature of both MA and CH is more prominent in CH.
