Abstract
The aim of this study was to examine the diagnostic spectrum of facial pain and to evaluate the clinical features relevant to the differential diagnosis in a neurological tertiary care centre. This is the first investigation comparing the first with the second edition of the International Classification of Headache Disorders (ICHD-I, ICHD-II) in consecutively referred patients comprising a broad spectrum of disorders without restricting the inclusion to certain diagnoses. Studying 97 consecutive patients referred for facial pain, we found trigeminal neuralgia or other types of cranial neuralgia in 38% and 39% according to ICHD-I and ICHD-II, respectively; persistent idiopathic facial pain was diagnosed in 27% and 21%, respectively. The proportion of patients who could not be classified was 24% in ICHD-I and 29% in ICHD-II. Six per cent of the patients had cluster headache or chronic paroxysmal hemicrania, the remaining 5% had various other disorders. The agreement between ICHD-I and ICHD-II was very good to perfect. In ICHD-II, sensitivity and specificity were similar to ICHD-I, the specificity and negative predictive value were imrpoved in single features of trigeminal neuralgia, but were widely unchanged in persistent idiopathic facial pain. The number of patients who could not be classified was larger in ICHD-II than in ICHD-I. Modifying the diagnostic criteria for different types of facial pain, in particular changes in the criteria of persistent idiopathic facial pain, might be helpful in reducing the number of patients with unclassifiable facial pain.
Keywords
Introduction
Facial pain may be caused by disorders of various facial structures such as the eyes, jaws, nose, sinuses or teeth, it may originate from cranial nerves or it may occur without detectable structural lesions (1, 2). Facial pain in association with acute sinusitis, acute dental disorders or other acute conditions usually represents no major diagnostic problem. However, recurrent or chronic facial pain may be a diagnostic (and therapeutic) challenge due to overlapping symptoms, complex history or equivocal findings in the diagnostic work-up (3–6). This is particularly true for a condition known in clinical practice as ‘atypical facial pain’ and called ‘facial pain not fulfilling criteria in groups 11 and 12’ in the 1988 International Headache Society (IHS) classification (1), which will be cited as first edition of the International Classification of Headache Disorders (ICHD-I) throughout this paper. Both terms are problematic, since it is contradictory to call a characteristic disorder ‘atypical’ and since the ICHD-I term is unsuitable for daily clinical practice. Therefore, the authors appreciate the renaming of the disorder in ‘persistent idiopathic facial pain’ in the second edition of the International Classification of Headache Disorders (ICHD-II) (7).
The majority of studies on pain involving the face focus on certain disorders such as trigeminal neuralgia, persistent idiopathic facial pain, trigeminal autonomic cephalgias or temporomandibular disorder (8–16). Only a few original papers (all from the same author) deal with the differential diagnoses of recurrent or chronic facial pain on a broader basis (17–19), but these studies do not apply ICHD criteria. Considering this lack of information as well as the clinical observation that the definite diagnosis remains uncertain in a considerable number of patients with facial pain, the objectives of this study were: (i) to examine the diagnostic spectrum of facial pain in a neurological tertiary care centre, (ii) to identify those clinical features most relevant to the differential diagnosis, and (iii) to compare the ICHD-I criteria with those of ICHD-II.
Patients and methods
We investigated 97 consecutive patients with recurrent or persistent facial pain referred for neurological investigation by an anaesthesiological pain clinic. The boundaries of the face were defined by the hairline, the frontal fixation of the ear, the posterior and lower edge of the lower jaw and corresponding to the osseous structures: the coronal suture and the external acoustic meatus, the lateral and caudal limitation of the lower jaw (3). All subjects underwent a clinical interview and a clinical neurological examination. The clinical interview covered the time since onset of pain, the duration, frequency, quality, intensity, and localization of pain, the presence or absence of precipitating factors, symptoms between attacks, concomitant autonomic symptoms, periods of remission, and presence or absence of an operation or injury preceding the pain. If necessary, we referred the patients to other specialists such as an otorhinolaryngologist, a maxillofacial surgeon, or a dentist. Plain X-rays, computed tomography, magnetic resonance imaging (MRI) or scintigraphic examinations were performed as required. Initially, one experienced neurologist (C.W.) established the final diagnosis according to ICHD-I (1). After ICHD-II had become available, all cases were re-diagnosed according to these criteria (7).
To study the spectrum of the clinical features we calculated the prevalence rates of the various characteristics in the entire group of patients as well as in subgroups according to ICHD-II. To evaluate the agreement between ICHD-I and ICHD-II we calculated κ coefficients. Furthermore, we separately calculated for ICHD-I and ICHD-II sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of the clinical features of trigeminal neuralgia and persistent idiopathic facial pain. Values ≥ 0.70 were regarded as clinically relevant. For statistical analysis SPSS 11.0 software (SPSS Inc., Chicago, IL, USA) was used.
Results
Diagnostic spectrum
In the total group of 97 patients, four patients had cluster headache, two had chronic paroxysmal hemicrania (CPH), another four had various types of symptomatic facial pain and one patient had external compression headache caused by glasses. In these diagnostic categories ICHD-I diagnoses did not differ from ICHD-II diagnoses (κ = 1, perfect agreement). The diagnoses of cranial neuralgia and persistent idiopathic facial pain did not fully correspond. Of 38 patients diagnosed as having a cranial neuralgia according to ICHD-II, four patients could not be diagnosed according to ICHD-I. Vice versa, three patients with an ICHD-I diagnosis of cranial neuralgia did not fulfil the corresponding ICHD-II criteria (κ = 0.85, very good agreement). In persistent idiopathic facial pain, all of the 20 patients with persistent idiopathic facial pain according to ICHD-II fulfilled ICHD-I criteria as well, but among 26 patients with an ICHD-I diagnosis of persistent idiopathic facial pain, six did not fulfil the ICHD-II criteria (κ = 0.83, very good agreement). The number of patients who could not be classified was higher in ICHD-II (n = 28) than in ICHD-I (n = 23). Detailed clinical characteristics are given in Table 1.
Clinical characteristics and ICHD-II diagnoses in 97 consecutive patients referred for facial pain

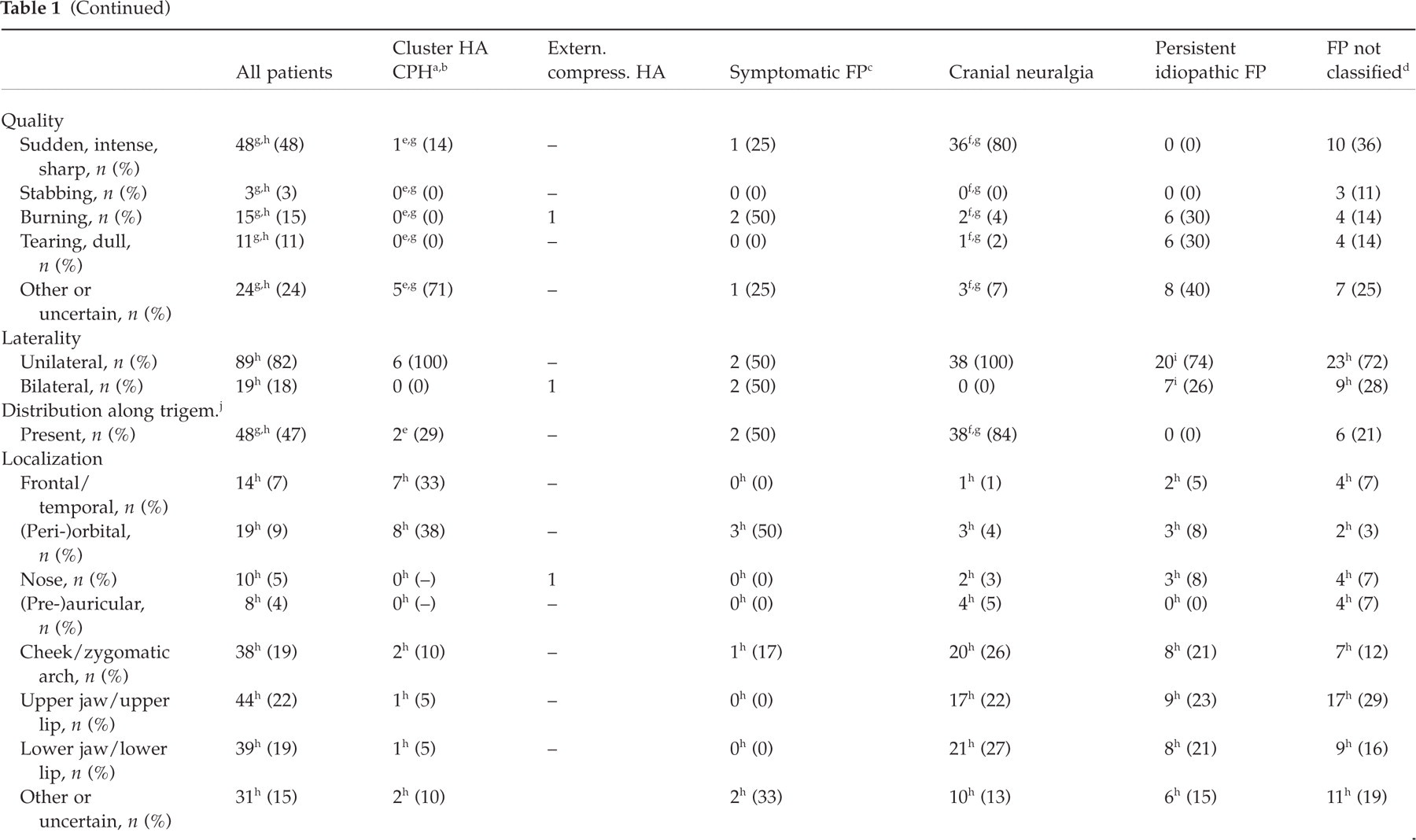

HA, Headache.
CPH, Chronic paroxysmal hemicrania.
FP, Facial pain.
FP not classif., Facial pain not classifiable.
One patient with additional facial pain not classifiable and one patient with additional persistent pain between the attacks.
Seven patients with additional persistent pain between the paroxysms.
Data missing in single patients.
Patients with two or more different characteristics. Therefore number > number of patients.
Seven patients with unilateral pain at onset and additional contralateral pain later.
Distribution along trigem., Distribution along one or more divisions of the trigeminal nerve.
Precipitation, Precipitation from trigger areas or by daily activities.
Persistent pain between attacks not included.
Operation or injury, Pain initiated by operation or injury.
Clinical and laboratory examinations
The clinical examinations by various specialists were normal in 42.1–75% and revealed abnormal findings not related to pain in 12.4–44.8% (Table 2). Only a small group of patients showed abnormal findings related to the facial pain (Table 2). Abnormal neurological findings most often consisted of hypaesthesia or hyperaesthesia. Similar to the clinical findings, only a small proportion of the radiological and laboratory examinations showed abnormal findings related to pain (Table 2).
Findings of clinical and laboratory examinations

Cluster headache and CPH
Among the patients with cluster headache, one patient had coexisting facial pain not classifiable according to ICHD-II (very easily distinguishable from cluster headache) and two other patients described an infraorbital pain maximum initially interpreted as ‘atypical facial pain’ by non-neurologists. One patient with CPH reported additional persistent pain between the attacks. All six patients got pain relief from standard therapies, i.e. sumatriptan, verapamil, lithium, methylprednisolone and indomethacin.
Cranial neuralgia
Among the patients with cranial neuralgia (Table 1), the vast majority had trigeminal neuralgia and two had glossopharyngeal neuralgia. One patient classified as superior laryngeal neuralgia according to ICHD-I did not fulfil the ICHD-II criteria because the local anaesthesia required in the new classification had not been performed. Accordingly, this patient was diagnosed as facial pain not classifiable in ICHD-II. Seven patients described an additional persistent, less severe pain between the attacks. In these patients, cranial MRI showed an aberrant vessel in one patient and a meningioma in another, whereas four patients had no relevant abnormality and one did not undergo MRI.
Four female patients with trigeminal neuralgia reported accompanying autonomic symptoms. Patient 1 had recurrent painful paroxysms for 10 years lasting a few seconds to 1 min. The paroxysms were located on the right side of the face spreading from the upper lip to the medial edge of the orbit. They were precipitated by touching the upper lip, the nose or the medial aspect of the eyebrow and they were accompanied by tearing and mild rhinorrhoea but not by conjunctival injection. Treatment with carbamazepine was moderately effective. Therefore, ganglionic local opioid analgesia (GLOA) was performed with a series of 10 injections of buprenorphine at the superior cervical ganglion. After this treatment the pain attacks were less severe and, interestingly, the autonomic symptoms had ceased completely. The improvement lasted for more than 3 months. Thereafter, pain intensity and frequency began to increase again. Patient 2 reported sudden, intense pain in the first and second branch of the right trigeminal nerve lasting a few seconds, triggered by brushing the teeth or speaking. Recurrence of attacks was relieved, for example, by ceasing to speak. The attacks were accompanied by tearing on the side of the pain. Improvement was achieved by administration of carbamazepine. Patient 3 had typical trigeminal neuralgia for almost 40 years. The attacks lasted up to 1 min and affected the first and second branches of the left side. She reported recent development of two new phenomena. First, she noticed additional persistent pain triggered by recurrent pain attacks. Second, she and her daughter observed autonomic symptoms associated with this pain, i.e. left-sided tearing and marked rhinorrhoea as well as paleness on the left side of the face. Despite the long history, carbamazepine had at least some effect. Patient 4 described a 10-year history of trigeminal neuralgia with attacks lasting less than 1 min and several remission periods. On presentation she reported a change in the pain characteristics with extremely frequent shortlasting stabbing pain in the maxillary division of the trigeminal nerve. These bursts of pain lasted up to 3 h and were associated with ipsilateral tearing and rhinorrhoea. Starting the patient on carbamazepine was followed by complete relief of pain and associated symptoms within 3 days, which was widely maintained over a 20-month follow-up.
The sensitivity, specificity, PPV and NPV of the clinical features of trigeminal neuralgia are shown in Table 3 comparing ICHD-I with ICHD-II. Establishing the diagnosis according to ICHD-I requires four obligatory and four out of five optional criteria, whereas ICHD-II requires four obligatory and one out of two optional criteria only. The sensitivity and NPV were good to excellent for all ICHD-I and ICHD-II criteria. The specificity was ≥ 0.70 in two of nine ICHD-I criteria (duration < 2 min; distribution along divisions of the trigeminal nerve) and four of six ICHD-II criteria (duration < 2 min; distribution along trigeminal nerve; quality intense, superficial, sharp or stabbing; precipitation). The PPV was sufficient in only one ICHD-I criterion (duration < 2 min) and in three ICHD-II criteria (duration < 2 min; distribution along trigeminal nerve; quality intense, superficial, sharp or stabbing). Additional clinical features of trigeminal neuralgia not included in ICHD-I and ICHD-II (i.e. daily occurrence, unilateral localization, absence of autonomic symptoms, presence of remission periods) did not add to the diagnostic accuracy (Table 3).
Sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of clinical features of trigeminal neuralgia: comparison of ICHD-I with ICHD-II

The specificity, PPV and NPV of the criterion ‘pain-free between attacks’ are not given, as a comparison against subjects with non-paroxysmal pain persisting all the day did not seem useful. +, Obligatory ICHD criterion; +/–, facultative ICHD criterion; –, no ICHD criterion.
Persistent idiopathic facial pain
Among the patients suffering from persistent idiopathic facial pain, the majority had a severe pain intensity. Remarkably, seven of 20 patients had bilateral pain from time to time. An operation or a trauma (tooth or jaw operation in four patients; fracture of the upper jaw followed by surgery, maxillary sinus operation and excision of a basalioma in one patient each) preceded the pain in seven patients (Table 1).
The sensitivity, specificity, PPV and NPV of the clinical features of persistent idiopathic facial pain are shown in Table 4. In both ICHD editions, the diagnosis is based on six features summarized in four obligatory criteria. The criteria are almost identical. The only difference refers to the spreading of pain which was moved from criterion B (in ICHD-I) to a footnote (in ICHD-II). Among the ICHD criteria ‘persistence of pain for all or most of the day’ and ‘deep and poor localization’ showed the best diagnostic value. ‘Daily presence’ and ‘pain on one side at onset’ had excellent sensitivity and NPV, but poor specificity and PPV. Among the additional clinical features of persistent idiopathic facial pain given in Table 4, sensitivity and NPV were good to excellent in the vast majority, specificity was ≥ 0.70 in five features and PPV in none.
Sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of clinical features of persistent idiopathic facial pain: comparison of ICHD-I with ICHD-II
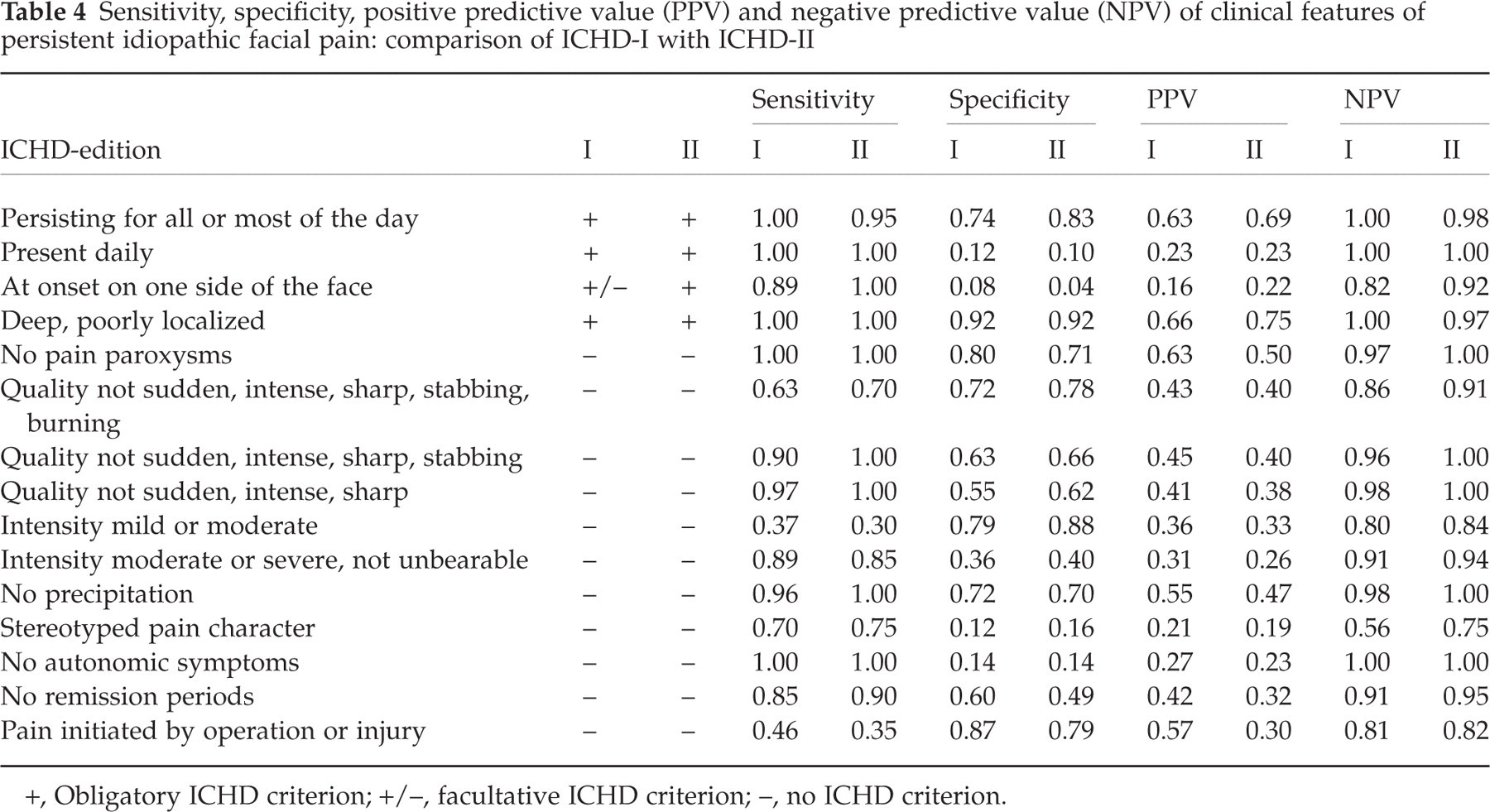
+, Obligatory ICHD criterion; +/–, facultative ICHD criterion; –, no ICHD criterion.
Facial pain not classifiable
A total of 28 patients suffering from facial pain (19 patients with paroxysmal and nine with non-paroxysmal pain) could not be classified according to the ICHD-II criteria. These patients had, for example, attacks lasting 10 or 20 min, occurring three or four times a day with or without precipitation by external stimuli, paroxysmal jaw pain after dental surgery associated with sensory deficits or facial pain spreading to the head (Table 1). But no one was suffering from de-afferentiation pain. Two patients with paroxysmal pain had autonomic symptoms. Patient A had three episodes with left-sided facial pain located in the maxillary (two episodes) or mandibular division (one episode) of the trigeminal nerve. The pain duration was initially less than 2 min, but increased to 20 min in the last episode. The pain was precipitated by brushing the teeth and other external stimuli. At the onset of a pain attack, the patient noticed left-sided tearing and a reddening of the left side of the face. Patient B described a 4-year history of facial pain with pain attacks in the right infraorbital region. The attacks lasted initially a few seconds, later the attacks lasted up to several minutes and one single attack lasted 1 h. Typically the attacks were precipitated by touching the face, shaving, chewing, speaking and head movements, and they were accompanied by marked ipsilateral conjunctival injection, tearing and rhinorrhoea. In an episode of frequent severe attacks the patient in addition developed a constant burning pain in the area of the right ophthalmic nerve. During several remission periods lasting up to 6 months the patient was entirely free of attacks. Neurological examination revealed mild hyperaesthesia in the area of the right ophthalmic nerve and cranial MRI showed an aberrant vessel at the right trigeminal root. Treatment with carbamazepine and GLOA was moderately effective.
Discussion
In this study, including 97 consecutive patients referred for facial pain to a neurological tertiary care centre, 39% of the patients had trigeminal neuralgia or other types of cranial neuralgia and 21% had persistent idiopathic facial pain. Almost 30% of the patients could not be classified according to the criteria of ICHD-II. Of the patients referred for facial pain, 6% had cluster headache or CPH. The remaining 5% of the patients had various other disorders. The prevalence of cluster headache in this study was much smaller and that of trigeminal neuralgia much higher than expected in epidemiological studies. The underrepresentation of cluster headache and overrepresentation of trigeminal neuralgia can be explained plausibly by the study design, the inclusion criterion, the specific referral policy in our hospital and the clinical features of the cluster patients. First, our study was not population based, but clinic based. Second, all patients included were referred from the out-patient pain clinic of the Department of Anaesthesiology with the diagnosis ‘facial pain’. Third, at our hospital patients with headache are usually referred to the headache out-patient clinic of the Neurological Department, whereas many patients with facial pain are referred to the pain clinic of the Department of Anaesthesiology (mostly for evaluation of possible invasive procedures). Fourth, among the patients with cluster headache included in our study, four had atypical features and therefore were not diagnosed correctly up to the visit at our clinic, whereas patients with typical cluster headache probably have a better chance to be diagnosed correctly and therefore were not referred for ‘facial pain’.
The finding that none of our patients had temporomandibular dysfunction (TMD) may be explained by the fact that the vast majority of patients with facial pain are seen by a dentist or maxillofacial surgeon before they are referred to a neurologist. Indeed, some of our patients underwent therapies directed against suspected TMD without success before referral to our department.
Trigeminal neuralgia
The most frequent diagnosis was trigeminal neuralgia, which was found in 36% of the patients. Considering that recurrent or even persistent facial pain is often uncritically classified as trigeminal neuralgia, it seems relevant to point out that 64% of the patients in this study had other types of facial pain.
Regarding the clinical features of trigeminal neuralgia, the occurrence of additional persistent pain and the manifestation of autonomic symptoms may cause difficulties in differential diagnosis. Persistent pain between attacks was found in seven patients with trigeminal neuralgia. In at least four of these patients, a causative lesion was excluded by MRI. This is remarkable, since ICHD-I allowed a persistence of aching between the paroxysms in symptomatic trigeminal neuralgia only (but not in idiopathic). In ICHD-II, a dull background pain is allowed in longstanding cases.
The occurrence of autonomic symptoms in trigeminal neuralgia is well known (4, 19) and requires the differentiation from SUNCT (shortlasting neuralgiform headache with conjunctival injection and tearing) (20, 21). This problem has been discussed recently in several papers. Benoliel and Sharav (5) described lacrimation in six out of 22 patients with trigeminal neuralgia. The authors argued against SUNCT, since the patients with and without lacrimation showed similar pain characteristics and all patients with lacrimation responded to carbamazepine at least in part. Sesso (22) described a patient with a 4-year history of trigeminal neuralgia affecting the ophthalmic division who experienced a change in clinical symptoms consisting of the occurrence of extensive unilateral lacrimation and conjunctival hyperaemia. The degree of these autonomic symptoms increased concomitantly to the increase in intensity and duration of the pain. The initial absence of autonomic symptoms led the author to the conclusion that the disorder has to be classified as trigeminal neuralgia. Goadsby et al. (4) argued that operationally, short-lasting pains of the ophthalmic division of the trigeminal nerve with prominent autonomic (parasympathetic) activation is SUNCT, but that modest cranial parasympathetic activation can occur in any form of first division nociceptive activation, including trigeminal neuralgia. Among our patients with trigeminal neuralgia and autonomic symptoms, all had tearing and three had rhinorrhoea, but none reported prominent conjunctival injection. In our opinion, the absence of prominent conjunctival injection as well as the fact that all four patients responded to carbamazepine are arguments against SUNCT, even in patient 3 reporting marked rhinorrhoea.
Among the clinical characteristics of trigeminal neuralgia, the sensitivity and the NPV were good to excellent in almost all features and they were similar in ICHD-I and ICHD-II. The number of features with a sufficient specificity and PPV, respectively, was larger in ICHD-II (n = 4) than ICHD-I (n = 2). The overall specificity of the criteria of trigeminal neuralgia was improved in ICHD-II by a different definition of the pain characteristics (‘intense, sharp, superficial, stabbing’ instead of ‘sudden, intense, sharp, superficial, stabbing, burning’) as well as by omitting ICHD-I criteria with poor specificity (frontal or facial localization, severe intensity).
Persistent idiopathic facial pain
Twenty-one per cent of our patients had persistent idiopathic facial pain according to ICHD-II. In these patients, there was a marked discrepancy between the severe pain intensity reported by the majority of subjects and the patients’ behaviour during the examination, which often suggested mild or moderate pain only. Some of the clinical features in our patients compare well with those of a previous study performed by Pfaffenrath et al. (15), others differ markedly. The patients’ age, the gender ratio, the daily occurrence and a high proportion of sometimes bilateral pain localization were similar in the two studies, whereas paroxysmal pain and remission periods occurred less often in our patients.
In persistent idiopathic facial pain, ‘persistence of pain for all or most of the day’ and ‘deep and poor localization’ showed the best diagnostic value among the ICHD criteria. ‘Absence of pain paroxysms’ and ‘absence of precipitation’ being not included in ICHD had also a very good diagnostic value. The limited utility of the criterion ‘at onset on one side of the face’ was due to the fact that several patients with otherwise typical persistent idiopathic facial pain reported bilateral pain and it cannot be excluded that they did not recall that pain was unilateral at onset. The finding that seven patients reported an operation or trauma preceding the pain does not allow any firm conclusions, since the events dated back up to 20 years. Prospective studies are required to clarify reliably the relationship between an operation or trauma and persistent idiopathic facial pain.
Facial pain not classifiable
Remarkably, a total of 28 patients (29%) with facial pain did not fulfil any of the ICHD-II criteria for facial pain or headache and their diagnoses remained uncertain. Regarding the two patients with autonomic symptoms, one (patient A) was similar to the four patients with autonomic symptoms in trigeminal neuralgia (with the exception that the pain did not fulfil the criteria of trigeminal neuralgia). The other (patient B), however, is highly interesting, as he illustrates the borderland between (symptomatic) SUNCT and trigeminal neuralgia without fulfilling the diagnostic criteria of these conditions in a strict sense (5, 11, 23).
Conclusion
We reported in detail the clinical features of patients with recurrent or chronic facial pain referred to a neurological tertiary care centre. To our knowledge, this is the first investigation in a clinic-based setting applying ICHD criteria covering a broad spectrum of disorders without restricting the inclusion to certain diagnoses. The agreement between ICHD-I and ICHD-II is very good to perfect. A considerable number of patients, however, cannot be classified according to ICHD and this number is even larger in ICHD-II than in ICHD-I. Modifying the diagnostic criteria for different types of facial pain, in particular changes in the criteria of persistent idiopathic facial pain, might be helpful to reduce the number of patients with unclassifiable facial pain.
Footnotes
Acknowledgements
The authors thank following colleagues from the Medical University of Vienna: Professor Dr A. Spacek, Dr B. Kraft and Dr F. X. Neiger, Department of Anaesthesiology and Intensive Care B (Head: Professor Dr H. G. Kress) for referring consecutively the patients included in this study; and Professor Dr A. Karwautz, Department of Neuropsychiatry of Childhood and Adolescence, for helpful advice in preparing the manuscript.
