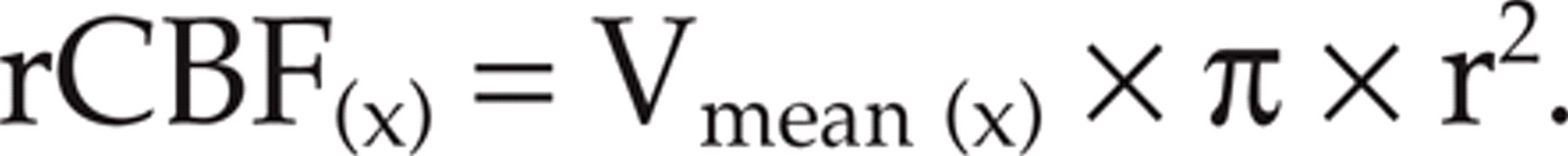Abstract
BIBN4096BS is a CGRP-antagonist effective in the treatment of migraine. Blocking the receptor of a strong vasodilator involves a theoretical risk of causing cerebral vasoconstriction, a probability not previously investigated with BIBN4096BS. Seven healthy volunteers completed this double-blinded placebo-controlled crossover study. The volunteers received randomly 10 min infusions of either placebo, 2.5 mg or 10 mg of BIBN4096BS on 3 separate days. Transcranial Doppler was used to measure the middle cerebral artery blood flow velocity (VMCA); global and regional cerebral blood flow (rCBFMCA) was measured by 133-Xenon inhalation SPECT. The diameter of the temporal and radial artery was measured by highresolution ultrasound. Systemic haemodynamics and partial pressure of CO2 (PetCO2), and adverse events were monitored regularly. BIBN4096BS had no influence on global or regional cerebral blood flow, or on the blood flow velocity in the middle cerebral artery. There was no effect on systemic haemodynamics and adverse events were minor. We conclude that there is no effect of CGRP-receptor blockade on the cerebral or systemic circulation in humans. Circulating CGRP is therefore not likely to exert a vasodilatory activity in the resting state and the use of BIBN4096BS for acute migraine seems to be without risk of cerebral vasoactivity. These data suggest that BIBN4096BS is the first specific antimigraine drug without vasoactive effect.
Introduction
Calcitonin gene-related peptide (CGRP) is probably one of the most potent vasodilators of human arteries (1–4). The vasoactive function of the neuropeptid is mediated through a receptor complex (5). Binding to this complex leads to an intracellular increase in cyclic nucleotides and vasodilatation. CGRP probably plays a protective role against vasospasm following subarachnoidal haemorrhage in both animals and humans (6, 7). CGRP is likely to play a causative role in migraine headache (8, 9).
Peptide fragments of calcitonin gene-related peptide, e.g. CGRP8-37 and [Asp31, Pro34, Phe35] CGRP27-37 have so far been the only available antagonists of CGRP. They have been used in in vitro studies to characterize CGRP function and CGRP-receptor properties. BIBN4096BS is a novel CGRP-antagonist. It has been well characterized in animal studies and can be used safely in humans (10, 11). A phase-2 study gave proof that BIBN4096BS is effective in treating acute migraine headache. BIBN4096BS therefore represents a new principle in acute migraine treatment (11). One potential concern of CGRP antagonism could be cerebral vasoconstriction, if CGRP exerts a tonic influence on the cerebral arteries.
In the present double-blinded placebo-controlled study, we therefore evaluated whether effective CGRP-antagonism achieved by BIBN4096BS would alter the resting tone of cerebral and extracerebral arteries.
Materials and methods
Design and participants
Nine healthy volunteers, five men and four women, were included in this double-blinded placebo-controlled triple crossover study. One subject withdrew her consent and one was excluded due to a knee injury requiring arthroscopy, both before entering the treatment phase of the study. In all seven healthy volunteers completed the study. The participants had an average age of 23.6 years, (range 20–28) and an average weight of 66.9 kg (range 54.8–82).
BIBN4096BS 2.5 mg or 10 mg or placebo (Xylitol 5%) was administered intravenously in a balanced random order over 10 min on 3 trial days separated by at least one week. BIBN4096BS was manufactured and supplied by Boehringer Ingelheim GmbH (BI). BI performed the blinding and randomization of the clinical supplies (ClinPro. version 6, Clinical Systems Inc. USA). The doses used in this study were determined from preliminary data from preclinical safety studies performed by BI (12). The dose of 2.5 mg was selected since this is the minimal effective dose found in the proof of concept study and the possible dose to be taken into future studies (11). Ten mg was the highest dose ever tested with BIBN4096BS and since adverse events was dose dependent; this dose was selected as the high dose to be administered.
A medical history was taken before inclusion. The participants had no present or previous cardiovascular, cerebrovascular, endocrine or neurological disorders, migraine, hypo-or hypertension. Episodic tension type headache with a frequency of ≤4 days/month was the only type of primary headache accepted. On the day of inclusion, a physical and a neurological examination were performed and an ECG and a panel of blood samples were taken.
All participants gave written informed consent before inclusion. The study was approved by the Ethical Committee of Copenhagen County (KA00089gs) and the Danish Medicines Agency (2612–1394) and was conducted according to the Helsinki II declaration and the guidelines for Good Clinical Practice (13).
Cerebral blood flow measurement
Global and regional cerebral blood flow was measured with 133-Xenon inhalation SPECT with a brain-dedicated camera (Ceraspect, DSI, Waltham, MA, USA). The apparatus consists of a stationary annular NaI crystal and a fast rotating collimator system. Each rotation took 10 s, thereby acquiring 1 frame in a 30-frame dynamic protocol of 133-Xenon inhalation, 3 backgrounds, 9 wash-in, 18 wash-out using the Kanno-Lassen algorithm (14). A photoelectric window of 70–100 keV was employed. Thirty-two slices were reconstructed in a 64 × 64 matrix with each pixel measuring 0.33 × 0.33 cm using a Butterworth 1D filter (cut-off 1.5, order 6). The 32 slices were reduced to sets of 8 transaxial slices generated by adding 4 slices together to a total slice thickness of 1.32 cm.
Attenuation correction using the Chang algorithm (µ= 0.05 cm) and correction for nose-artefact were performed. The output for each pixel was the ki-value and flow values were estimated from these using the partition coefficient (λ) of 0.85 (grey matter).
A Datex Normacap 200 (Dameca, Roedovre, Denmark) was used for partial pressure of CO2 (PetCO2) measurements during the CBF acquisitions. A Ceretronix XAS SM 32C (Randers, Denmark) was used for the 133-Xenon administration. Each measurement lasted 5 min. Calculations of flow in the perfusion territories of the major cerebral arteries were performed by fitting standard vascular regions of interest on the five rostral slices at 3.6, 5.0, 6.3, 7.6 and 9 cm above the orbito-meatal line. Flow in the territory of the MCA (rCBFMCA) was calculated as a mean of the left and right side.
Transcranial doppler (TCD) and C-scan
Transcranial Doppler ultrasonography (2 MHz, Multidop X Doppler: DWL, Sipplingen, Germany) was used for the measurement of blood flow velocity. The recordings were done simultaneously and bilaterally as previously described, but with handheld probes (15). Along the middle cerebral artery (MCA), a fixed point was found for the measurement. The fixed point was chosen as close as possible to the bifurcation of the anterior cerebral artery and MCA. The same fix-point was used for each individual and for each recording, for which the signal was optimized. Based upon the envelope curve (the spectral TCD-curve), a time-average mean (Vmean) over approximately four cardiac cycles or four seconds was calculated by the built-in software (TCD7.40x for MDX). The final measure used for each time point was an average of four cycles (VMCA). Simultaneously with the TCD recording, end-tidal PetCO2 (POET; Criticare Systems, Waukesha, WI, USA) was measured using a mask loosely covering the nose and mouth of the subject.
A high-resolution ultrasound scanner, C-scan (Dermascan C, 20 MHz, bandwidth 15 MHz, Hadsund Denmark) (16), was used for the direct measurement of the diameter of the temporal and radial artery. The temporal artery diameter was measured at the frontal branch of the superficial temporal artery; both the temporal and radial recordings were done on the volunteer's left side. To ensure that TCD and C-scan were done in the same place during the repeated measurements on the separated trial days, marks were drawn on the skin. After the last recording on the first trial day, the coordinates of the marks were stored for re-use on the following trial days.
Recording of adverse events (AEs)
The intensity/severity of the AEs were graded mild, moderate or severe and their relationship to study medication was classified as related or not related. The volunteers were questioned for the presence of AEs every 15 min during the study from T0 (baseline) to T240 (end of study day). In between questioning the participants self-reported any changes that they might experience. After discharge the participants recorded AEs every hour until 24 h after the infusion of placebo or BIBN4096BS.
Plasma sampling for BIBN4096BS concentration measurements
Plasma samples of BIBN4096BS were taken at the following time points: TBaseline(− 10), ½, T30, T60 and T180 on each trial day in Vacutainer® EDTA K3 10 ml glasses. Samples were stored on ice for a maximum of 30 min before centrifugation for 10 min (2000 r.p.m) at 4°C. Plasma was stored at −20°C until analysed at Boehringer Ingelheim Pharma GmbH & Co. (KG, Biberach Germany).
Analytical determination of BIBN4096BS in plasma
The procedures were conducted in accordance with current international guidelines (17). BIBN4096BS was analysed by high performance liquid chromatography coupled to tandem mass spectrometry (HPLC-MS/MS) using [D8]-labelled BIBN4096BS as internal standard. The assay comprises sample clean up by automated solid phase extraction in the 96-well plate format. Chromatography was achieved on an analytical C18 reversed-phase HPLC column with gradient elution. The substances were quantified by HPLC-MS/MS in the positive electro spray ionization mode. Assay performance during the study was assessed by back-calculation of calibration standards, tabulation of the standard curve fit function parameters and measurement of quality control samples. No interference of endogenous compounds was observed in human blank plasma and in predose samples. The linear calibration curves for undiluted samples covered a range of plasma concentrations from 0.5 to 500 ng/ml for BIBN4096BS. The validation data document the specificity, accuracy and precision of the HPLC-MS/MS assay employed for the study.
Trial procedures
Participants met at the clinic at 0800h and had abstained from coffee, tea, and caffeine containing beverages, tobacco and medication except oral contraceptives for 8 h prior to the meeting time. The volunteers were headache free. They were placed in supine position. Two intravenous catheters (Optiva∗2, 18G, Johnson & Johnson, Ethicon S.p.A., Italy) were inserted in the cubital veins, one for the infusion of placebo or BIBN4096BS and the second for blood withdrawal. After an adjustment period of at least 30 min, baseline measurements of CBF, VMCA, temporal and radial artery diameter, blood samples, blood pressure (BP), heart rate (HR) and ECG were recorded. At time T0, placebo or BIBN4096BS (2.5 mg or 10 mg) was infused by a time and volume controlled infusion pump (Braun® perfusor, B. Braun Melsong AG, Germany) over 10 min.
All measurements, except BP, HR and CBF were recorded at 15 min intervals for 3 h (the study period). BP and HR were measured every 5 min for the first 30 min and thereafter every 15 min using an automatically inflating cuff (Omega 1400, In vivo Research Laboratories Inc. Copiagne, NY, USA). For safety reasons BP, HR, EKG and adverse events were monitored for an additional hour, after the end of the 3-h study period.
Three SPECT-scans were done: at baseline, at 60 min and at 180 min. The estimated perfusion (rCBFx) in the area of a given artery (x) in the brain is depending on the mean blood flow velocity (Vmean(x)) and the cross-sectional area, π × r2, of the artery. The following equation is valid for the regional cerebral blood flow:
If the CBF is unchanged the following equation is valid:
Δ Diameter is the relative percentage change in diameter; VMCA1 is the mean blood flow velocity before infusion of BIBN4096BS or placebo and VMCA2 the velocity at a specific time point after or during the infusion (18, 19). Global CBF and rCBFMCA were corrected for PetCO2 changes by 2% per mmHg change in PetCO2 (20) and VMCA by e0.034 per mmHg, when required (21).
Statistics
Baseline was calculated as the mean of the measurements of time point T−20 and T−10. Values are presented as means ± SD. P < 0.05 was considered significant. All data processing was performed using SPSS statistical software version 10.0 (Chicago, IL, USA).
For changes over time on each trial day all recordings were analysed by a univariate analysis of variance with the factors time and participant included in the model. Due to the risk of mass significance, data were reduced to five points of interest: baseline, T30, T60, T120 and T180. If a significant change was found, a Dunnett's multiple comparisons test was performed to localize the change. Absolute values were used for the statistical analysis.
For the comparison of differences between the three trial days (placebo, BIBN4096BS 2.5 mg, and BIBN4096BS 10 mg) a univariate analysis of variance was performed, treatment and participant was included in the model. As a summary measure for the univariate analysis, the area under the curve (AUC), calculated on percentage changes of global CBF, rCBFMCA, VMCA, temporal and radial artery diameter and peripheral haemodynamic from baseline, was used.
Results
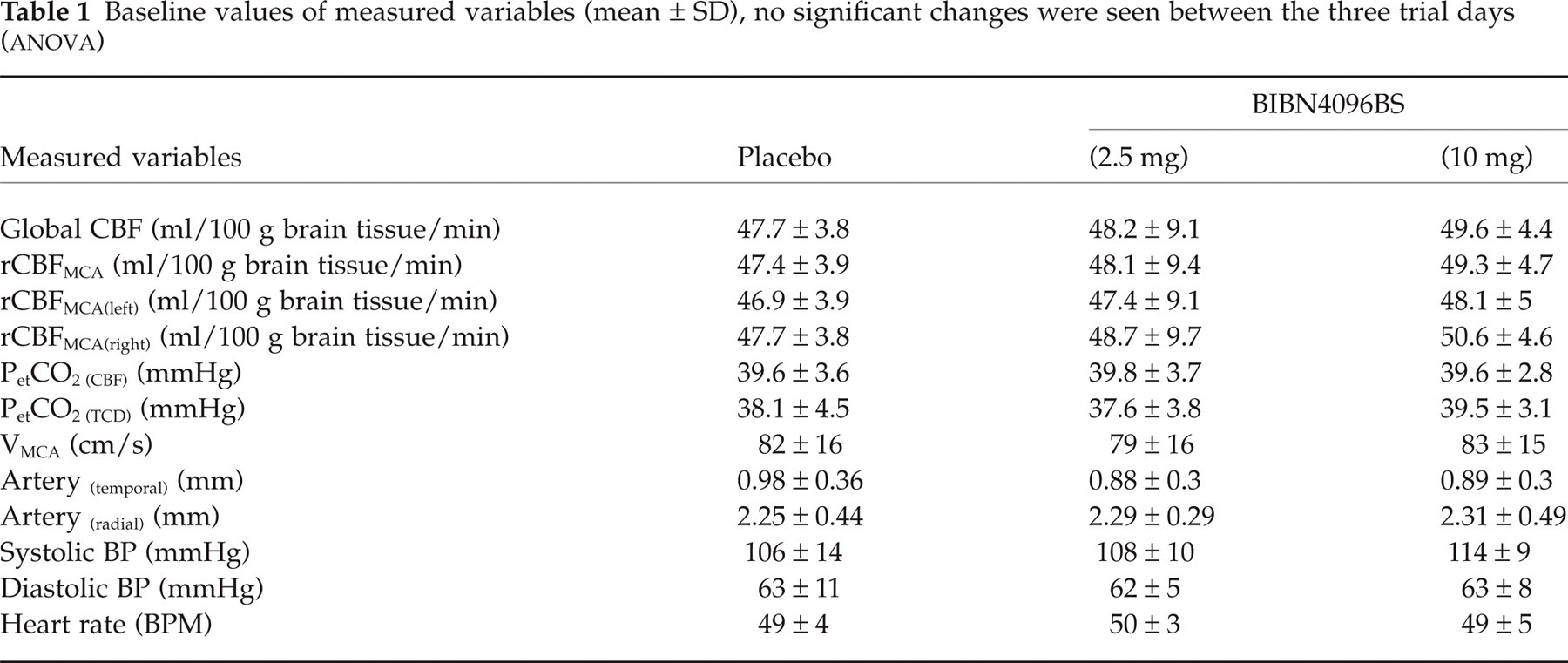
Baseline values of the measurements are summarized in Table 1. No significant difference was found between the baseline values of the three different trial days.
Baseline values of measured variables (mean ± SD), no significant changes were seen between the three trial days (
Haemodynamics
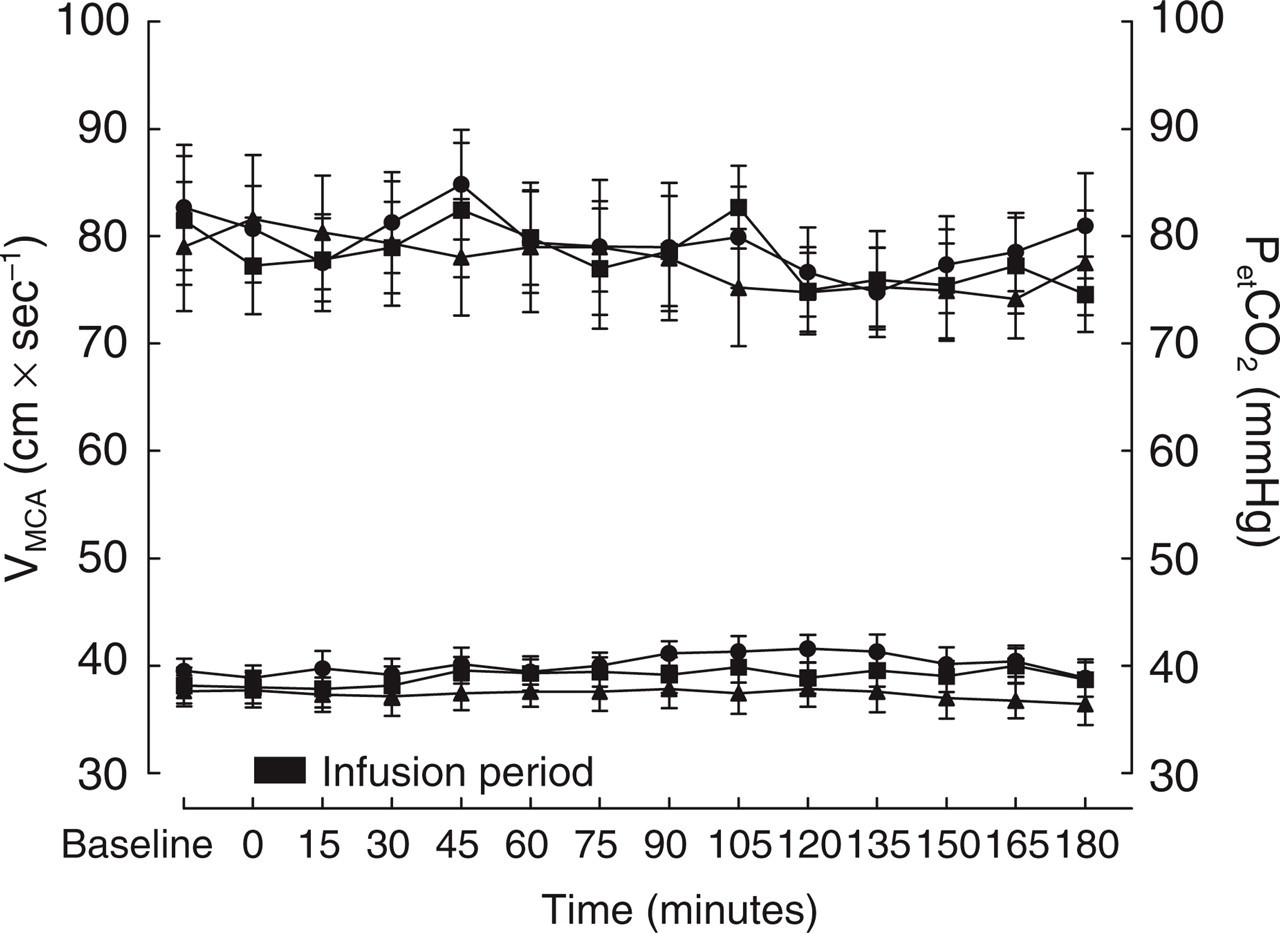
One CBF measurement was not included in the analysis, due to technical problems with the delivery of 133-Xenon (time 60 min on the trial day with administration of BIBN4096BS 10 mg). There was no significant difference in rCBFMCA (Left) (P = 0.74), rCBFMCA (Right) (P = 0.3), rCBFMCA (P = 0.49), or global CBF (P = 0.46) between the 3 treatment groups (placebo, BIBN4096BS 2.5 mg and 10 mg). No significant changes over time were found for either global CBF or rCBFMCA on the three trial days (Fig. 1). PetCO2 showed no significant changes over time on any of the treatment days or between treatment days (P = 0.18).

Measurements of the regional cerebral blood flow in the area supplied by MCA on the three different treatment days are shown (mean ± s.e.m). ▪ Placebo; ▴ BIBN4096BS 2.5 mg; • BIBN4096BS 10 mg.
Since PetCO2 during TCD recordings were performed independently and at separate time points from the PetCO2 during the CBF recordings, the obtained data on PetCO2 were analysed separately. PetCO2 increased significantly over time after BIBN4096BS 10 mg (P = 0.005), the increase was at time T120 and from 39.5 ± 3.1 mmHg to 41.6 ± 3.5 mmHg. There were no significant differences between treatment days (P = 0.29). TCD recordings (VMCA) were analysed with and without correction for PetCO2 changes. Both the uncorrected and corrected VMCA decreased significantly over time on placebo days (P = 0.02 and P = 0.01), the decrease was seen at T120 and T180. No changes were seen with BIBN4096BS 2.5 mg (P = 0.38 and P = 0.33). When administering BIBN4096BS 10 mg a significant (P < 0.001) decrease in corrected VMCA from 83 ± 15 to 72 ± 12 cm/s occurred, the decrease was seen at T120; without PetCO2 correction a significant change was not seen (P = 0.12).
However, no significant difference in VMCA was found between placebo and the two different doses of the CGRP-antagonist, P = 0.18 (PetCO2 corrected) and P = 0.73 (uncorrected, Fig. 2). When calculating the percentage diameter changes, no difference was seen between treatments P = 0.68 (uncorrected).
(a) PetCO2-uncorrected VMCA values (mean ± s.e.m) are illustrated, units for data on left y-axis, ▪ placebo; ▴ BIBN4096BS 2.5 mg; • BIBN4096BS 10 mg. (b) the corresponding PetCO2 values (mean ± s.e.m) are plotted with reference to the right y-axis, ▪ placebo; ▴ BIBN4096BS 2.5 mg; • BIBN4096BS 10 mg.
The luminal diameter measurement of the temporal and radial artery revealed no significant difference between the three treatment groups (P = 0.36 temporal artery and P = 0.33 radial artery). On placebo days the temporal artery significantly (P = 0.04) increased in diameter from 0.98 ± 0.36 mm to 1.09 ± 0.30 mm (T120) and at time point T60 the radial artery significantly (P = 0.046) increased from 2.25 ± 0.44 mm to 2.41 ± 0.38 mm. When infusing BIBN4096BS 2.5 mg no changes were seen. On days when BIBN4096BS 10 mg was infused the temporal artery increased significantly (P = 0.02) from 0.89 ± 0.30 mm to 1.17 ± 0.39 mm (T30).
For none of the measured systemic haemodynamic variables a significant change between the three treatment groups was found: systolic blood pressure (BP) (P = 0.15), diastolic BP (P = 0.52), and HR (P = 0.42). Table 2 summarizes the measurements of BP and heart rate. There were no ECG changes at any time in any of the three treatment arms.
Summary of peripheral haemodynamics (mean ± SD), all time points are not included. No significant difference between the three treatment groups was seen for any of the measure variables

An analysis over time of each treatment day showed no statistically significant change of the diastolic BP. When administering BIBN4096BS 2.5 mg a significant change in systolic BP was seen over time (P = 0.03), this was not seen on the two other trial days. HR changed significantly over time after placebo and 2.5 mg (P = 0.01 and P = 0.02). Changes after placebo occurred at T60 and T180, and after BIBN4096BS 2.5 mg at T180. No change was seen after BIBN4096BS 10 mg (P = 0.4).
Adverse events
Thirty-six AEs were recorded either by the investigator or the volunteers. Two of the AEs occurred on placebo days, 16 and 18 with BIBN4096BS infusion, 2.5 mg and 10 mg. Of the 36 AEs, 16 were considered to have no relation to the study medication. The 20 remaining AEs that were considered related to study medication were graded as mild 16, or moderate 4. Of these 20 AEs, 13 were registered on days with infusion of BIBN4096BS 10 mg, and 7 after 2.5 mg. The AEs with a relation to BIBN4096BS and on placebo days are listed in Table 3.
Adverse events. In this table, BIBN4096BS and placebo related adverse events are described by: number of occurred AEs, severity and dose on which they occurred

Head, neck and chest,
face and chest,
place of infusion,
scalp.
BIBN4096BS plasma concentration
No detectable BIBN4096BS levels were recorded in predose or placebo samples. Plasma concentrations above detection level were recorded for all participants during the active drug administration (Fig. 3). The time courses of plasma concentration of BIBN4096BS following intravenous infusion were characterized by a rapid increase in plasma concentrations during infusion, followed by a rapidly declining distribution phase, and then by a slower terminal phase. The plasma concentrations of BIBN4096BS just before the end (½ minutes) of an intravenous infusion of 10 mg over 10 min had a mean maximum observed concentration (Cmax) of 768.71 ± 136.85 ng/ml. Three hours after the end of administration, the mean plasma concentration was about 6% of the corresponding Cmax value. After 2.5 mg BIBN4096BS, Cmax was 202 ± 47.72 ng/ml and the three hours value 4% of its corresponding Cmax.

Plasma concentrations of BIBN4096BS (ng/ml) measurements are shown for the individual participants. (a) Plasma concentrations after administration of 2.5 mg BIBN4096BS (b) Plasma concentrations after administration of 10 mg BIBN4096BS. Blood samples were drawn from the opposite cubital vein of infusion at the following time points: 10 min before start of the BIBN4096BS infusion and ½, 30, 60 and 180 min post infusion start. Analysis was performed using HPLC-MS/MS.
Discussion
This is the first human study of the effect of a novel CGRP-antagonist, BIBN4096BS, on the cephalic and peripheral circulation in healthy volunteers. We found that BIBN4096BS compared to placebo in resting healthy volunteers had no significant effect on cerebral blood flow or the diameter of the middle cerebral artery. In addition, no effect on the extracranial arteries or systemic haemodynamics was recorded. These data suggest that circulating CGRP does not exert a sustained vasodilator activity in humans under resting conditions. The possibility that BIBN4096BS does not cross the blood–brain barrier (BBB), dictates some caution on conclusions regarding the function of CGRP released from perivascular nerves.
CGRP in the regulation of cerebral haemodynamics
CGRP is found throughout the human body. In the cerebral circulation, the peptide has primarily been localized to perivascular sensory c-fibre terminals, originating in the trigeminal ganglion (22). When released, CGRP induces vasodilatation mediated through receptor binding and intracellular increase of cyclic nucleotides.
The CGRP-receptors, CGRP-1 and GCRP-2 are part of a receptor family also including the adrenomedullin receptor (23). The differentiation of ligand affinity within the receptor family is ascribed to the binding of receptor activity modifying proteins (RAMPs) to the calcitonin receptor like receptor (CRLR) (5).
It has been suggested, in animal experiments, that CGRP increases CBF (24). Furthermore CGRP seems to be involved in the cerebral vasodilatation in response to hypotension (25), in postocclusive hyperaemia (26) and in cortical spreading depression (27).
If CGRP plays a role in the regulation of the vascular tone of cerebral arteries or CBF in human volunteers, the administration of a potent CGRP-antagonist could be expected to induce vasoconstriction of cerebral arteries and/or arterioles. Based upon the measurements performed in this study, no vasoconstriction occurred in the middle cerebral artery or in the arterioles. We therefore suggest that in healthy volunteers the neuropeptide has no influence on the resting vascular tone of cerebral arteries.
CGRP is believed to play a pivotal role in migraine pathogenesis. Administration of CGRP to migraine patients induces an immediate headache in 8 out of 9 and a delayed migraine attack, fulfilling the IHS criteria, in 3 out of 9 (8). The implication of CGRP in migraine pathogenesis was further substantiated by the finding of the clinical antimigraine efficacy of BIBN4096BS (11).
BIBN4096BS
In animals and human cell-lines, the selectivity of BIBN4096BS for the CGRP-receptor compared to CGRP-related binding sites such as calcitonin, amylin and adrenomedullin, has been demonstrated. It has an approximately 200-fold higher affinity for the primate compared to the non-primate receptor (10, 28). In vivo studies performed in marmosets and rats have all confirmed the inhibitory effect of the antagonist on CGRP-mediated responses, induced either indirectly by electrical stimulation of the trigeminal ganglion or by intravenous CGRP administration (10, 29). In isolated human temporal, cerebral pial arteries, and coronary arteries BIBN4096BS effectively inhibited CGRP elicited dilatation. The BIBN4096BS was equally effective in intracranial and coronary arteries (3, 30). High concentration of BIBN4096BS (10 n
By nature BIBN4096BS is hydrophilic and is unlikely to pass the BBB, however, no data exists to confirm this.
The present study showed no effect on any of the measured variables, in particular no vasoactive effect was seen after BIBN4096BS 2.5 mg and 10 mg compared to placebo. Flushing and dilatation of the temporal artery after the highest dose occurred in three participants, in none after 2.5 mg and in one after placebo. Whether this was caused by BIBN4096BS cannot be excluded, but both vasodilatation and flushing was also seen on placebo days. Flushing after 10 mg of BIBN4096BS has been reported elsewhere (12). Vasodilatation and flushing might therefore occur, although our numbers are too small for any definite conclusion. 3 participants recorded a mild headache on four occasions after administration of BIBN4096BS 2.5 mg (n = 2) and 10 mg (n = 2), none after placebo. The headache was reported between 5 and 9 h after the infusion of the compound. It cannot be concluded whether the compound induced this or it was a coincidence. However, headache has been reported on previous occasions following the administration of BIBN4096BS (11, 32).
To clarify the interpretation of our findings, a descriptive analysis based on the least detectable diameter changes of the MCA was carried out. This assessment was based on the calculated diameter as described by Dahl et al. (18). The analysis was performed as a comparison (paired t-test) between placebo and the highest dose of BIBN4096BS (10 mg) since the higher dose was the most likely to induce vasoactive changes. The 95% confidence interval was −5.4% to 3.5%, indicating that the study was powered to detect a vasocontriction larger than 5.4% and a vasodilatation larger than 3.5%.
Adverse events and safety
There were more adverse events after BIBN4096BS than after placebo. There seemed to be a dose-dependent trend towards increased drug related adverse events. However, all adverse events were mild or moderate and none lead to discontinuation of the infusion. Even 10 mg of BIBN4096BS, which is four times higher than the clinically effective dose (11), had no vasoactive effect, within the limitation of the study, in the cerebral or the extracerebral vascular bed. This suggests cerebrovascular safety of CGRP-antagonism in the treatment of acute migraine.
Footnotes
Acknowledgements
The used technical equipment was partly sponsored by The Villum Kann Rasmussen Foundation, The Toyota Foundation, and The Simon Fougner Hartmann Foundation. A special thank to Lene Elkjær and Kirsten Bruunsgaard for excellent technical support and Dr Kirsten Kassøe for making everything work perfect. Boehringer Ingelheim sponsored the study and provided BIBN4096BS. The authors were independently responsible for the study design, data analysis and manuscript.