Abstract
Unpleasant sensory symptoms are commonly reported in association with the use of 5-HT1B/1D-agonists, i.e. triptans. In particular, pain/pressure symptoms from the chest and neck have restricted the use of triptans in the acute treatment of migraine. The cause of these triptan induced side-effects is still unidentified. We have now tested the hypothesis that sumatriptan influences the perception of tactile and thermal stimuli in humans in a randomized, double-blind, placebo-controlled cross-over study. Two groups were tested; one consisted of 12 (mean age 41.2 years, 10 women) subjects with migraine and a history of cutaneous allodynia in association with sumatriptan treatment. Twelve healthy subjects (mean age 38.7 years, 10 women) without migraine served as control group. During pain- and medication-free intervals tactile directional sensibility, perception of dynamic touch (brush) and thermal sensory and pain thresholds were studied on the dorsal side of the left hand. Measurements were performed before, 20, and 40 min after injection of 6 mg sumatriptan or saline. Twenty minutes after injection, sumatriptan caused a significant placebo-subtracted increase in brush-evoked feeling of unpleasantness in both groups (P < 0.01), an increase in brush-evoked pain in migraineurs only (P = 0.021), a reduction of heat pain threshold in all participants pooled (P = 0.031), and a reduction of cold pain threshold in controls only (P = 0.013). At 40 min after injection, no differences remained significant. There were no changes in ratings of brush intensity, tactile directional sensibility or cold or warm sensation thresholds. Thus, sumatriptan may cause a short-lasting allodynia in response to light dynamic touch and a reduction of heat and cold pain thresholds. This could explain at least some of the temporary sensory side-effects of triptans and warrants consideration in the interpretation of studies on migraine-induced allodynia.
Introduction
Scalp allodynia (normally non-noxious stimuli induce pain) during and after migraine attacks was described already in 1873 (1) and has later been confirmed by clinical studies (2– 4). Interictally, the pain perception thresholds of the scalp have been found to descend acutely after light stimulation and in association with nausea (5, 6). Extracranial tenderness of the skin sometimes appears early during the prodrome, and commonly accompanies the headache phase (7, 8). Based on repeated examinations during an untreated attack, and on clinical interviews, it has been suggested that most individuals experience cutaneous allodynia in the referred pain area of the scalp early in the course of the migraine attack, which later develops into a more widespread cutaneous allodynia, suggesting a central sensitization and/or disruption of central pain modulation (2, 9–11). More than every third migraineur reports an increased cutaneous sensitivity to thermal and mechanical stimuli even during headache-free intervals (12). Nevertheless, there is no robust laboratory evidence for an interictal difference in pain sensitivity in the extremities of patients with unilateral migraine or between migraineurs and control (2, 13–15). It is therefore generally considered that the cutaneous allodynia is related to the headache phase and sometimes remains during the postdrome, i.e. that it is a reversible symptom related to the attacks per se. The prevalence is still unknown.
A problem that has to be taken into account when interpreting the results obtained in research on the natural course of migraine attacks is that it is very difficult for patients to refrain from taking any drugs. It is often uncertain whether a symptom is part of the attack or an effect of the treatment. More than 70 years ago, paresthesias and allodynia to thermal stimuli were reported in association with the use of ergotamine (16), and it was later found that muscular tenderness to palpation during acute migraine was related to ergot-use but not duration of pain (17), and that scalp tenderness was greater during and after attacks in migraineurs who had taken antiemetic agents, ergotamine tartrate or prophylactic medication on the day of examination (2). Unpleasant symptoms of pressure and stiffness in the throat, neck and chest are commonly induced by all triptans. The cause of these side-effects is still unknown, but many patients discontinue treatment because of them (18–20). Cutaneous allodynia or hyperalgesia has also been described after acute intake of triptans (21, 22). For example, this may be experienced as a burning and painful sensation in the skin when taking a normally tempered shower or a dislike of being touched, especially after injection of the drug. Most patients state that the adverse events are mild or moderate, that they vanish within 10–30 min, and that they do not appear in attacks treated with analgesics only.
This has now led us to explore the hypothesis that triptans per se may induce cutaneous allodynia. We will show, for the first time, that a 5-HT1B/1D-agonist, which is designed and used for the acute treatment of migraine, paradoxically can induce a transient cutaneous allodynia. A preliminary report has been published (23).
Methods
Design
Prospective, randomized, double-blind, placebo-controlled cross-over study. The protocol was approved by the Ethics Committee at the Göteborg University.
Subjects
Twenty-four persons participated. The required number of participants to achieve adequate power could not be calculated beforehand. A sample size that could be managed by the two examiners during 6 months was chosen. One group consisted of 12 subjects (mean age 41.2 years, range 25–55, 10 women) with migraine (1 with aura only, 6 without aura only, 5 with and without aura, attack-frequency 1–6/month) diagnosed by a neurologist (M.L) according to the criteria of the International Headache Society (24) and with a history (reported spontaneously or when specifically asked for) of allodynia after injection of sumatriptan. Otherwise, the migraineurs did not have more spontaneously reported side-effect from sumatriptan than other patients. The other group consisted of 12 healthy subjects (mean age 38.7 years, range 24–58, 10 women) without migraine.
Sensory testing
Each individual participated in two sessions (≥ 7 days interval, median 14, SD 15.3) where the assessments were made during pain- and medication-free intervals (≥ 3 days since migraine/medication and no indication of an approaching attack). Each subject had one session with placebo and one with the active drug. After the second injection, 14 of the participants (58% of all) thought they could tell a difference between active drug and placebo. The subjects had been instructed to refrain from any physical effort such as riding a bicycle earlier during the day. Examinations were performed in a quiet room with a temperature of 21–23°C by the same examiners (L.L and M.L) and at the same time of the day. Before any measurements, the participant rested for at least 30 min. The subjects’ skin-temperature as measured by a digital thermometer (Ellab, Copenhagen, Denmark) varied between 30 and 33°C, and pulse rates varied between 54 and 88 beats/min. During all stimulations the subjects were instructed to keep their eyes closed, to concentrate and not to speak.
Three different steps of quantitative sensory tests were performed in a standard sequence on the skin overlying the first dorsal interosseus muscle of the left hand before and 20 + 40 min after s.c. injection with a thin needle in the right thigh of either 6 mg sumatriptan or a corresponding volume (0.5 ml) of placebo (saline). The injection devices were prepared and masked according to a randomized schedule by a research nurse in an adjacent room.
Tactile directional sensibility
The stimulation procedure has previously been described in detail (25). Briefly, a hand held stimulator was manually moved in proximo-distal orientation over the skin surface. The stimulation load was set to 4 g and speed of movement was approximately 10 mm/s. A protocol based on forced choice between ‘up’ or ‘down’ was used for 32 strokes. Three correct answers at a given distance (initially 18 mm) led to a shorter stroke (minimum 3 mm) (26). One incorrect answer led to a longer distance. The threshold for correct direction discrimination was considered to be the shortest distance for which the subject had >75% correct responses and not below that level at longer distances.
Perception of dynamic mechanical stimulation
A soft brush (SenseLab-05™, Somedic, Hörby, Sweden) was manually moved back and forth (length 3 cm) in proximo-distal orientation with a frequency of 1 Hz during 20 s. Subjects rated their perceptions of touch, unpleasantness and pain on visual-analogue (VA) scales with endpoints ‘no sensation of touch’–‘strongest sensation of touch imaginable’, ‘not at all unpleasant’–‘most unpleasant sensation imaginable’, and ‘not at all painful’–‘worst pain imaginable’. The participants used a pencil to mark their responses on the scale which was continuous and without anchor-points.
Thermal stimulation
Thermal sensory and pain thresholds were measured according to the ′method of limits’ (27). A thermostimulator (TSA-II™, Medoc, Ramat Yishai, Israel) with a precision of ± 0.1°C was used. For safety, the maximum temperature was set to +50°C. The thermode with a contact-area of 32 × 32 mm was attached lightly to the hand with a strap. With randomized time-intervals the computer initiated repeated ramps of thermal stimulation at a rate of 1°C × s−1 starting from a baseline of +32°C (28). The subject operated a lever with the right hand and was instructed beforehand to indicate first perception of cool sensation (CS), warmth sensation (WS), cold pain threshold (CP) and heat pain threshold (HP). We specifically emphasized that it was not a test of pain tolerance. Thresholds were calculated as the mean of four (CS, WS) or three (CP, HP) recordings.
Statistical approach
Sign-test for paired samples was used to compare the change in all VA-scales and tactile directional sensibility from baseline between injection of sumatriptan and placebo. Wilcoxon sign-rank test was used in a corresponding fashion for changes of thermal sensory and pain thresholds (29). All analyses were performed using the Statistical Package for Social Sciences (SPSS™) software program (version 11.0 for MacOSX, SPSS, Inc., Chicago).
Results
Tactile directional sensibility
All subjects were at baseline before sumatriptan and/or placebo capable of discriminating the shortest distance of stimulation (3 mm). There was no significant placebo-subtracted change in tactile directional sensibility by sumatriptan, neither among migraineurs (20 min: suma > placebo = 3, ties = 7, suma < placebo = 2, 40 min: suma > placebo = 5, ties = 6, suma < placebo = 1), nor among controls (20 min: suma > placebo = 2, ties = 9, suma < placebo = 1, 40 min: suma > placebo = 2, ties = 10, suma < placebo = 0).
Intensity of brush-stimulation
Before injection, the median ratings were 97 in migraineurs and 96 in controls (0–100 scales). There was no significant placebo-subtracted change in brush-evoked feeling of touch by sumatriptan, neither among migraineurs (20 min: suma > placebo = 5, ties = 2, suma < placebo = 5, 40 min: suma > placebo = 5, ties = 2, suma < placebo = 5), nor among controls (20 min: suma > placebo = 6, ties = 2, suma < placebo = 4, 40 min: suma > placebo = 6, ties = 2, suma < placebo = 4).
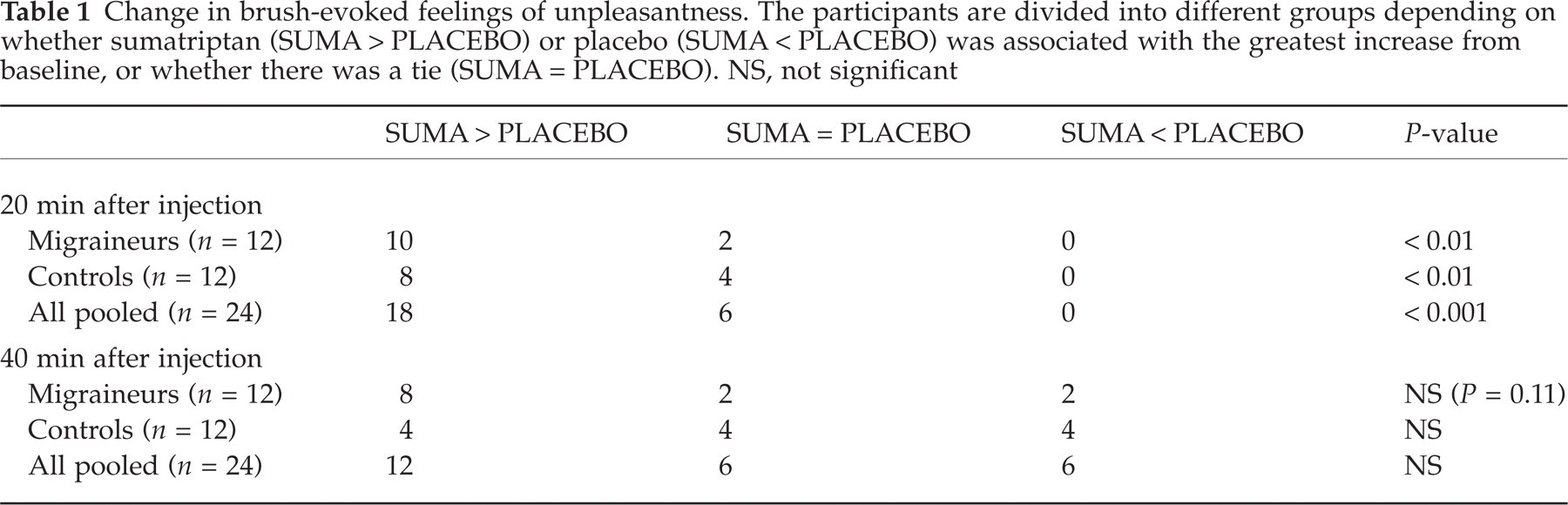
Brush-evoked feeling of unpleasantness (BEU)
Before injections, brush strokings were not perceived as unpleasant, i.e. the ratings were close to zero (Fig. 1). However, 20 and 40 min after injection of sumatriptan there was a clear increase in unpleasantness ratings notably in migraineurs but also in controls. When the effects of placebo were subtracted, the increase was significant at 20 but not at 40 min after injection in both groups (Table 1).

Values of brush-evoked feeling of unpleasantness at different assessment points. Plots give the interquartile range (boxes), median (line within boxes), highest and lowest values (whiskers) excluding outliers. (a) migraineurs; (b) controls;  sumatriptan, □ placebo.
sumatriptan, □ placebo.
Change in brush-evoked feelings of unpleasantness. The participants are divided into different groups depending on whether sumatriptan (SUMA > PLACEBO) or placebo (SUMA < PLACEBO) was associated with the greatest increase from baseline, or whether there was a tie (SUMA = PLACEBO). NS, not significant
Brush-evoked feeling of pain
Before injection, brushing was not perceived as painful, i.e. the ratings were close to zero (Fig. 2). After injection of sumatriptan there was a clear increase in pain ratings only among migraineurs and only at 20 min. However, the effect at 20 min was significant after placebo was subtracted, not only in the migraine group, but also when results from both groups were pooled (Table 2). No placebo-subtracted differences were significant at 40 min.

Values of brush-evoked feeling of pain at different assessment points. Plots give the interquartile range (boxes), median (line within boxes), highest and lowest values (whiskers) excluding outliers. (a) migraineurs; (b) controls;  sumatriptan, □ placebo.
sumatriptan, □ placebo.
Change in brush-evoked feelings of pain. The participants are divided into different groups depending on whether sumatriptan (SUMA > PLACEBO) or placebo (SUMA < PLACEBO) was associated with the greatest increase from baseline, or whether there was a tie (SUMA = PLACEBO). NS, not significant

Cool sensation threshold
Before injection the median cool sensation threshold was 31.4°C (range 26.1–31.5°C) among migraineurs and 31.3°C (range 30.2–31.6°C) among controls. There was no significant placebo-subtracted change in cool threshold by sumatriptan, either among migraineurs (20 min: suma > placebo = 6, ties = 1, suma < placebo = 5, 40 min: suma > placebo = 5, ties = 0, suma < placebo = 7), or among controls (20 min: suma > placebo = 5, ties = 3, suma < placebo = 4, 40 min: suma > placebo = 7, ties = 1, suma < placebo = 4).
Warmth sensation threshold
The median warmth sensation threshold at baseline was 33.9°C (range 32.7–37.2°C) among migraineurs and 33.2°C (range 32.5–34.7°C) among controls. There was no significant placebo-subtracted change in warmth threshold by sumatriptan, either among migraineurs(20 min: suma > placebo = 5, ties = 1, suma < placebo = 6, 40 min: suma > placebo = 3, ties = 1, suma < placebo = 8), or among controls (20 min: suma > placebo = 6, ties = 1, suma < placebo = 5, 40 min: suma > placebo = 6, ties = 1, suma < placebo = 5).
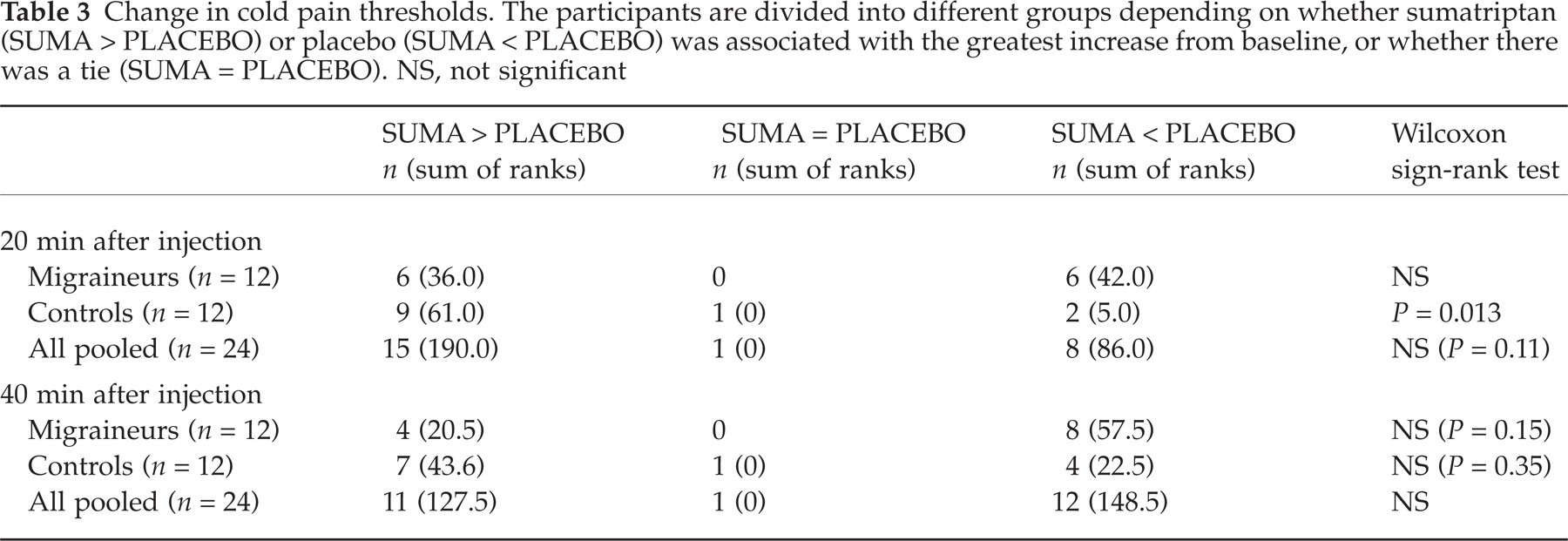
Cold pain threshold
The median cold pain threshold at baseline was 23.8°C (range 0.9–30.7°C) among migraineurs and 25.3°C (range 0.0–29.9°C) among controls. After injection of sumatriptan there was a reduction of cold pain threshold among controls at 20 min, which was significant after the placebo effect was subtracted (Fig. 3, Table 3). At 40 min no differences were significant.

Values of cold pain thresholds at different assessment points. Plots give the interquartile range (boxes), median (line within boxes), highest and lowest values (whiskers) excluding outliers. (a) migraineurs; (b) controls;  sumatriptan, □ placebo.
sumatriptan, □ placebo.
Change in cold pain thresholds. The participants are divided into different groups depending on whether sumatriptan (SUMA > PLACEBO) or placebo (SUMA < PLACEBO) was associated with the greatest increase from baseline, or whether there was a tie (SUMA = PLACEBO). NS, not significant
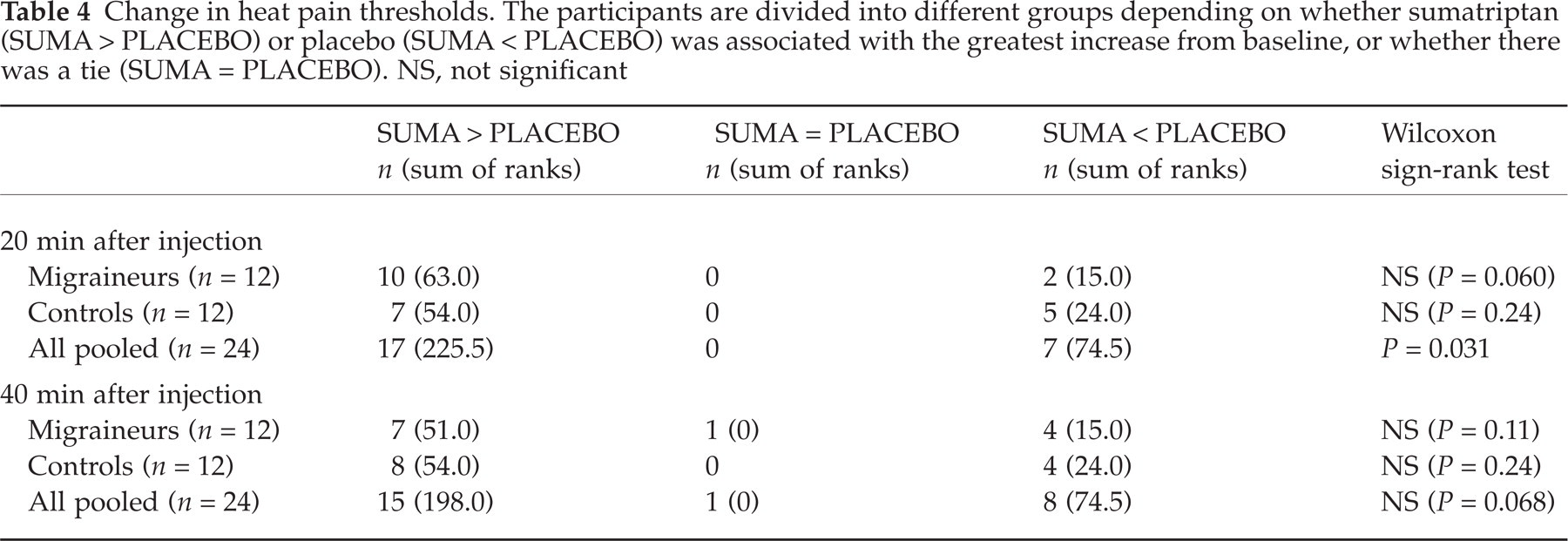
Heat pain threshold
The median heat pain threshold at baseline was 41.0°C (range 35.8–49.3°C) among migraineurs and 39.5°C (range 34.4–50.0°C) among controls. After sumatriptan had been injected there were slight reductions of pain thresholds among migraineurs as well as controls at 20 min. When data for both groups were pooled the effect was significant after placebo subtraction (Fig. 4, Table 4). All changes were nonsignificant at 40 min.

Values of heat pain thresholds at different assessment points. Plots give the interquartile range (boxes), median (line within boxes), highest and lowest values (whiskers) excluding outliers. (a) migraineurs; (b) controls;  sumatriptan, □ placebo.
sumatriptan, □ placebo.
Change in heat pain thresholds. The participants are divided into different groups depending on whether sumatriptan (SUMA > PLACEBO) or placebo (SUMA < PLACEBO) was associated with the greatest increase from baseline, or whether there was a tie (SUMA = PLACEBO). NS, not significant
Discussion
This study shows that sumatriptan may induce cutaneous allodynia, and that the phenomenon is short-lasting. We found a significant placebo-subtracted allodynia in response to light brush stroking and a reduction of thermal pain thresholds, not only among migraineurs who were selected because they had complained of sensory side-effects of sumatriptan, but also in normals who had no previous experience of the drug. It can thus be concluded that the phenomenon is not dependent on having developed the disease migraine per se. In line with this, additional statistical analyses were performed where the migraineurs and control groups were eliminated and all participants were grouped together. This increased the significance of our results.
Since it is not known what should be interpreted as a clinically relevant difference in a symptom scale score such as the one used, any measurable (i.e >1 mm) change was counted (30). However, when we post hoc tested to use a minimum of 10 mm to count as a change, placebo-subtracted increase of BEU still fell out significant 20 min after injection.
The finding of a reduced cold pain threshold in controls (only) was unexpected, and it may possibly be an artefact due to the considerably different cold pain thresholds reported by controls at baseline before injection of sumatriptan (median 19.3°C) compared to saline (median 26.1°C). Since all triptans look similar from the perspective of dose-dependent tolerability it is very likely that the observed findings can be extrapolated to other triptans.
It seemed likely that any possible triptan-effect would be generalized, and we therefore chose to study only one anatomical area (the left hand). We injected sumatriptan subcutaneously since that is the most effective route of administration although with the most side-effects. The time-intervals 20 and 40 min for testing after injection seemed appropriate considering the duration for reported sensory side-effects and the pharmacodynamics with a time to maximum plasma concentration (tmax) of 12–14 min (31). As in earlier placebo-controlled trials of sumatriptan, a limitation of the method is that a majority of the participants, also those without earlier experience of the drug, probably could tell a difference between active drug and placebo after the second injection. It is, however, unlikely that this has had any major influence on the data, since out of all studied parameters only those that had been hypothesized beforehand fell out positive. Also the normalization after 40 min is well in line with earlier reported clinical experience (21). Furthermore, it is very difficult to report false sensory thresholds in quantitative sensory testing without an increase in variance (32), and the homogenous intraindividual standard deviations in our findings suggest trustworthy results. The fact that sumatriptan caused a significant placebo-subtracted increase in BEU not only among migraineurs but also within the group of controls (P < 0.01), shows that the results are not confined to the selected migrainuers.
Our findings raise questions concerning the underlying mechanisms. According to our clinical experience, visual or auditory sensation does not appear to be enhanced by triptans, which might indicate a specific phenomenon related to the somatosensory system. We cannot, however, exclude a generalized state of hyperexcitability, although we found no indications of a change in sensory thresholds for nonpainful modalities (cool sensation, warmth sensation or tactile directional sensibility). A consistent challenge when working with psychophysical tests such as quantitative sensory testing is that they do not specify the anatomical level along the neurological pathway from peripheral receptors to cortex/mind, where an abnormality resides (33, 34). We have not assessed if there is an increased receptive field which definitely would be a sign of central sensitization. The use of defined and restricted test areas cannot discriminate between peripheral and central sensitization.
One possibility is that sumatriptan causes allodynia via an altered function of primary afferent fibres. Recent data from studies in the cat indicate that the 5-HT1B/1D agonist naratriptan sensitizes peripheral cutaneous nerves or receptors to stimulation following intravenous administration (35). Nowhere is the plasticity of function of the primary afferent fibres more evident than at the peripheral terminals (36, 37). It is not necessarily the case that peripheral neurons are of significant importance for the development of allodynia (38), but sensitization of primary afferents appears to account for some of its characteristics due to an increase of spontaneous activity, a lowered threshold for activation, and increased and prolonged firing to a suprathreshold stimulus. Contrary to former belief, peripheral sensitization is a specific process caused by substances binding to highly specific receptor molecules in the membrane of the nerve ending (38–41). Via alterations at this location, serotonin can lower the mechanical thresholds of nociceptors into the innocuous range, thus enabling weak stimuli to excite the nerve and elicit allodynia (36, 39–48). For example, both animal and human studies have shown that serotonin sensitizes muscle nociceptors for various stimuli, thereby causing allodynia and hyperalgesia (41, 49, 50). This appears to be mainly via a direct action on 5-HT1A/1B, but perhaps also 5-HT2 or 5-HT3 receptors (36, 48, 49, 51). Depending on the dose, route of administration, nociceptive test and studied species, agonists selective for certain 5-HT-receptor-subtypes may either facilitate or inhibit nociceptive transmission. 5-HT1B-agonists are, for example, usually antinociceptive, but can facilitate the tail-flick reflex in rats when given intrathecally (52–55). One can therefore not rule out the possibility that sumatriptan at its peak plasma concentrations may excite other 5-HT1 receptors (56). As a consequence of drug distribution, the plasma concentration falls rapidly, which could explain the short duration of the allodynia. There seems to be a trend in our results also after 40 min, although it did not reach statistical significance.
It has been suggested that sensitized peripheral receptors are unlikely to explain allodynia if detection thresholds are unchanged, as was the case here. Instead it has been suggested that allodynia may be due to a loss of central pain-inhibitory control (disinhibition/unmasking) and/or sensitization of higher-order neurons (5, 39, 46, 57). In both cases, the same afferent input leads to an increased central response, although the exact mechanism remains debated (43, 58, 59). Imaging studies in humans show that allodynic stimulation induces brain activity normally implicated in pain processing, reflecting a ‘misinterpretation’ of somatosensory information (38, 60, 61). Although the neuropharmacology of central sensitization has been described in quite some detail (62), information is sparse on the role of serotonin. Pharmacological experiments in animal models have shown that triptans work at least partly by a central mechanism of action (63). But it is still not known if sumatriptan passes over the blood–brain barrier in humans although the CNS-related side-effects such as sedation support a central action.
Our findings of triptan-induced allodynia warrant consideration in the design and interpretation of studies on the treatment of migraine (64). It has been hypothesized that there is a sequential development of sensitization along the trigeminovascular pain pathway during acute migraine (10, 11, 65, 66). The implication of this theory is that triptans should be administered immediately at the onset of the attack to block impulses from the periphery (10, 11). Could it be then, that triptans have both an excitatory and inhibitory effect on the somatosensory system? A future challenge will naturally be to separate the therapeutic effect of triptans on migraine-induced allodynia from the allodynia caused by these drugs per se. When comparing migraine-induced and triptan-induced allodynia, the former seems to be of slower onset with a start in the scalp area and subsequent recruitment of anatomical areas. The pain threshold changes seem also to be of much greater magnitude and longer duration. One must bear in mind though, that data from different methods of quantitative sensory testing cannot easily be compared (67). There is a need for controlled studies of acute migraine. Triptans should then be given at different times, in various doses and modes of administration, and the net effect on allodynia should be studied.
It is possible that the findings of allodynia can help us to better understand the common and often unpleasant sensations induced by triptans. These symptoms can, for example, remind of angina pectoris, but the underlying mechanism is not known (21). There are no robust abnormalities in ECG:s taken during sumatriptan-induced chest-pain, and the symptoms are also seen in patients after intake of the selective 5-HT1D-agonist PNU-142633 which lacks effect on vascular receptors. Our results are well in line with the possibility that such sensory side-effects of triptans are of direct neural origin (68, 69). Presumably, mild vasoconstriction in peripheral skeletal muscle can be the source of some sensory adverse events such as a short-lived feeling of weakness (70), but neural sensitization seems to be a more likely explanation of the allodynia (19). Patients commonly describe a short-lasting (minutes) but extremely painful exaggeration of the migraine-headache directly after intake of a triptan. Perhaps this hitherto obscure phenomenon can be explained in terms of hyperalgesia. Another important issue that should be explored in clinical studies is whether neural sensitization from chronic 5-HT1B/1D-receptor stimulation plays a role in the development of triptan-overuse headache.
In summary, the finding that triptans can cause allodynia raises further questions concerning other sensory side-effects of triptans and warrants consideration in the interpretation of studies on migraine-induced allodynia.
Footnotes
Acknowledgements
We would like to thank Annsofie Mellberg and Tomas Sundberg for skilful technical assistance and Martin Gellerstedt for valuable statistical comments during the preparation of the manuscript. In March 2003, the study received the Migraine Innovators Award (sponsored by Astrazeneca) on behalf of the faculty and delegates.
