Abstract
Botulinum toxin is increasingly advocated as effective treatment in chronic tension-type headache. We conducted a randomized, placebo-controlled clinical trial to prove efficacy of botulinum toxin in chronic tension-type headache. Patients were randomly assigned to receive botulinum toxin (maximum 100 units) or placebo (saline) in muscles with increased tenderness. After 12 weeks there was no significant difference between the two treatment groups in decrease of headache intensity on VAS (-3.5 mm, 95% confidence interval (CI) -20 to +13), mean number of headache days (-7%; 95% CI -20 to +4), headache hours per day (-1.4%; 95% CI -3.9 to +1.1), days on which symptomatic treatment was taken (-1.9%; 95% CI -11 to +7) and number of analgesics taken per day (-0.01; 95% CI -0.25-0.22). There was no significant difference in patient's assessment of improvement after week 4, 8 and 12. Botulinum toxin was not proven effective in treatment of chronic tension-type headache. Increased muscle tenderness might not be as important in pathophysiology of chronic tension-type headache as hitherto believed.
Introduction
Tension-type headache (TTH) is common with a life-time prevalence of almost 80% (1). In the general population 4–5% suffer from chronic daily headache and about half of them have chronic tension-type headache (CTTH), with headache for more than 15 days per month (2). Pathophysiology of chronic tension-type headache is unknown. It has been strongly suggested that increased myofascial tenderness and muscle hardness play an important role (3–7), although evidence for a centrally mediated origin of CTTH is increasing (8). Today, treatment of these patients is unsatisfactory and includes physical therapy, simple analgesics or antidepressant drugs (9, 10). Recently, open studies report botulinum toxin to be successful in patients with CTTH (11–13). Botulinum toxin decreases muscle tone by presynaptic blockade of the release of acetylcholine at motor endplates. It has been proven effective in diseases characterized by increased painful muscle tone, like cervical dystonia (14) and blepharospasm. Randomized data on treatment of botulinum toxin in CTTH is scarce, and inconclusive (15, 16). We report on a double-blind placebo-controlled randomized trial to determine clinical efficacy of botulinum toxin in treatment of CTTH.
Methods
Patients
Patients were eligible if they had chronic tension-type headache according to the International Headache Society criteria (17). Exclusion criteria were:
age < 18;
pregnancy;
neuromuscular disorders;
previous use of other investigational new drugs in the past 30 days prior to the screening visit;
previous use of botulinum toxin;
migraine frequency of more than one attack per month; analgesics or caffeine abuse (IHS-criteria).
The institutional ethics committee approved the study protocol, and patients gave informed consent before inclusion.
Study design
The trial had a double-blind, placebo-controlled parallel design. After patient's consent was obtained, we recorded baseline demographics (age, gender). During a baseline period of 4 weeks, patients used a diary to record the presence of headache, number of headache hours per day, number of days on which medication was taken, and number of tablets taken per day. At baseline (week 0) patients recorded their mean headache intensity scored on 100 mm Visual Analogue Scale of the previous 4 weeks (VAS-pretreatment). At 12 weeks follow-up they recorded again their mean headache intensity (VAS-post-treatment). Pericranial tenderness was measured at baseline using the Total Tenderness Score (TTS) (3). A higher score indicates more tenderness. After this baseline period, patients were randomly assigned to receive 1 unit botulinum toxin (Botox®) per kg with a maximum of 100 units or placebo (saline) in 2 cc syringes. The local trial pharmacist prepared the drug, coded the syringes, and kept treatment codes. The pharmacist had no further participation in the trial. An experienced clinical neurophysiologist (DT) performed all injections. Injection sites were selected individually in muscles with clinically increased muscle tone or muscle tenderness and included m. occipitofrontalis, m.temporalis, m. masseter, m. sternocleidomastoideus, m. splenius capitis, m. trapezius and m. semispinalis with a maximum of 10–20 unit's Botox® per muscle.
Outcomes
After patients were injected at week 0, clinical characteristics were recorded using diaries during the 12 weeks post-treatment. Primary outcome measure was the intensity of the headache of the total study period, week 0–12, scored on the VAS at week 12 (one single measurement) compared to baseline. Patients were asked to rate (in a single value) severity of headache which at times may be mild and perhaps at others severe, by ‘integrating severity over time’ (18).
Secondary outcome measures were average number of headache days, average number of headache hours per day, average number of days on which symptomatic treatment was taken, mean number of symptomatic tablets per day at 12 weeks compared to baseline. Additionally, a blinded investigator asked patients whether there was any improvement in their condition after 4, 8 and 12 weeks in the two treatment groups (five-point scale: great worsening, any worsening, no improvement, any improvement and great improvement). We also divided all patients in responders and nonresponders at week 12. We considered at least 45% reduction in the VAS compared to baseline to be clinically relevant (e.g. responder). All adverse events during the 3 months follow-up were recorded by the investigators.
Sample size determination
At baseline we expected mean headache intensity on the VAS of 40/100 mm. The estimated placebo effect was a 8 mm reduction to a VAS intensity of 32/100 at 12 weeks. We expected a 18 mm reduction in the treatment group to a VAS intensity of 22/100. To detect this clinically relevant effect (8 vs. 18 mm improvement) at a 5% level of statistical significance (two-tailed), with a power of 80%, a total of 40 patients was needed.
Statistical analysis
We calculated mean differences between baseline VAS data and VAS data after 12 weeks, using the t-test; statistical uncertainty was expressed in 95% confidence intervals. The responders at week 12 in each treatment group were compared by use of the χ2 test.
Improvement scores were compared with use of two-sided Mann–Whitney U-tests. All these analysis were done with SPSS (version 10.0).
Results
Between October 1999 and August 2001 40 patients were included at Leyenburg hospital The Hague(see Fig. 1). No patient was lost to follow-up.
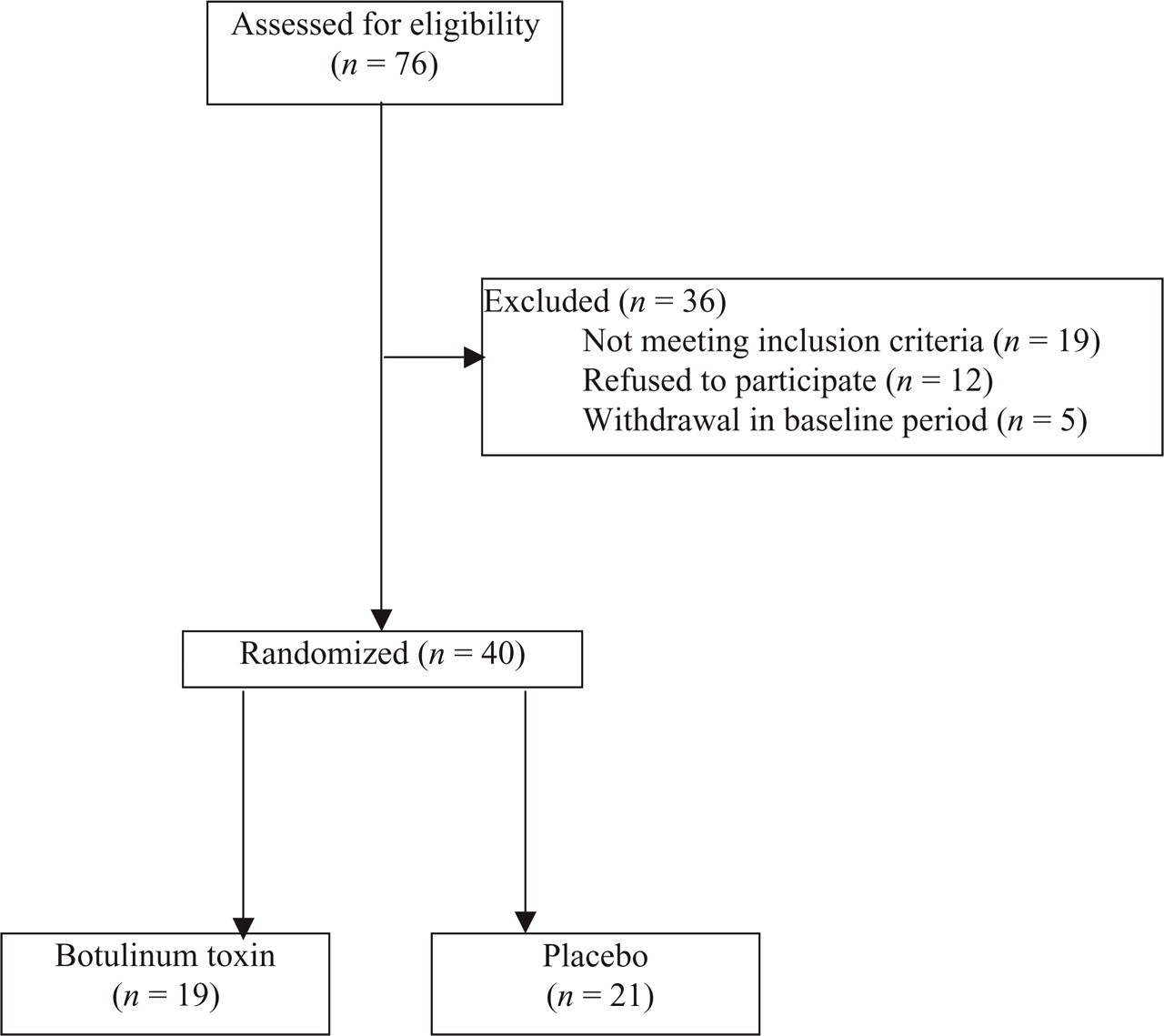
Profile of the trial.
In three patients the headache diary for the post-treatment period was incomplete (missing data on headache duration, days on which medication was taken and mean number of tablets per day for one patient in the botulinum toxin group and for 2 patients in the placebo group). There were no differences in baseline characteristics between the two treatment groups. Headache intensity, VAS at baseline was 64.5 ± 16.0 mm (mean ± SD) in the botulinum group and 69.3 ± 25.3 mm in the placebo group (Table 1). At 12 weeks the VAS scores were 53.9 ± 33.1 mm in the botulinum group and 62.2 ± 29.1 mm in the placebo group. Mean differences between VAS pre- and post-treatment was +10.6 in the botulinum group and +7.1 in the placebo group. The therapeutic gain (= difference in improvement between the two treatment groups measured in mm) was −3.5 (P = 0.68; 95% confidence interval −20 to +13). Six patients in the botulinum group were responders as defined, vs. three in the placebo group (NS, P = 0.19) (Table 2). Patients’ assessment of improvement showed no significant difference between the two groups at week 4 (P = 0.68), 8 (P = 0.51) and 12 (P = 0.37) (Table 3). There were patients with worsening of their complaints: at week 4 one patient in both groups. At week 8 two patients in both groups, and at week 12 two in the treatment group vs. one patient in the placebo group. There were no significant differences between the two treatment groups in mean number of headache days, mean number of headache hours per day, mean number of days on which medication was taken, and mean number of tablets taken per day pre- and post-treatment (Table 4). Twenty-one patients reported minor side-effects, 13 in the placebo group and 8 in the botulinum toxin group. Main complaint was short-lasting pain at the injection sites (Table 5).
Baseline characteristics

(n=18);
(n=19).
Responders

Improvement scale

Secondary outcome measures

one missing data,
two missing data.
Side-effects

Discussion
We could not demonstrate botulinum toxin to be more effective than placebo in patients with chronic tension-type headache.
The sample size was estimated to detect a clinical relevant difference for all patients with CTTH. A sample size of approximately 800 patients would be necessary to significantly detect the present small difference of 3.5 mm improvement on the VAS. This difference in favour of botulinum toxin, however, is too small to be of clinical relevance. It would certainly not justify this expensive therapy for all patients with CTTH.
Only patients with CTTH (IHS-criteria) were included. In clinical practice CTTH is often diagnosed if patients do not fulfil criteria for migraine, cluster headache or other well–defined primary headache syndromes. Not seldom CTTH patients have a mixture of diverse headache syndromes, and analgesic abuse and depression is often coexisting. We excluded patients with medication or caffeine abuse, but depression itself was no exclusion criterion. We also excluded patients with episodic tension-type headache, in which probably the transition to a chronic state not has taken place. As there were still more responders in the botulinum group (six vs. three in placebo group), botulinum toxin might still be helpful in some patients with CTTH. It cannot be ruled out that CTTH is not the single disease entity as hitherto believed, and that a subgroup of patients diagnosed as CTTH do respond to botulinum toxin (or to the injections itself).
Results of previous clinical studies are contradictory. Open label studies tend to present positive results (11–13), but double-blind placebo-controlled trials do not clearly prove a beneficial effect for botulinum toxin. In most randomized studies there was a standardized design, with predefined injection sites (19–21) and relatively low dosages of botulinum toxin (dosage 20 U Botox® to 100 U). We used injections at trigger points on a individual basis, with a rather high dosage (total dosage of 100 U Botox®). Nevertheless, one single treatment-session could still be insufficient to treat this chronic pain syndrome. Indeed some report a step-like therapeutic effect after repeated injections in CTTH (13, 22). Likewise in other chronic diseases, e.g. cervical dystonia it has been shown that results of botulinum toxin tend to be more successful after repeated injections (14).
The positive effect of botulinum toxin in open-label studies seems remarkable, and has been attributed to a high placebo effect from injectable therapies. However, the outcome measures chosen in these studies were often not well-defined or clinically irrelevant (e.g. area under the headache curve in a defined period) (12). The effect of botulinum toxin (placebo effect included) would probably have been less impressive in open studies if more relevant outcome measures were chosen. The placebo effect (15%) in our study seems relatively low, but is comparable to other studies with a randomized design (19, 21).
Pathophysiology of CTTH is not well established. Pericranial muscle tenderness is increased in patients with CTTH (3, 6, 23), although its initiating role in tension-type headache is not certain (8). Botulinum toxin has a direct myotonolytic effect, and therefore decreases muscle tone. If CTTH was only based upon increased muscle tenderness a beneficial effect of botulinum toxin would have been expected.
However, central mechanisms might play an important role in CTTH. It is assumed that initially, prolonged excitation of peripheral nociceptive afferent fibers generates pain. This leads to changes in second order neurons at the level of the spinal dorsal horn and trigeminal nucleus. These changes, so-called central sensitization, make these afferents more responsive to nociceptive stimuli, or responsive to otherwise no-nociceptive stimuli (8). Evidence for abnormal endogenous pain control mechanisms in tension-type headache is contradictory. Alterations of brainstem inhibitory interneurons were found in patients with tension-type headache (24), but could not be reproduced (5, 25–27). The nitric oxide synthesis (NOS) inhibitor (L-NMMA) aims to influence NO-related central sensitization (28). L-NMMA proved effective in patients with CTTH (29). So, successful pain management in CTTH probably also needs to influence central mechanisms of pain control. At present botulinum toxin cannot be advocated as standard treatment for chronic tension-type headache patients. Muscle contraction as an aetiological factor might not be as important in TTH as previously believed. However, it cannot be excluded that a subgroup of patients with CTTH still might benefit from botulinum toxin, especially after repeated injections (30).
Footnotes
Acknowledgements
Appreciation is expressed to M.J.A.M van Putten for his technical support and Prof M.D. Ferrari for his comments on the study protocol. Allergan provided the study medication, but was not involved in the collection, analysis and interpretation of the data.
