Abstract
The effect of exposure to a rotating optokinetic drum on the electrically evoked blink reflex was investigated in 20 healthy volunteers. Pain ratings and the area under the curve of the R2 component of the blink reflex to innocuous and nociceptive trigeminal stimulation decreased substantially during and after optokinetic stimulation. At low shock intensities, R2 decreased most during optokinetic stimulation in subjects who did not develop symptoms of motion sickness. In contrast, during the recovery period after optokinetic stimulation, suppression of R2 to moderate and intense stimuli was greatest in the most nauseated subjects. These Findings suggest that a mechanism that suppresses symptoms of motion sickness during sensory conflict also inhibits activity in wide dynamic range neurones in the trigeminal nucleus caudalis. Nausea in the absence of sensory conflict may inhibit R2 to intense electrical stimulation by provoking diffuse noxious inhibitory controls.
Keywords
Introduction
Motion sickness arises from a mismatch between vestibular and visual or proprioceptive cues (1). The symptoms that this mismatch produces resemble a combination of non-specific ‘poison’ and stress responses (2). Many of these symptoms are similar to those of migraine. For example, nausea, vomiting, dizziness and headache develop in both conditions.
During and after attacks of migraine, the scalp is usually tender to touch (3). Scalp tenderness also develops in nauseated subjects during motion sickness (4), suggesting some overlap in the mechanism that triggers these symptoms. Burstein et al. (5, 6) reported that scalp tenderness developed after the onset of migraine, and speculated that sensitization of dural nociceptors early in the attack provokes sensitization of second-order neurones in the trigeminal nucleus caudalis that receive convergent input from the facial skin. In support of this hypothesis, Kaube et al. (7) recently detected electrophysiological signs of trigeminal sensitization during attacks of migraine. They used a thin wire cathode positioned at the centre of a concentric anode to selectively stimulate trigeminal nociceptive afferents in the supra-orbital skin. The R2 component of the blink reflex evoked by stimuli delivered from the concentric electrode was over six times greater during migraine than during the headache-free period, consistent with sensitization of cutaneous nociceptive afferents or second-order neurones in the trigeminal nucleus caudalis. However, signs of trigeminal sensitization were not detected when stimuli were delivered from standard electrodes, presumably because of segmental interaction between nociceptive and non-nociceptive impulses (e.g. ‘gate-control’ effects) (8).
Noxious stimulation applied anywhere to the external surface of the body inhibits nociceptive reflexes such as the RIII limb withdrawal reflex (9) and the second (R2) component of the blink reflex (10, 11). The inhibitory effect of one noxious stimulus on another (counter-irritation) is thought to involve a process of ‘diffuse noxious inhibitory controls’ (DNIC) that originates in neurones in the subnucleus reticularis dorsalis of the caudal medulla (12). These neurones appear to have ‘whole-body’ receptive fields for noxious stimulation, and project to wide dynamic range neurones in the trigeminal nuclei and dorsal horn at all levels of the spinal cord. Stimulation of the neurones responsible for DNIC may boost the ‘signal-to-noise’ ratio of strong nociceptive signals by inhibiting responses to weak nociceptive and non-nociceptive signals that converge on the widely dispersed wide dynamic range neurones (12). Bouharrisa et al. (13) investigated the effect of noxious gastric distension on the withdrawal reflex elicited by painful electrical stimulation of the sural nerve. Gastric distension was achieved by inflating a balloon connected to an oesophageal tube inside the stomach. Inflating the balloon to pressures that induced epigastric discomfort strongly inhibited the RIII component of the leg withdrawal reflex, consistent with DNIC.
Since gastric distension induces DNIC, it seems conceivable that noxious gastrointestinal sensations from other sources would also inhibit nociceptive reflexes and subsidiary sources of pain. In particular, if the aversive sensations experienced during motion sickness trigger DNIC, the R2 component of the blink reflex to innocuous or nociceptive stimulation of the trigeminal nerve should decrease. On the other hand, if the headache and scalp tenderness that accompany motion sickness are associated with sensitization of wide dynamic range neurones in the trigeminal nuclei, the R2 component of the blink reflex to nociceptive stimulation of the trigeminal nerve should increase. These hypotheses were investigated in the present study.
Methods
Subjects
Nine male and 11 female university students aged between 17 and 29 years (mean 21.2 ± 3.3 years) were subjected to optokinetic stimulation to provoke motion sickness. Since one aim of the study was to investigate the association between signs of trigeminal sensitization (i.e. blink reflex facilitation) and symptoms of motion sickness, no attempt was made to screen out subjects with a history of migraine. However, as might be expected from the prevalence of migraine in the general community, only two subjects had experienced attacks that fulfilled International Headache Society criteria for migraine (14). Individuals with neurological disorders or other conditions that required medical treatment were excluded from participation. In particular, individuals with a history of epilepsy did not participate because of concern that optokinetic stimulation might induce an epileptic seizure. Blink reflexes were also measured in five males and five females aged between 18 and 31 years (mean 23.1 ± 4.9 years) while they sat quietly for a 45-min observation period (the control group). Subjects gave their informed consent for the procedures, which were approved by the Murdoch University Human Research Ethics Committee.
Blink reflexes
Blink reflexes were elicited by electrical stimulation of the supra-orbital nerve with standard 5 mm diameter surface electrodes and also with a concentric electrode. Kaube et al. (15) reported that topical application of a local anaesthetic cream virtually abolished the blink reflex induced by a concentric electrode but had little effect on blink reflexes induced by standard surface electrodes. They concluded that the high-density current from the concentric electrode stimulates superficial (predominantly nociceptive Aδ and C) fibres, whereas the more dispersed current emanating from standard surface electrodes stimulates deeper non-nociceptive Aβ fibres as well. Preliminary tests with the concentric electrode used in the present study confirmed that R2 virtually disappeared after topical application of local anaesthetic cream. The concentric electrode consisted of a central cathode made of copper wire 0.5 mm in diameter, fixed in the centre of the ring described by a stainless steel washer with an internal diameter of 10 mm and an external diameter of 20 mm. Since the cathode was placed approximately 1 cm above the right supra-orbital notch, only about two-thirds of the anode made contact with the skin. The stimuli were monopolar square wave pulses (pulse width 0.3 ms, starting at 1 mA and increasing in 1-mA increments to 5 mA) that were delivered at an interstimulus interval of approximately 10 s to minimize habituation. The cathode of the standard electrodes was placed over the left supra-orbital foramen, and the anode was placed approximately 2 cm rostrally on the forehead. The stimuli were monopolar square wave pulses (pulse width 0.3 ms, starting at 2 mA and increasing in 2-mA increments to 10 mA) delivered at an interstimulus interval of 10 s.
Electromyograph (EMG) signals were recorded bilaterally from the orbicularis oculi muscles with 5 mm diameter cup electrodes filled with electrode paste and attached to the mid-lower eyelids. Reference electrodes were attached lateral to the outer canthus, and a ground electrode was attached to the wrist. EMG signals were amplified by biopotential amplifiers (Grass Instrument Co., Quincy, MA, USA), and were sampled at 1000 Hz and stored for later off-line analysis on a personal computer via an MP100 Biopac Systems Analogue/Digital Channel Receptor and Acqknowledge software (Biopac Systems, Goleta, CA, USA).
Procedures
Subjects in the control condition sat on a stationary chair for 45 min. Blink reflexes were induced in 15 series at 3-min intervals (Fig. 1). Each series began with a 1-mA stimulus delivered from the concentric electrode, followed by 2–5-mA stimuli at 10-s intervals. Stimuli were then delivered from the standard electrodes, starting at 2 mA and followed by 4–10-mA stimuli at 10-s intervals. After each stimulus, subjects rated pain intensity verbally on a scale where 0 corresponded to no sensation, 2–3 to mild pain, 4–6 to moderate pain, 7–8 to somewhat severe pain, 9 to severe pain, and 10 to extremely severe pain. Subjects were also given the option of rating stimuli as a ‘painless sensation’ (rated as 1). A verbal rating scale was used instead of a visual analogue scale because looking at a visual analogue scale during optokinetic stimulation would have interrupted the optokinetic effect. The pain threshold was defined as the lowest intensity in each block of five series at which three stimuli were rated as painful (i.e. a rating of ≥ 2). Since ratings of a ‘painless sensation’ obviously could not be included in a scale of pain intensity, ratings of 1 were recoded to 0, 2–1, 3–2 and so on, to form a pain intensity scale ranging between 0 and 9.

Summary of procedures. Blink reflexes were recorded in 15 3-min series. In the experimental group, series 6–10 were recorded during optokinetic stimulation. Each series began with 1–5-mA stimuli delivered at 10-s intervals from a concentric electrode. Shock intensities of 2–10 mA were then delivered from standard electrodes.
After each series of electrical stimuli, subjects rated dizziness, nausea and headache on 0–10 scales: 0 corresponded to no sensation and 1 to slight dizziness or stomach or head awareness. Thereafter, ratings of 2–3 corresponded to mild, 4–6 to moderate, 7–8 to somewhat severe, 9 to severe, and 10 to extremely severe dizziness, nausea or headache. The subject rested quietly for approximately 1 min before the next series started.
In the experimental condition, blink reflexes were induced in five series before, five series during, and five series after optokinetic stimulation (Fig. 1). To provoke symptoms of motion sickness, the subject sat on a stationary chair with his or her head and shoulders inside a drum 50 cm in diameter, 70 cm in height, and painted internally with 24 pairs of vertical black and white stripes (16). The drum revolved 10 times per minute for 15 min or until vomiting was imminent. To enhance the illusion of movement created by the moving stripes, the subject was asked to focus on a distant point rather than to watch the stripes moving past. The electrical stimuli started 1 min after the drum began to rotate, and the series continued at 3-min intervals as described above. The subject continued to sit with his or her head and shoulders inside the drum for 15 min after the drum had stopped rotating. The first series of electrical stimuli during this recovery period started 1 min after the drum had stopped rotating.
Data reduction and statistical analysis
Signals were rectified and integrated prior to analysis of the R1 and R2 components of the blink reflex. For the standard electrodes, the area under the curve (AUC) of the R1 component was assessed in the time window 9–26 ms after the stimulus (11). Stimulation with the concentric electrode did not induce the R1 component of the blink reflex (15). For both the standard and concentric electrodes, the R2 AUC was measured in the 27–87-ms window after the stimulus (11). Values during and after optokinetic stimulation were expressed as a percentage of the mean value before optokinetic stimulation. In the control group, mean values during series 6–10 and 11–15 were expressed as a percentage of mean values during series 1–5. The data points obtained from one subject who withdrew from the optokinetic drum after only 3 min were not included in the analyses. Missing values were replaced by mean scores in two other subjects who withdrew after 12–13 min of optokinetic stimulation. The other 17 subjects completed the full 15 min of optokinetic stimulation. Blink reflex latencies were not measured due to the relatively low sampling rate (1000 Hz) and to frequent uncertainty about the point of onset of R1 and R2 in individual (non-averaged) waveforms.
To simplify the interpretation of findings, the effects of stimulation with the standard and concentric electrodes were investigated separately. Changes in the pain threshold over the course of the experiment were investigated in 2 × 3 [group (experimental, control) × phase (before, during and after optokinetic stimulation)] analyses of variance. Analyses that investigated supra-threshold ratings of pain had an additional factor of shock intensity (five levels). The effect of optokinetic stimulation on blink reflexes was investigated in relation to (i) the shock intensity at the initial pain threshold, and (ii) the full range of shock intensities. The first set of analyses had factors for group, phase, and side (ipsilateral vs. contralateral to stimulation for the R2 component of the blink reflex). The second set of analyses had an additional factor of shock intensity (five levels). The multivariate solution was used for all factors with more than two levels. Spearman's non-parametric correlation coefficient (ρ) was used to investigate the relationship between mean blink reflex suppression at each stimulus level and peak ratings of dizziness, nausea and headache during and after optokinetic stimulation.
Results
Symptoms of motion sickness
As shown in Fig. 2, mild nausea and dizziness developed shortly before optokinetic stimulation and gradually intensified during stimulation. Dizziness and nausea started to subside shortly after subjects withdrew from the drum, whereas headache persisted. In contrast, symptom ratings were minimal in the control condition.

Pain thresholds and ratings
Optokinetic stimulation did not influence the pain threshold to electrical stimulation of the supra-orbital nerve, which increased over the course of the experiment both in experimental and control subjects (Fig. 3). Supra-threshold pain ratings were higher in experimental subjects than in controls before optokinetic stimulation. As shown in Fig. 4, ratings to shock intensities above 2 mA decreased during and after optokinetic stimulation, but decreased only slightly during the equivalent period in controls [for the concentric electrode, group × phase interaction F(2,26) = 5.73, P < 0.01; for the standard electrodes, group × phase interaction F(2,26) = 3.75, P < 0.05].

Intensity of electrical stimulation at the pain threshold before, during and after optokinetic stimulation (•), and during a similar time period in controls (○). The pain threshold increased over the course of the experiment, but did not differ between experimental subjects and controls.
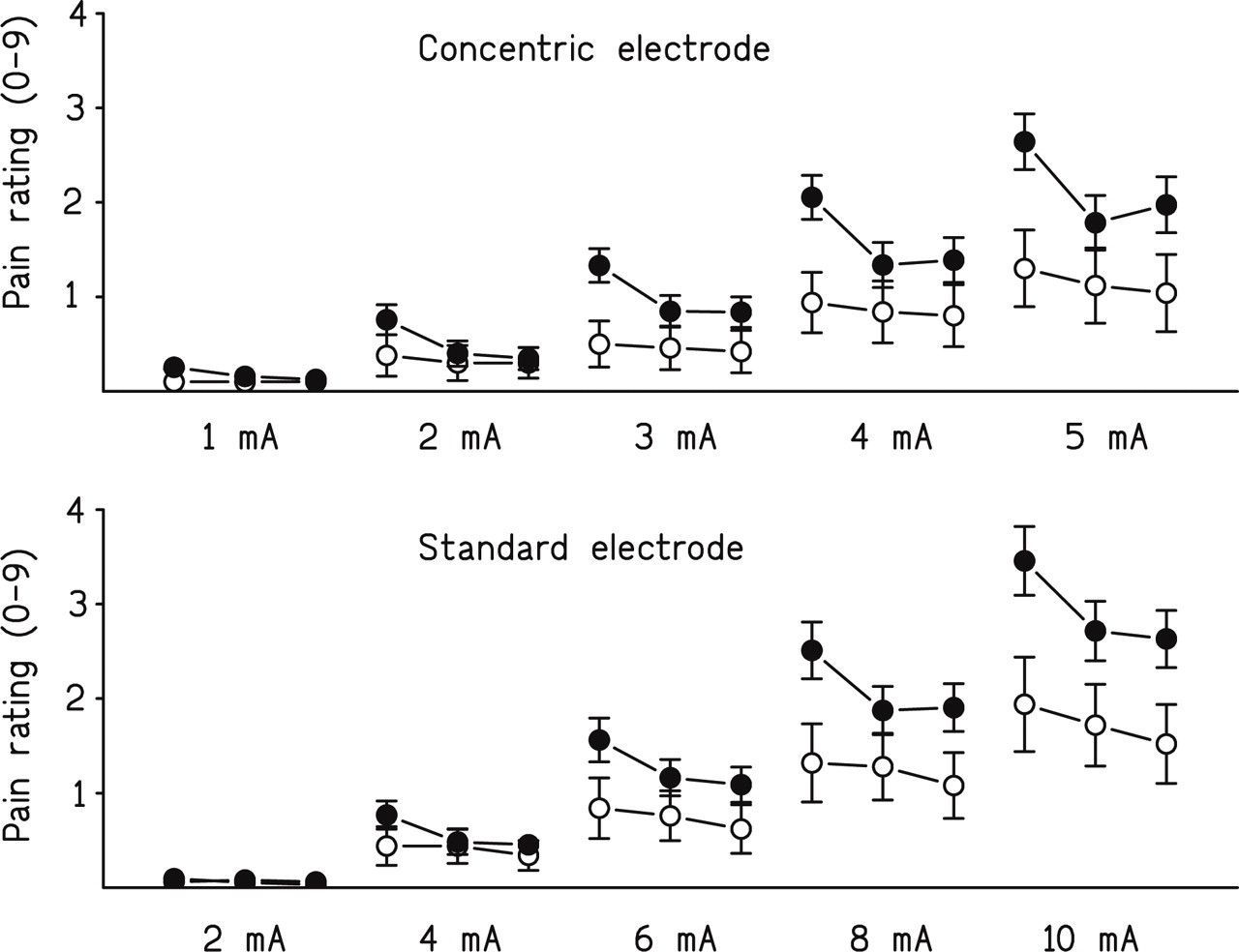
Pain ratings before, during and after optokinetic stimulation at each shock intensity (•), and during a similar time period in controls (○). Ratings for shock intensities above 2 mA were greater in experimental subjects than in controls during the baseline period, but decreased during optokinetic stimulation.
Blink reflexes
Examples of blink reflexes recorded from a single individual before and during optokinetic stimulation are presented in Fig. 5. In the group as a whole, the R1 component of the blink reflex did not change from baseline during or after optokinetic stimulation, either at the initial pain threshold or across the full range of shock intensities. In contrast, the R2 component decreased during optokinetic stimulation to shock intensities below and above the pain threshold, and this decrease persisted to some extent during the recovery period (Fig. 6). For the concentric electrode, the decrease across the full range of shock intensities averaged 32.9 ± 3.2% in experimental subjects compared with 11.6 ± 4.4% in controls [F(1,27) = 15.1, P < 0.001]. For the standard electrodes, the decrease averaged 22.3 ± 2.2% in experimental subjects compared with 6.3 ± 3.0% in controls [F(1,27) = 18.5, P < 0.001]. No other effect that involved comparisons between experimental and control subjects achieved statistical significance. During optokinetic stimulation, R2 suppression to the 2-mA stimulus delivered from the standard electrodes (thought to stimulate mainly Aβ fibres) did not differ from R2 suppression to the 2-mA stimulus delivered from the concentric electrode (thought to stimulate cutaneous nociceptive fibres) (R2 suppression averaged 33.1 ± 6.7% and 37.2 ± 6.0%, respectively, for the standard and concentric electrodes).

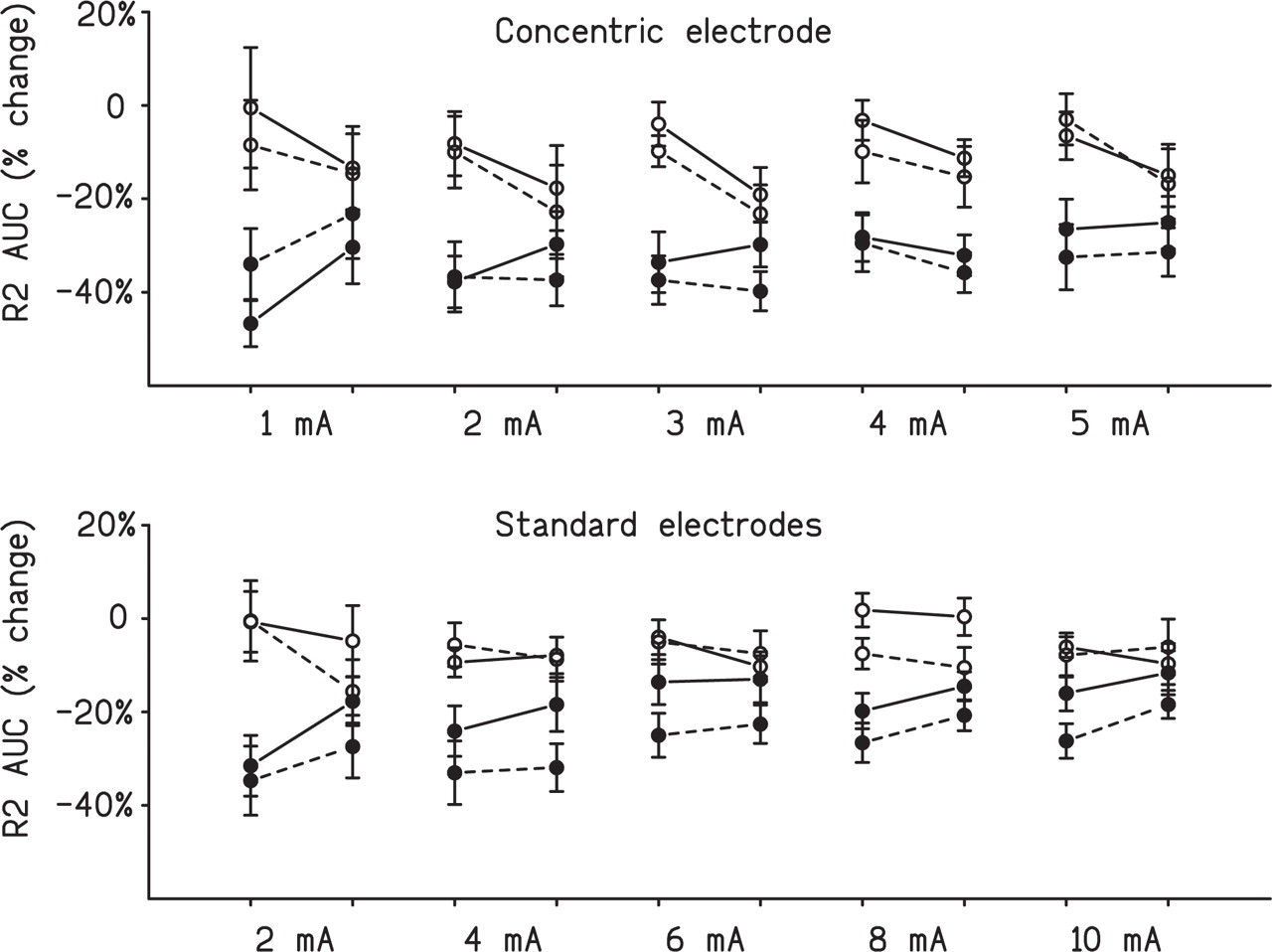
Suppression of the R2 component of the blink reflex to electrical stimuli during and after optokinetic stimulation (•), and changes in the area under the curve (AUC) during a similar time period in controls (○). The solid lines represent the response ipsilateral to electrical stimulation, and the dashed lines represent the contralateral response.
Association between suppression of R2, symptoms, and pain ratings
The decrease in pain ratings after optokinetic stimulation was associated with R2 suppression for the 2-mA stimulus delivered from the concentric electrode [Spearman's ρ (17) = 0.75, P < 0.001], and the association approached significance for the 1-mA stimulus [Spearman's ρ (17) = 0.40, P < 0.1]. There was no significant association between pain rating changes and R2 suppression to stimuli delivered from the standard electrodes, either during or after optokinetic stimulation.
At the lowest shock intensities, suppression of the R2 component of the blink reflex during optokinetic stimulation was weakest in subjects with the most severe motion sickness (Table 1). This association did not hold for shock intensities> 2 mA. In contrast, suppression of R2 to 4-mA and 10-mA stimuli delivered from the standard electrodes was greatest in the most nauseated subjects during the recovery period after optokinetic stimulation (Table 2).
Relationship between suppression of the R2 component of the blink reflex and symptoms of motion sickness during optokinetic stimulation

Spearman's ρ statistically significant:
∗P < 0.05;
∗∗P < 0.01.
Relationship between suppression of the R2 component of the blink reflex and symptoms of motion sickness after optokinetic stimulation

Spearman's ρ statistically significant:
∗P < 0.05;
∗∗P < 0.01.
Discussion
Subjective feelings of anxiety and dread, associated with nausea and signs of sympathetic nervous system activity, often precede and usually accompany vomiting in motion sickness (2). In the present study, nausea and dizziness started to increase before optokinetic stimulation, suggesting that anticipating motion sickness initiated stress-linked symptoms. Jokerst et al. (17) reported that slow, deep breathing before and during optokinetic stimulation suppressed the tachygastric component of the electrogastrogram (which correlates with nausea), and helped to control symptoms of motion sickness. Similarly, relaxation and other forms of stress management help to prevent recurrent attacks of migraine (18), possibly by suppressing stress-linked symptoms.
Pain ratings and R2 to innocuous and noxious stimulation of the supra-orbital nerve decreased during optokinetic stimulation, and these effects persisted to some extent afterwards. However, R1 remained unchanged. Painful infrared laser stimulation that selectively activates nociceptive Aδ fibres in the forehead evokes a bilateral blink reflex at a latency corresponding to R2, but does not evoke an early ipsilateral R1 component (19). Thus, nociceptive Aδ fibres that project to the trigeminal nucleus caudalis apparently contribute to R2 but not R1. Ellrich and Treede (11) reported that painful thermal stimulation of the forearm inhibited R2 to innocuous stimulation of the supra-orbital nerve whereas R1 was unaffected, consistent with involvement of wide dynamic range neurones in R2 but not R1. Since R2 to the full range of shock intensities decreased during optokinetic stimulation, the present findings suggest that sensory conflict or a secondary effect of sensory conflict (e.g. nausea, dizziness or headache) inhibited activity in trigeminal wide dynamic range neurones.
An association was detected between the decrease in pain ratings and R2 suppression to low-intensity stimuli delivered from the concentric electrode after optokinetic stimulation; however, R2 suppression to all other stimuli was unrelated to pain rating changes. Several effects may account for this general lack of association. Since the full range of stimuli delivered from standard electrodes and high-intensity stimuli delivered from concentric electrodes appear to activate non-nociceptive as well as nociceptive fibres (15), segmental interaction between neural impulses from different nerve fibre groups (8) may have influenced pain sensations. Pain thresholds increased and pain ratings and R2 declined slightly over the course of the experiment in control subjects, possibly because of adaptation to the sensory effects of repetitive electrical stimulation or decreases in arousal (20). These effects may also have contributed weakly to R2 suppression and decreases in pain during and after optokinetic stimulation. To ensure that the optokinetic effect was not interrupted, subjects rated pain verbally on an 11-point scale; however, this ordinal method of rating is not particularly sensitive to change, and an association between pain rating changes and R2 suppression may have been overlooked at some stimulus intensities. Pain ratings were greater during the baseline period before optokinetic stimulation than in the control condition, presumably because of anticipatory anxiety. Subsequent changes in anxiety during and after optokinetic stimulation may have contributed to variation in R2 suppression and pain ratings. In addition, the distracting effects of the optokinetic stimulus may have influenced pain reports. Despite these potential sources of variation, pain decreases were moderately related to R2 suppression for weak stimuli delivered from the concentric electrode after optokinetic stimulation, consistent with inhibition of activity in trigeminal wide dynamic range neurones.
At low stimulus intensities, R2 decreased most during optokinetic stimulation in subjects who did not develop symptoms of motion sickness. This effect achieved statistical significance in only a few instances. However, the direction of association was consistent for all stimuli delivered from the concentric electrode and for painless stimuli delivered from the standard electrodes, suggesting that the effect would have been detected at a wider range of stimulus intensities in a larger sample of subjects. Presumably the recruitment of additional influences on R2 (e.g. segmental interaction between nociceptive and non-nociceptive impulses; or DNIC triggered by nausea, see below) masked the association at higher stimulus intensities. This new finding suggests that a mechanism that suppressed activity in trigeminal wide dynamic range neurones also inhibited symptoms of motion sickness. A mechanism such as this may help to counteract the immediate development of headache and other symptoms of motion sickness during transient or minor episodes of sensory conflict encountered in everyday life (e.g. overbalancing while walking on uneven ground, slipping on smooth surfaces, or being pushed).
Since susceptibility to motion sickness is greater than normal in migraine sufferers (21), it is tempting to speculate that an inherent failure to suppress symptoms such as nausea, dizziness and headache increases vulnerability to both conditions. Consistent with this notion, we previously found that nausea was more likely to develop in migraine sufferers than in controls during optokinetic stimulation (4). In addition, pain sensitivity in the fingertips increased in migraine sufferers but not in controls after optokinetic stimulation, suggesting that sensory conflict disrupted pain inhibitory mechanisms more readily in migraine sufferers than in controls. Further investigations are required to determine whether failure of a mechanism that suppresses activity in trigeminal wide dynamic range neurones contributes to symptoms (e.g. headache, scalp tenderness and photophobia) during attacks of migraine.
If the aversive sensations associated with motion sickness trigger DNIC, it might be expected that DNIC would be greatest in subjects with the most intense symptoms. During the recovery period after optokinetic stimulation, suppression of R2 to the 4-mA and 10-mA stimuli was greatest in the most nauseated subjects. This relationship was not detected during optokinetic stimulation, and did not hold for stimuli delivered from the concentric electrode or for the 2-mA, 6-mA or 8-mA stimuli delivered from the standard electrodes. Thus, further investigations are required to determine whether nausea triggers DNIC.
The hypothesis that the headache and scalp tenderness that accompany motion sickness would facilitate R2 to trigeminal nerve stimulation was not supported. Hence, some other mechanism apparently mediates scalp tenderness in nauseated subjects after optokinetic stimulation (4). In this respect motion sickness differs from migraine, which is associated with an increase in AUC (an index of mean amplitude) and a decrease in the latency of R2 to nociceptive supra-orbital stimulation during attacks (interpreted by Kaube et al. (7) to be signs of trigeminal sensitization). The headache associated with motion sickness is usually described as a dull ache across the forehead, in the temples or behind the eyes; unlike migraine, only occasionally does the headache throb or affect one side more than the other (4). Perhaps a more intense or persistent headache than that generally experienced during motion sickness is required to induce trigeminal sensitization. Further studies are needed to determine whether neurophysiological signs of trigeminal sensitization develop more readily in migraine sufferers than in controls during motion sickness.
In conclusion, the present findings suggest that the sensory conflict provoked by optokinetic stimulation inhibited activity in trigeminal wide dynamic range neurones, and also inhibited symptoms of motion sickness. A process that ultimately overpowers this inhibitory mechanism may trigger symptoms of motion sickness during prolonged sensory conflict. Whether failure of this inhibitory mechanism also initiates symptoms during attacks of migraine requires further investigation.
Footnotes
Acknowledgements
This study was supported by grants from the National Health and Medical Research Council of Australia and the U.K. Migraine Trust. I wish to thank Ms Shiree Hassard for technical assistance.
