Abstract
This study was conducted to investigate the effect of 5-HT1B/1D receptor activation on nitroglycerin (NTG)-induced cerebral hyperaemia and neuronal nitric oxide synthase (nNOS) expression in trigeminovascular neurones. NTG (10 mg/kg) was infused intravenously to adult male Wistar rats with or without pretreatment with 5-HT1B/1D receptor agonist, sumatriptan (0.4 mg/kg, intravenously). Cortical blood flow and expression of nNOS enzyme in trigeminal ganglia, trigeminal nucleus caudalis and perivascular nerve fibre surrounding superior sagittal sinus were measured. The results showed that pretreatment with sumatriptan could significantly shorten the period of NTG-induced cerebral hyperaemia without compromising the magnitude of hyperaemic peak. Sumatriptan pretreatment also attenuated the NTG-evoked expression of nNOS in all studied areas. Based on these findings, we suggest that 5-HT1B/1D receptor has an important role in stabilizing the trigeminovascular system by attenuating the expression of nNOS enzyme, hence reducing nitric oxide production.
Introduction
Serotonin (5-HT) and nitric oxide (NO) are two transmitters significantly involved in the pathogenesis of migraine. Several lines of evidence indicate that both transmitters may have opposed effects in control of the trigeminovascular system. Clinically, migraine attacks have been shown to coincide with a decrease in platelet 5-HT level (1) and increase in nitrate metabolites in jugular venous blood (2). It was also demonstrated that the cerebral microvascular system was supersensitive to NO during the period of 5-HT depletion (3). However, the interaction between these two systems may be more complex due to the fact that 5-HT can exert its physiological functions via several receptor subtypes. For instance, we have recently shown that activation of 5-HT2A receptor can enhance the expression of neuronal nitric oxide synthase (nNOS) enzyme in the trigeminal pathway (4).
Characterization and localization of 5-HT1B/1D serotonin receptors is an important advance in migraine research during the last decade. These findings offer a new avenue for a therapeutic approach to abort migraine headache. The agonists to these receptors, namely triptan compounds, are considered the most effective treatment for acute migraine attacks. These receptors are expressed in vascular smooth muscle (mainly 5-HT1B receptors) and perivascular nerve fibres (mainly 5-HT1D receptors) (5). Activation of vascular 5-HT1B receptors causes vasoconstriction. Because the first compound, sumatriptan, cannot pass through the blood–brain barrier, its efficacy in aborting migraine attacks is explained solely by this peripheral vasoconstrictive effect. However, subsequent studies showed that triptans might exert their anti-migraine effect via multiple mechanisms. For example, administration of sumatriptan can minimize the amount of the ictal release of calcitonin gene-related peptide. This effect was evident in both animal models and clinical migraine attacks (6). Besides cranial vessels, 5-HT1B/1D receptor has also been identified in various brainstem areas involved in the process of cranial nociception. These include trigeminal nucleus caudalis, area postrema, nucleus of solitary tract, etc. (7). This anatomical evidence raises the possibility of central anti-migraine effect of triptan compounds. The effect of 5-HT1B/1D receptor upon the NO system has not yet been well described.
This study was conducted to determine the possible interaction between 5-HT1B/1D receptor activity and the NO system in the process of cerebrovascular control. The objectives of this study included investigating the effect of 5-HT1B/1D receptor agonist on cerebrovascular response to NO and determining the effect of 5-HT1B/1D receptor agonist in control of nNOS expression in the trigeminovascular pathway.
Materials and methods
Animals
Adult male Wistar rats (National Laboratory Animal Centre of Mahidol University, Thailand) weighing 250–350 g were housed in stainless cages with free access to food and water, and maintained in a 12-h dark–light cycle, under a controlled environment. All tests were conducted under the ethical guidelines for investigation of experimental pain in conscious animals (8). The protocol was approved by the Ethical Committee of Chulalongkorn Medical School.
Study design
Rats were divided into four groups of eight to ten animals each. The first group received sumatriptan succinate (0.4 mg/kg body weight) intravenously, followed by normal saline. This dose was previously shown to be able to block c-fos expression within rat trigeminal nucleus caudalis caused by chemical stimulation of the meninges (9). The same dose of sumatriptan was given to the second group 5 min before nitroglycerin (NTG) (10 mg/kg body weight, intravenously) infusion. The third group was treated with NTG alone without sumatriptan pretreatment. The last group, which served as control, received only vehicle and normal saline. After drug treatment, parietal cortex blood flow was continuously monitored for 1 h with a laser Doppler flowmeter. Animals were then killed with an overdose of pentobarbital and were perfused for the study of nNOS immunoreactivity.
Cortical blood flow monitoring
After being anaesthetized with pentobarbital sodium (50 mg/kg i.p.), rats were mechanically ventilated by a positive pressure ventilator (Rodent ventilator model 683; Harvard Apparatus, South Natick, MA, USA) via tracheostomy opening. Blood pressure was monitored throughout experiments with an intra-arterial pressure transducer (Nikhon model TP-300T; Nihon Khoden, Tokyo, Japan) placed in a femoral artery and recorded on a polygraph (Nikhon RM 6000; Nihon Khoden). Arterial blood was collected periodically for determination of pH, PaO2 and PaCO2 by a pH/blood gas analyser (238 pH/blood gas analyser; Ciba Corning Diagnostics, Halstead, Essex, UK). The blood gas and pH were controlled in the physiological range throughout the experiment.
Parietal craniotomy was performed and the dura was opened to expose the cortical surface. The cortex was continuously superfused with artificial cerebrospinal fluid. The fibreoptic needle probe of a laser Doppler flowmeter (ALF 21; Advance Co. Ltd, Tokyo, Japan) was placed perpendicularly at a distance of 2 mm above the cortical surface. The wavelength of laser beam was 780 nm. Data were calculated as percent changes from the baseline value.
nNOS immunohistochemical studies
After completion of cortical blood flow recording, rats were killed and perfused transcardially with 300 ml of phosphate buffer saline (PBS; pH 7.4) followed by 300 ml of 4% paraformaldehyde in 0.1 M PBS. After perfusion, the brains, cervical spinal cords, trigeminal ganglia and superior sagittal sinus were removed. These areas were selected for nNOS immunoreactivity as they include the trigeminal nucleus caudalis (TNC), the trigeminal ganglia and NO-containing perivascular nerves surrounding the superior sagittal sinus. Tissues were then cryoprotected by immersing in 30% sucrose in 0.1 M phosphate buffer solution at 4 °C. The brainstem and trigeminal ganglia were cut coronally in a cryostat (HM 505N; Microm, Waldorf, Germany) at a thickness of 30 µm. The sections were then collected in the PBS for further processing. Free-floating sections and whole-mounted preparation of superior sagittal sinus were rinsed in PBS 2 × 5 min and were treated with normal swine serum in PBS for 30 min in order to minimize the non-specific binding. Sections were incubated with a specific rabbit anti-NOS antiserum (dilution 1 : 500) (Chemicon Int., Temecula, CA, USA) at 4 °C for 24 h. The sections were washed with PBS and were incubated with biotinylated swine anti-rabbit IgG (dilution 1 : 400) (Dako, Glostrup, Denmark) for 60 min. After incubation, tissue sections were rinsed with PBS (3 × 5 min). The tissue sections were reacted with streptavidin–biotin horseradish peroxidase complex (dilution 1 : 200) for 60 min at room temperature and then washed. Finally, they were reacted for peroxidase activity in a solution containing 0.025% 3,3′-diaminobenzidine (Sigma, St Louis, MO, USA) and 0.01% H2O2 in 0.05 M Tris–HCl buffer pH 7.6 for 30 min. The stained tissue sections were washed with distilled water (2 × 5 min), mounted on gelatin-coated slides, dried and coverslipped.
Neuronal NOS immunoreactive (nNOS-ir) neurones were distinguished by their darkly stained cell bodies and processes. Expression of nNOS in TNC was determined by counting nNOS-ir neurones in lamina I and II of TNC from five sections of the first cervical spinal cord and five sections from caudal medulla. The data were averaged and expressed as number of cells per slide. Expression of NOS in trigeminal ganglia in each rat was determined by counting the NOS-ir cells from 1000 randomly selected ganglionic cells. Data were expressed as percent of NOS-ir cells per rat. A hundred NOS-ir varicosities on the perivascular plexus surrounding the superior sagittal sinus were randomly selected from each group. The area of NOS-ir varicosities was then calculated using Leica Q win program (Leica, Deerfield, MA, USA). The counting procedure was performed manually by an author (C.S.) who was blinded to the treatment groups.
Statistical analysis
All data were presented as mean ± SD. Serial blood flow changes were subjected to statistical analyses using ANOVA for repeated measures with post hoc Dunnett's t-test. The numbers of nNOS-ir cells were tested for possible statistical difference among groups using Mann–Whitney method. All the statistical analyses were performed using computer program SPSS version 9 for windows (SPSS, Chicago, IL, USA). Probability values of < 0.05 were considered statistically significant.
Results
Infusion of NTG led to a transient reduction in systemic blood pressure. Systemic blood pressure rapidly became normalized after NTG discontinuation and remained stable throughout the study. (Fig. 1) Besides transient hypotension, NTG did not cause any substantial changes in other physiological parameters. Infusion of sumatriptan did not produce significant change in systemic arterial pressure.

The effect of nitroglycerin (NTG) and sumatriptan on mean arterial blood pressure. The graph demonstrates the mean value of mean arterial pressure. Infusion of NTG causes immediate decrease in mean arterial pressure. This hypotension is rapidly normalized after NTG discontinuation. Infusion of sumatriptan does not produce any significant change in systemic blood pressure. To minimize the complexity of the figure, the error bars are omitted. The standard deviation is approximately 20% of the mean value (coefficient of variation = 0.2). ▪, Sumatriptan + NTG; □, saline + NTG; •, sumatriptan + saline; ○, saline + saline.
Effect of 5-HT1B/1D agonist and NTG on Cortical Blood Flow
NTG infusion caused an initial decline in cortical blood flow, which was followed by long-lasting hyperaemia. The blood flow reached its peak (140 ± 38% of the baseline value) almost immediately after NTG discontinuation. Degree of cerebral hyperaemia slowly declined to the magnitude of 20% above baseline value 15 min post-infusion and remained stable until the end of the experiment. The observed blood flow at the end of monitoring was 121 ± 18% of baseline value. No significant change in flow was demonstrated in the control group. The flow changes observed in NTG-treated rats was significantly different compared with the control group (P < 0.05, ANOVA for repeated measures).
Pretreatment with sumatriptan substantially shortened the period of NTG-induced cerebral hyperaemia without compromising its peak amplitude. No significant difference was observed when the magnitudes of NTG-induced initial hyperaemic peaks between rats with and without sumatriptan pretreatment were compared. In the sumatriptan-treated group, the blood flow gradually decreased and was normalized at the 15 min post-NTG infusion. The inhibitory effect of sumatriptan upon NTG-induced hyperaemia was first observed 6 min post-NTG infusion. The difference in NTG-induced hyperaemia between rats with and without sumatriptan pretreatment became statistically significant 10 min post-infusion (ANOVA for repeated measures, post hoc Dunnett's test, P = 0.03). Such a statistically significant difference was observed throughout the monitoring period (Fig. 2). Administration of sumatriptan alone did not produce any substantial change in cortical blood flow.

The effect of sumatriptan on nitroglycerin (NTG)-induced cerebral hyperaemia. The graph demonstrates the mean value of cortical blood flow (CBF) measured by laser Doppler flowmetry. The period of cerebral hyperaemia is significantly shorter in rats receiving sumatriptan pretreatment (▪) compared with those without pretreatment (□) (∗P < 0.05, ANOVA for repeated measures with post hoc Dunnett's t-test between NTG-treated rat with and without pretreatment with sumatriptan). To minimize the complexity of figure, the error bars are omitted. The standard deviation is approximately 20% of the mean value (coefficient of variation = 0.2). ▪, Sumatriptan + NTG; □, saline + NTG; •, sumatriptan + saline; ○, saline + saline.
Effects of 5-HT1B/1D agonist on NTG-evoked nNOS expression in the trigeminovascular pathway
The results showed that the brief period of NTG exposure could increase nNOS expression in several areas of the trigeminovascular pathway, as evident from a greater number of NOS-ir cells in all studied areas. The numbers of NOS-ir cells in the TNC were 52 ± 27 and 30 ± 14 cells per section for NTG and control groups, respectively (P < 0.01, Mann–Whitney test). The same pattern was also true in the trigeminal ganglia where NOS-ir cells accounted for 24 ± 3% and 10 ± 3% (P < 0.01, Mann–Whitney test) in the NTG and control groups, respectively. Administration of NTG also enlarged the size of nNOS-ir boutons surrounding the superior sagittal sinus (Fig. 3). The surface areas of the perivascular NOS-ir boutons of the NTG-treated and control groups were 1.9 ± 1.2 µm2 and 1.3 ± 1.0 µm2, respectively (P < 0.01, Mann–Whitney test).
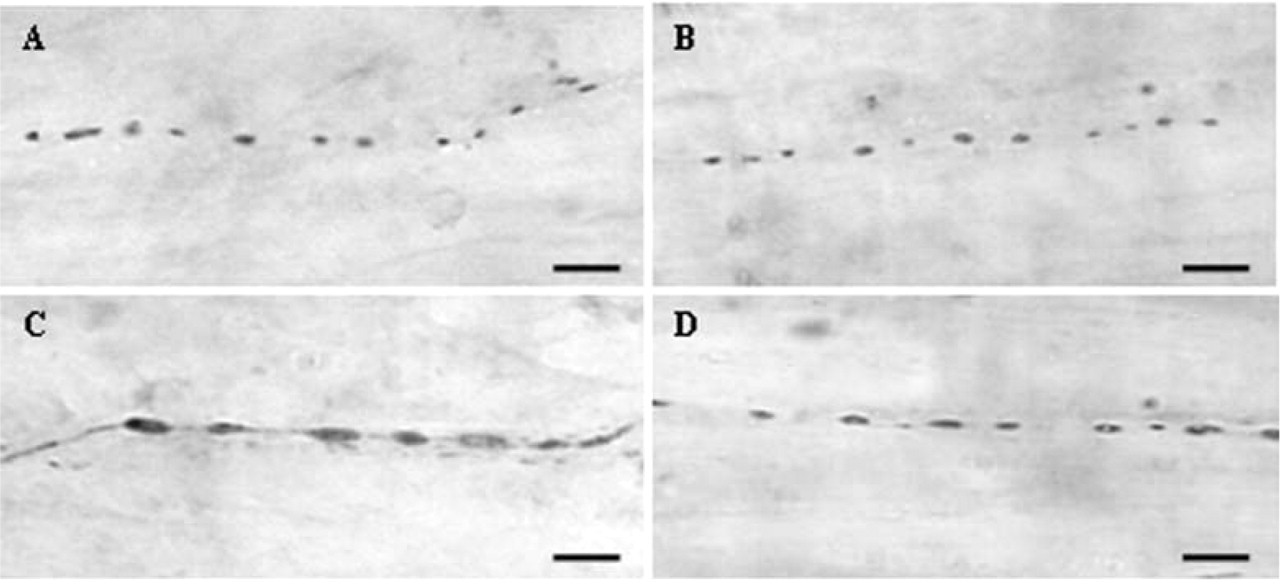
The effect of sumatriptan on nitroglycerin (NTG)-evoked neuronal nitric oxide synthase (nNOS) expression in perivascular nerve fibres. Infusion of NTG can enlarge the perivascular nNOS-ir varicosities. This effect can be blocked by sumatriptan pretreatment. A, Control; B, sumatriptan alone; C, NTG without sumatriptan pretreatment; D, NTG with sumatriptan pretreatment. Bars = 5 µm.
Pretreatment with sumatriptan prevented NTG-evoked nNOS-ir expression in all areas studied. The surface area of nNOS-ir fibre in the sumatriptan-pretreated group was 1.5 ± 0.9 µm2 compared with 1.9 ± 1.2 µm2 in those without pretreatment. The difference between animals receiving NTG with and without sumatriptan pretreatment was statistically significant (P < 0.01, Mann–Whitney test). The number of NTG-evoked nNOS-ir cells in trigeminal ganglia and TNC in sumatriptan-pretreated rats was also significantly lower than those observed in rats receiving no pretreatment. No significant change was evident in the group receiving sumatriptan alone (Figs 4 and 5 and Table 1).

The effect of sumatriptan on nitroglycerin (NTG)-evoked neuronal nitric oxide synthase (nNOS) expression in trigeminal ganglia. Infusion of NTG can increase the number of nNOS-ir neurones in trigeminal ganglia. This effect can be attenuated by sumatriptan pretreatment. A, Control; B, sumatriptan alone; C, NTG without sumatriptan pretreatment; D, NTG with sumatriptan pretreatment. Bars = 50 µm.
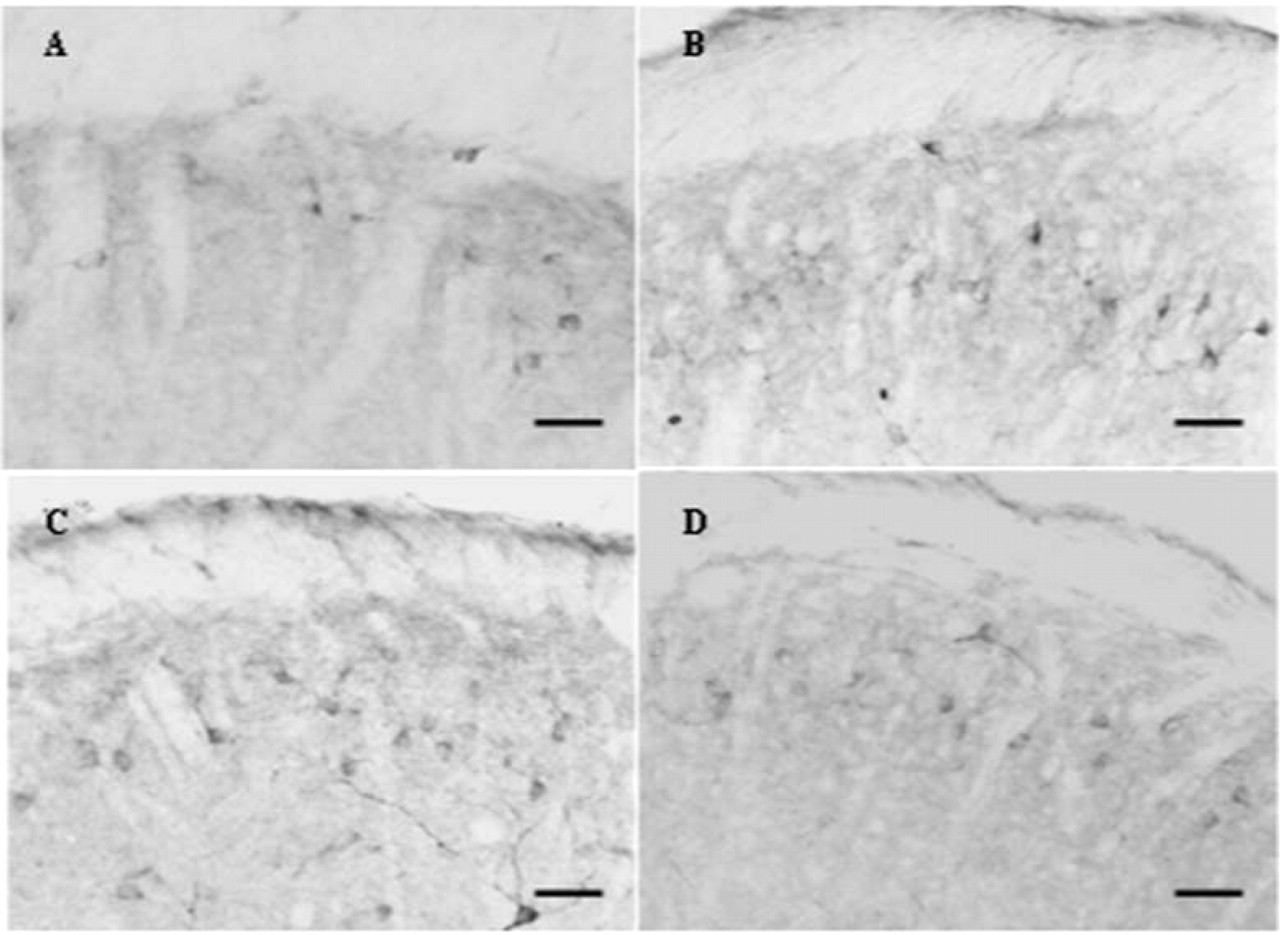
The effect of sumatriptan on nitroglycerin (NTG)-evoked neuronal nitric oxide synthase (nNOS) expression in trigeminal nucleus caudalis (TNC). Infusion of NTG can increase the number of nNOS-ir neurones in TNC. This effect can be attenuated by sumatriptan pretreatment. A, Control; B, sumatriptan alone; C, NTG without sumatriptan pretreatment; D, NTG with sumatriptan pretreatment. Bars = 100 µm.
Effect of 5-HT1B/1D agonist and nitroglycerin (NTG) on neuronal nitric oxide synthase (nNOS) expression in the trigeminovascular pathway

Data are expressed as mean ± SD.
∗Significant difference between group receiving saline + NTG and the others (P < 0.01, Kruskal–Wallis test).
Discussion
Activation of 5-HT1B/1D receptor is the primary target in aborting migraine attacks. Agonists to these receptors abort migraine headache via several mechanisms. These include constricting cranial vessels, inhibiting the release of algogenic substances and stabilizing thalamic projection neurones in the TNC. The present study shows that, in addition to previously described mechanisms, 5-HT1B/1D receptor agonists can minimize the effect of NTG on cerebral blood flow and nNOS expression in the trigeminal neurones and perivascular nerve fibres. Therefore, modulation of NO transmission in the trigeminal pathway may be another mechanism underlying anti-migraine activity of 5-HT1B/1D receptor agonists. The finding of an attenuating effect of sumatriptan on NTG-evoked nNOS expression in TNC also suggests that this substance may also act centrally.
NO is an important transmitter involved in the development of migraine pain. Clinically, headache is a common adverse effect occurring during infusion of NTG, a vasodilator and NO donor. Infusion of NTG can trigger migraine attacks in migraine patients and can also induce attacks of migraine without aura in patients who usually have migraine with aura (10). The involvement of NO in clinical migraine is further supported by an observation of increased levels of nitrate metabolites and cGMP in jugular venous blood taken from migraine patients during their attacks (2). In animal studies, infusion of NTG can induce c-fos expression in trigeminal caudalis neurones, implying activation of the cranial nociceptive system (11). Noteworthy, some of these NTG-evoked c-fos immunoreactive neurones were also NOS immunoreactive. The observation of the NTG-induced expression of nNOS-ir has been reported previously in TNC 4 h after NTG infusion (12). Also, 4 h after NTG exposure, Reuter et al. (13) reported an increased expression of inducible NOS in meningeal microglia. Here, we show that the changes in nNOS expression can occur within a shorter period (1 h), a finding possibly explained by differences in the dose of NTG used between the experiments. In Reuter's study (13), NTG was intravenously infused at a dose of 2 µg/kg per min for 30 min, therefore the total dose was usually less than 15 µg. Pardutz et al. (12) used NTG of the same dose as ours (10 mg/kg), but the drug was given subcutaneously. Lower dose of NTG may induce a more gradual response and may explain the absence of NOS expression in the period before 4 h. A finding of increased NOS immunoreactivity after exposure to NTG indirectly supports the hypothesis that exposure to exogenous NO donor can trigger endogenous nNOS activity and hence increasing the NO production.
We also showed that sumatriptan can shorten the duration of NTG-evoked cerebral hyperaemia without changing the basal cortical blood flow. In rats pretreated with sumatriptan, cortical blood flow was normalized 15 min post-NTG infusion, whilst it was still increased in the saline-pretreated rats. Given the hypothesis that NTG can induce endogenous NO production, this change implied that sumatriptan modified the NTG-induced cerebral hyperaemia by reducing endogenous NO synthesis. This hypothesis was confirmed by our immunohistochemical study. Indeed, we found that sumatriptan was able to attenuate the magnitude of NTG-induced enlargement of the perivascular nNOS-ir varicosities, and the number of cell expressing nNOS in the trigeminal ganglia and TNC. Such an effect of sumatriptan on TNC may suggest that, at high dose of NTG, the cerebral capillaries increase their permeability and disrupt the blood–brain barrier, as suggested by our previous observation (3). It should be noted that pretreatment with sumatriptan did not minimize the amplitude of the initial peak of NTG-induced cerebral hyperaemia, possibly due to the high dose of NTG used in this study.
Modification of the NO system by triptan compound has been previously demonstrated. In 1999, Read et al. showed that administration of NTG led to increased NO and decreased superoxide concentrations. They also found that sumatriptan was able to stabilize the level of superoxide (14). More recently, the same group found that sumatriptan pretreatment attenuated all phases of NO release associated the cortical spreading depression and reduced cortical NO level (15). It was also shown that sumatriptan could partially block NO-induced meningeal vasodilation (16), possibly by inhibiting trigeminal activation and calcitonin gene-related peptide release. 5-HT1B/1D agonists can also abolish the N-methyl-D-aspartate-evoked enhancement of NOS activity and cGMP concentration (17). Our NOS immunohistochemical study thus confirms the potential inhibitory effect of 5-HT1B/1D receptor agonist on NO production.
Despite the above evidence favouring the effect of 5-HT1B/1D receptor agonists in minimizing NO production or antagonizing NO effect, some contradictory evidence was also reported. For example, Ayajiki et al. (18) showed that sumatriptan counteracted the neurogenic relaxation in canine cerebral arterial strips without interfering with NO release. It was also shown that sumatriptan failed to abolish the cortical spreading depression-evoked cGMP accumulation (15).
In conclusion, this study showed that activation of 5-HT1B/1D receptor could reduce the stimulating effect of NO donor on cerebral hyperaemia and nNOS expression in the trigeminal system. This finding may offer an additional explanation for the therapeutic effect of triptan compounds in aborting the migraine headache, and suggests that modulation of NO production may be another potential target in development of migraine drugs.
Footnotes
Acknowledgements
The authors gratefully acknowledged Glaxo Welcome Co. for generous supply of sumatriptan. This study was partly supported by a grant from the Minister of University Affairs of Thailand.
