Abstract

Introduction
Chronic tension-type headache is one of the most common and important types of primary headaches (1) and represents a considerable health and socio-economic problem (2). Increased tenderness of pericranial myofascial tissues to manual palpation is the most prominent abnormal finding in patients with chronic tension-type headache (3–6). Painful impulses from these tissues may be referred to the head and perceived as headache, and myofascial mechanisms may therefore play a major role in the pathophysiology of tension-type headache (7). Progress in molecular neurobiology of pain (8) and an increasing number of studies on tension-type headache (9) have increased our knowledge about the mechanisms underlying chronic head pain. Thus, substantial experimental evidence indicates that central sensitization, i.e. increased excitability of neurons in the central nervous system (CNS) generated by prolonged nociceptive input from the pericranial myofascial tissues, plays an important role in the pathophysiology of chronic pain (8) and chronic tension-type headache (9). Furthermore, discovery of neurotransmitters and neuromodulators such as nitric oxide (NO), calcitonin gene-related peptide (CGRP), substance P (SP), neuropeptide Y (NPY) and vasoactive intestinal polypeptide (VIP) involved in the pain processing provides new insights to our understanding of the biology of chronic head pain. To explore the neurobiology of human chronic pain conditions, it is necessary to utilize advances made in basic research. The purpose of the present thesis was to study the neurobiology of chronic tension-type headache. Specific aims were: (i) to investigate NO mechanisms in chronic tension-type headache sufferers; (ii) to study plasma levels of CGRP, SP, NPY and VIP in patients with chronic tension-type headache; and (iii) to study in vivo skeletal muscle blood flow during static exercise in patients with chronic tension-type headache.
Nitric oxide in chronic tension-type headache
Biosynthesis of nitric oxide
The free radical NO is a messenger molecule involved in various biological functions (10–12). NO is synthesized by a complex family of nitric oxide synthase (NOS) enzymes (13). Because NO is highly reactive and unstable, much of the research on its functions is based on characterization of NOS. Three distinct NOS enzymes [neuronal NOS (nNOS), endothelial NOS, inducible NOS) have been purified, cloned and biochemically characterized (13). The precise mechanism of NO formation is not fully understood. It is known that NO is synthesized from L-arginine and that the reaction also yields citrulline (Fig. 1).
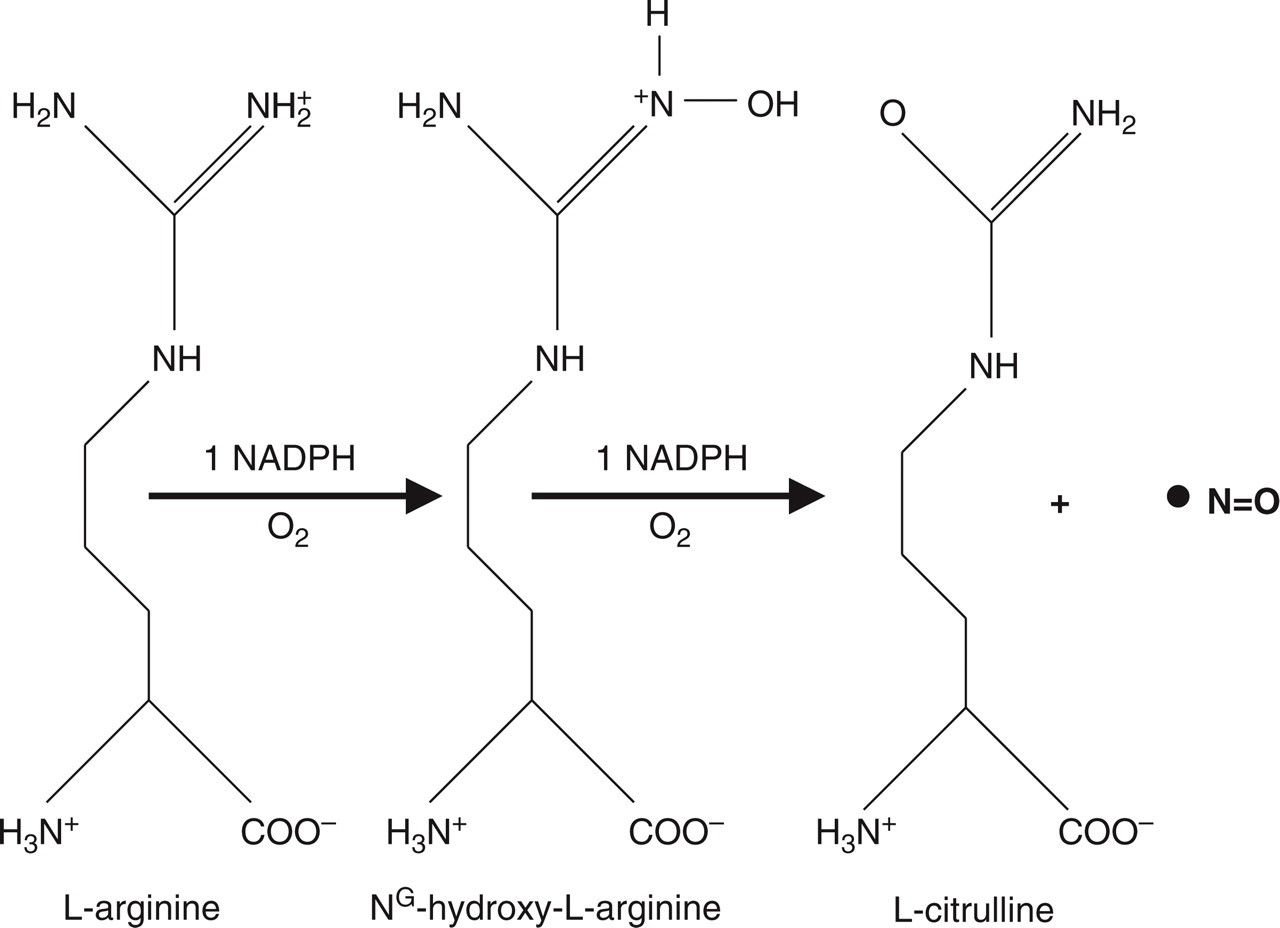
Nitric oxide synthase-catalysed oxidation of L-arginine. Nitric oxide is synthesized from L-arginine and the reaction also yields citrulline. (Reproduced from ‘Nitric oxide in the nervous system’. In: V. B. Mayer, editor. Biochemistry and molecular pharmacology of nitric oxide synthases, Chapter 2, 1995:21–38, with permission from Elsevier.)
NO and nociception in animal studies
NO or NOS immunoreactivity has been identified in the peripheral and central nervous system. In the peripheral nervous system, nNOS immunoreactivity was demonstrated in dorsal root ganglia in both man and rat (14) and in perivascular nerves of large cerebral arteries of the rat (15) and human brain (16). In the central nervous system, nNOS is present exclusively in neurons and NOS neurons were demonstrated in the spinal trigeminal nucleus (17) and in the dorsal horn of the spinal cord (14, 18).
NO derived from neurons was first recognized when N-methyl-D-aspartate (NMDA) receptor activation in cerebellar cultures resulted in NO generation (19). Involvement of NO in neuronal signalling was confirmed by demonstrating that NOS inhibitors blocked the stimulation of cyclic guanosine monophosphate in brain that is associated with activation of NMDA receptor (20, 21). Later, it was demonstrated that sensitization of pain pathways in the spinal cord may be caused by or associated with activation of nNOS and the generation of NO (22–24). Moreover, prolonged elevation of NO levels within the spinal dorsal horn is important in maintaining the central sensitization (25). Finally, it has been shown that inhibition of NOS reduces central sensitization in pain models (22, 26, 27) and that nociceptive responses in these models are enhanced by NO donors (28, 29). Taken together, these data suggest that NO is an important transmitter in pain pathways of the spinal cord and that NO contributes to development and maintenance of central sensitization at the spinal level.
Central sensitization and chronic tension-type headache
Nociception from pericranial myofascial tissues may play a major role in the pathophysiology of tension-type headache. Thus, several studies have consistently reported increased myofascial tenderness as the most prominent abnormal finding in patients with chronic tension-type headache (3–6). Further support for myofascial involvement is the finding of increased muscle hardness (30) and a positive correlation between muscle hardness and tenderness in chronic tension-type headache (31). In addition to findings in the periphery, chronic tension-type headache sufferers also exhibit signs of increased sensitivity in the CNS. Thus, pressure pain detection and tolerance thresholds to mechanical stimuli have been found decreased in these patients (32, 33). Furthermore, Bendtsen et al. (34) demonstrated that patients with chronic tension-type headache had a qualitatively altered pain perception. On the basis of these findings and data from basic pain research (35), it has been suggested that the central sensitization and thereby the chronic pain state in patients with chronic tension-type headache may be due to sensitization at the level of the spinal dorsal horn/trigeminal nucleus induced by prolonged nociceptive input from pericranial myofascial tissues (9, 36).
Inhibition of NOS in chronic tension-type headache
In order to test the hypothesis that inhibition of NO and thereby central sensitization would reduce chronic headache, Ashina et al. (37) investigated the analgesic effect of the NOS inhibitor L-NG methyl arginine hydrochloride (L-NMMA) in patients with chronic tension-type headache. In a double-blind, placebo-controlled crossover study patients received L-NMMA or placebo on 2 days. L-NMMA reduced headache intensity significantly more than placebo (Fig. 2). To explore the mechanisms of this analgesic effect Ashina et al. (38) also studied the relationship between myofascial factors and NOS inhibition. This study showed that both muscle hardness and tenderness were significantly reduced following treatment with L-NMMA, while there was no significant reduction at any time after treatment with placebo (Figs 3 and 4). The muscle hardness was significantly reduced following treatment with L-NMMA compared with placebo. The reduction in tenderness following treatment with L-NMMA did not reach statistical significance compared with placebo. The pressure pain detection thresholds in the finger and temporal region were largely unchanged following treatment with L-NMMA. Thus, these studies (37, 38) demonstrated that the NOS inhibitor, L-NMMA, reduces headache intensity and muscle hardness in patients with chronic tension-type headache.

Percent changes from baseline pain intensity on a 100-mm visual analogue scale (VAS) in 16 patients with chronic tension-type headache. The pain intensity was significantly more reduced following treatment with L-NMMA (▪) compared with placebo (□) (P = 0.01). ∗P < 0.05 compared with baseline (time = 0). The plots represent mean scores. (Modified from Ashina et al. 1999, by permission of the Lancet.)

Percent changes in muscle hardness in 16 patients with chronic tension-type headache. Muscle hardness was significantly more reduced following treatment with L-NMMA (▪) than with placebo (□) in patients with chronic myofascial pain (P = 0.04). ∗P < 0.05 compared with baseline (time = 0). The plots represent mean scores. (Reproduced from Ashina et al. 1999, by permission of Oxford University Press.)

Percent changes in total tenderness score (TTS) in 16 patients with chronic tension-type headache. The TTS tended to be reduced following treatment with L-NMMA (▪) compared with placebo (□) (P = 0.11). Within each treatment, the TTS was significantly reduced at 60 and 120 min after start of the infusion of L-NMMA, while there was no significant change at any time after treatment with placebo. ∗∗P < 0.01 compared with baseline (time = 0). The plots represent mean scores. (Reproduced from Ashina et al. 1999, by permission of Oxford University Press.)
An important question is how L-NMMA modulates myofascial factors in patients with chronic tension-type headache and whether the effects of L-NMMA are due to an action in muscle, peripheral nerves or the CNS. It is well established that persistent activity in peripheral nociceptors may lead to sensitization of neurons in the spinal dorsal horn, partly via activation of NMDA receptors (8). Many of the effects of NMDA receptor activation are mediated via production of NO (23) and, as described earlier, animal models of persistent pain have shown that inhibitors of NOS decrease sensitization of the spinal dorsal horn induced by continuous painful input from the periphery (22, 24, 26, 27). On the basis of these findings Ashina et al. (37, 38) suggested that the anti-nociceptive effect of NOS inhibition in patients with chronic tension-type headache is probably due to reduction of central sensitization at the level of the spinal dorsal horn/trigeminal nucleus. One should, however, also consider other possible mechanisms of action. Thus, it is possible that L-NMMA has direct anti-nociceptive effects in myofascial tissues. It has been demonstrated that NOS inhibitors have anti-nociceptive effects after peripheral administration, probably due to inhibition of endothelial NOS (eNOS) (22, 39, 40). However, the exact role of NO in the periphery is still far from understood, and additional research is needed to clarify whether NO may activate or sensitize nociceptors in myofascial tissues.
The anti-nociceptive effect of L-NMMA might also be due to blocking of vascular input. Excessive vascular nociception may contribute to a primary myofascial nociception in patients with tension-type headache (41). L-NMMA inhibits all three types of NOS, including eNOS. Thus, it is possible that L-NMMA exerts its action by blocking eNOS and thereby moderate vasodilatation of cephalic/extracephalic arteries in patients with chronic tension-type headache. Because of convergence of nociceptive input from facial tissues at the spinal/trigeminal level (42), blocking of vascular input might also lead to reduction of myofascial nociception from pericranial muscles (38).
Taken together, these data indicate that the NOS inhibitor L-NMMA elicits its anti-nociceptive effect in chronic tension-type headache by modulation of nociceptive information from myofascial tissues. This anti-nociceptive effect may mainly be due to reduction of central sensitization at the level of the spinal dorsal horn or trigeminal nucleus, or both.
NO induction in chronic tension-type headache
In 1989, Iversen and colleagues introduced a NO donor, glyceryl trinitrate (GTN), model of experimental headache (43). Using this model, it has been demonstrated that patients with migraine are hypersensitive to NO, i.e. migraineurs develop significantly stronger headache after GTN infusion than healthy subjects (44, 45). Furthermore, it has been shown that GTN may induce strong immediate headache in patients with episodic tension-type headache compared with healthy subjects (44). To explore further the role of NO in development of headache and modulation of myofascial pain input, Ashina et al. (46) studied the effect of GTN in patients with chronic tension-type headache. GTN infusion in patients resulted in a biphasic response with an immediate and a delayed headache (46). Patients developed significantly stronger immediate and delayed headache on a GTN day than on a placebo day (Fig. 5). Furthermore, patients developed significantly stronger headache after GTN than controls (Fig. 5).

Median headache intensity over time during (20 min) and after infusion of glyceryl trinitrate (GTN) (▪, patients; •, controls) and placebo (□, patients; Δ, controls) in 16 patients with chronic tension-type headache and in 16 healthy subjects. Headache was scored on a 10-point verbal rating scale (VRS). The patients developed significantly stronger headache than healthy controls both during the first hour (immediate headache) (P = 0.02) and during the subsequent 11 h (delayed headache) (P = 0.008). (Modified from Ashina et al. 2000, by permission of Oxford University Press.)
The mechanisms responsible for the GTN-induced headache in patients with primary headaches are unknown. CGRP is an important neuromodulator of the sensory system (47). An experimental study has shown that GTN dilates cerebral arteries in cats via liberation of CGRP, and it has been suggested that this mechanism may explain the occurrence of vasodilatation and headache in humans (48, 49). However, this effect of GTN was not confirmed in isolated guinea pig basilar arteries (50). Furthermore, Iversen and colleagues (51) reported that plasma levels of CGRP are unchanged in the cranial circulation of healthy subjects after GTN infusion. To study the role of CGRP in NO-induced immediate headache, Ashina et al. (52) measured plasma levels of CGRP during and after infusion of GTN in patients with chronic tension-type headache (52). No significant changes in plasma CGRP after GTN infusion were found in either patients or controls. Interestingly, the dosage of GTN used in that study is known to liberate CGRP in cats (48) and vasodilatation in humans (53).
The unchanged sensitivity of pericranial myofascial pain pathways seems also to rule out sensitization of myofascial peripheral and central pathways as a mechanism of the immediate headache (52). Unchanged pressure-pain detection thresholds in the finger, i.e. outside of the pain area, may also indicate no alteration in sensitivity of third-order neurons (52).
NO evokes pain in humans when injected paravascularly or perfused through a vascularly isolated hand vein segment (54). These findings suggest that NO may directly activate or sensitize nociceptors around blood vessels. Intravenous infusion of GTN induces dilatation of the middle cerebral artery in healthy subjects (53), and in migraineurs and patients with episodic tension-type headache (49). In these studies the dilatation lasted at least until 1 h after cessation of GTN infusion and 3 h in another study (55). Collectively, these studies suggest that immediate headache after GTN infusion in patients with chronic tension-type headache may originate from NO-induced activation or sensitization of sensory nerves around cephalic arteries, or from NO-induced arterial dilatation, or both (46).
The most important finding in the study by Ashina et al. (46) was that systemic administration of NO donor in patients with chronic tension-type headache resulted in biphasic response with an immediate and a delayed headache (8 h after start of infusion) (Fig. 5). Interestingly, the time profile of the GTN-induced headache in patients with chronic tension-type headache was strikingly similar to the time profile of GTN-induced headache in patients with migraine (45). Thus, patients with migraine without aura developed an immediate headache during GTN infusion and a delayed headache fulfilling International Headache Society (IHS) (56) criteria for migraine several hours after cessation of the infusion. The characteristics of the delayed headache in chronic tension-type headache were, however, different from those in patients with migraine. Eighty percent of migraine patients developed migraine without aura after infusion of GTN (45), while 87% of patients with chronic tension-type headache developed a tension-type headache (46). These data suggest that patients with chronic tension-type headache are supersensitive to NO, similar to patients with migraine, and that the majority of patients in both groups develop their usual headache several hours after the infusion of GTN.
What are the mechanisms of the delayed headache and why do patients with chronic tension-type headache and patients with migraine develop delayed headache resembling their usual type of headache, and why do most healthy subjects develop no delayed headache or only a minor one? Studies in rats and in humans have shown that after intravenous administration of GTN very little drug remains in the blood and that the majority of the GTN is distributed to tissues (57). In the anaesthetized cat, intravenous infusion of GTN induces a prolonged (60 min) increase of NO in brain parenchyma (58), and a prolonged increase of NO levels in the spinal dorsal horn was demonstrated during central sensitization (59). Furthermore, Wu and colleagues (60) demonstrated that during central sensitization both endogenous and exogenous nitric oxide induce c-fos (an immediate–early gene) which can further activate the production of other substances in the CNS. In addition, Pardutz et al. (61) reported that subcutaneous GTN produced a significant increase of NOS- and c-fos-immunoreactive neurons in the cervical part of trigeminal nucleus caudalis in rats after 4 h. These data indicate that GTN infusion may result in storage and subsequent liberation of NO or it may trigger endogenous NO production in the CNS, thereby enhancing sensitization of nociceptive pathways in the CNS of patients with chronic tension-type headache.
Alternatively, sustained NO-induced vascular nociception may lead to central sensitization and subsequent convergence of nociceptive input from blood vessels and myofascial tissue. Thus, NO may activate or sensitize nociceptors around blood vessels directly (54) or by dilatation (53). Dilatation of meningeal blood vessels in rats causes sensitization of central trigeminal neurons and facilitation of convergent sensory responses (62). It is therefore possible that excessive vascular nociception caused by GTN may gradually augment the sensitizing effect of preexisting myofascial input in chronic tension-type headache sufferers (41).
As mentioned earlier, patients with chronic tension-type headache and patients with migraine develop increased delayed headache with different characteristics. The most likely explanation is that preexisting facilitation of distinct nociceptive central pathways in chronic tension-type headache sufferers (myofascial pathways) and migraineurs (vascular pathways) may be enhanced by NO-induced central sensitization. This may explain why the delayed headache fulfilled tension-type headache criteria in patients with chronic tension-type headache and migraine criteria in migraineurs. This could also explain why NO does not induce strong delayed headache in healthy subjects when no preexisting sensitization is present.
Summary
Studies of NO mechanisms in chronic tension-type headache suggest that NO plays an important role in the pathophysiology of this disorder (37, 38, 46). The anti-nociceptive effect of NOS inhibitor suggests that inhibition of NOS may become a novel principle in treatment of chronic tension-type headache. It is probable that the anti-nociceptive effect is due to reduction of central sensitization at the level of the spinal dorsal horn or trigeminal nucleus, or both. Studies with selective NOS inhibitors are needed to determine which type of NOS is involved and its exact site of action in chronic tension-type headache. Data from the GTN model of experimental headache indicate that NO-induced delayed headache in patients with chronic tension-type headache is due to augmentation of preexisting central sensitization. Moreover, these data indicate that NO contributes to mechanisms of several types of primary headaches and that NO-related central sensitization may be an important common denominator in the pain mechanisms of primary headaches, although their basic pathophysiological mechanisms are different.
Neuropeptides in chronic tension-type headache
CGRP, SP, NPY, VIP and nociception in animal studies
Immunohistochemical studies have shown that CGRP, SP, NPY, VIP are present in the peripheral and central nervous system (47, 63–69). Experimental studies demonstrated that acute and chronic nociception leads to altered release of CGRP, SP, NPY and VIP from sensory nerve endings and from central terminals of sensory afferents into dorsal horn of the spinal cord (70–73). These data suggest that CGRP, SP, NPY and VIP are important neurotransmitters or neuromodulators in the nervous system and that these neuropeptides may play a role in the process of central sensitization.
CGRP, SP, NPY, VIP in chronic tension-type headache
In the last 10 years there has been increasing interest in the role of neuropeptides in primary headaches. Particularly, a role for CGRP has been implicated in the pathophysiology of migraine (74, 75) and cluster headache (76, 77). However, the role of CGRP in generating headache pain is still unclear (75). Studies of SP, NPY and VIP in patients with primary headaches or other chronic pain conditions have not led to consistent results (78). This is probably due to variations in methodology [different methods of analysis and/or field of sampling (plasma, platelets, CSF, saliva)] or patient populations.
As mentioned above, nociception from the pericranial myofascial tissues may be of importance in the pathophysiology of chronic tension-type headache. In rat muscle, CGRP sensory fibres are preferentially located in the wall of arteries (79) and nerve fibres containing CGRP, SP, NPY and VIP accompany small blood vessels in human cranial muscles (80). These findings indicate that ongoing activity in sensory neurons in the cranial muscles may be reflected in changes of plasma levels of neuropeptides in patients with chronic tension-type headache. The role of neuropeptides in chronic tension-type headache has been investigated in three studies (78, 81, 82). Bach and colleagues (81) reported normal CGRP levels in the cerebrospinal fluid (CSF) in patients with chronic tension-type headache. It was not reported whether patients were examined during headache or in headache-free period or whether there was any relationship between CGRP levels and headache quality. To explore a possible role of neuropeptides in chronic tension-type headache, Ashina and colleagues measured plasma levels of CGRP (82) and SP, NPY and VIP (78) in the cranial and peripheral circulation of patients and controls. These studies showed that plasma levels of CGRP, SP, NPY and VIP are normal in both cranial and peripheral circulation of patients. Moreover, plasma levels of neuropeptides were largely unrelated to headache state (78, 82). Findings of normal plasma CGRP are particularly important because they are clearly different from our previous study in migraine patients with elevated levels of CGRP (75). However, exploratory testing in relation to headache characteristics showed that eight patients with a pulsating pain quality, although fulfilling the IHS criteria for tension-type headache and not for migraine, had higher plasma CGRP in the headache-free period than controls (82). Plasma levels of CGRP in patients with predominantly pressing headache in the past did not differ from plasma CGRP in controls. In addition, there was no relationship between CGRP levels and muscular factors (82).
How can we explain increased plasma CGRP in chronic tension-type headache patients with pulsating headache quality? Since CGRP levels are increased in migraineurs (75) and in patients with pulsating headache in the headache-free period (82), ongoing nociception from cephalic or extracephalic vasculature, or both, seems to be ruled out. Circulating CGRP may be involved in the regulation of blood flow and in the maintenance of vascular tone (83). It is possible that increased interictal CGRP levels in patients with pulsating headache like migraineurs may reflect altered vascular control due to abnormal release of CGRP from sensory neurons (75). Therefore, the most likely explanation is that these patients, although fulfilling the IHS criteria for chronic tension-type headache and not having any migraine history, are in fact pathophysiologically related to migraine. Although the finding of increased plasma CGRP in patients with pulsating headache quality is very interesting, one should be very cautious with interpretation of post hoc analysis, particularly with small numbers of patients. Therefore, a prospective study with a large number of patients is required to confirm this observation.
Summary
Studies on the role of neuropeptides in chronic tension-type headache indicate that plasma levels of CGRP, SP, NPY and VIP are normal in patients and largely unrelated to headache state (78, 82). Furthermore, findings of normal plasma CGRP add to the growing list of differences between migraine and tension-type headache. It is possible that changes of neuropeptide levels at the spinal/trigeminal level or in the myofascial tissue of these patients do not reach the cranial or peripheral circulation. It would have been optimal to measure SP, NPY and VIP levels in the pericranial muscles and CSF or even at the neuronal level. However, this is difficult for both practical and ethical reasons. Thus, the findings of normal levels of CGRP, SP, NPY and VIP in the cranial and peripheral circulation do not exclude that abnormalities of these neuropeptides at the neuronal or peripheral (pericranial muscle) levels play a role for the pathophysiology of chronic tension-type headache. Investigation of neuropeptides in other compartments with new sensitive methods of analysis is necessary to clarify their role in chronic tension-type headache. Finally, future CGRP studies in chronic tension-type headache sufferers with pulsating pain quality and in migraine patients with pressing pain quality, but otherwise fulfilling the IHS migraine criteria, are required to clarify whether increased CGRP levels are associated with pulsating pain quality.
Microdialysis and chronic tension-type headache
Muscle pathology in tension-type headache?
It is a common experience for individuals who have been exposed to static or repetitive work for a long period to develop tender areas in the pericranial muscles and tension-type headache. It has been hypothesized that local muscle ischaemia, disturbances in metabolism, microcirculation and mitochondria function in the tender areas may explain myofascial pain in tension-type headache and in other myofascial pain disorders such as trapezius myalgia (84). Various in vitro and in vivo methods, such as muscle biopsy, single-fibre laser-Doppler and magnetic resonance spectroscopy, have been used to explore the mechanisms responsible for myofascial pain. The results of these studies have been conflicting. While open studies suggested abnormalities in microcirculation (85, 86), controlled and blinded studies failed to find firm evidence of peripheral abnormalities (87) in patients with chronic myofascial pain (88). Using 133xenon clearance technique, Langemark and co-authors (89) found normal resting blood flow and relative flow increase during isometric work in temporal muscle in patients with chronic tension-type headache. However, in that study muscle blood flow was measured from a large muscle area and not in a tender point. Thus, firm evidence of peripheral muscle pathology as a cause of muscle pain and chronic headache is still lacking. More sensitive techniques are needed to answer the question of whether tension-type headache and other myofascial pain disorders are associated with peripheral pathology in tender points.
Muscle blood flow in chronic tension-type headache
Microdialysis is a unique technique for investigating and monitoring local muscle blood flow and metabolism in vivo within a tissue volume of < 1 cm3 (90). Using the microdialysis technique, Ashina and collaborators (91) estimated blood flow and interstitial lactate concentrations in the trapezius muscle at rest and in response to static exercise in patients with chronic tension-type headache. The major finding of that study was a decreased blood flow in response to static exercise in a tender point in patients. Thus, the increase in muscle blood flow from baseline to exercise and post-exercise periods was significantly lower in patients than in controls (Fig. 6) (91). There was no difference in resting blood flow between patients and controls.
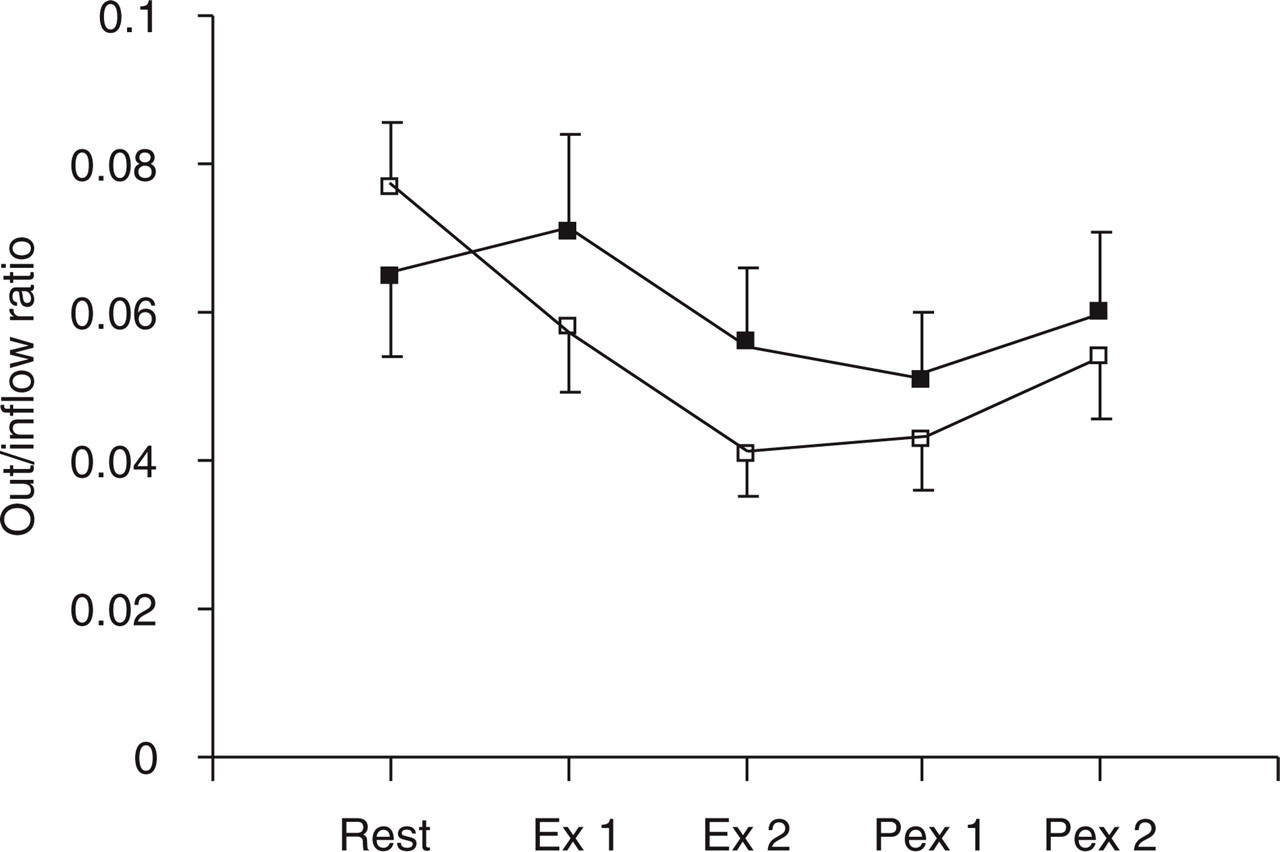
Mean nutritive muscle blood flow in 16 patients with chronic tension-type headache and in 17 healthy control subjects. The figure shows that the increase in muscle blood flow from baseline (Rest) to exercise (Ex 1 and Ex 2) and post-exercise periods (Pex 1 and Pex 2) was significantly lower in patients (▪) than in controls (□) (P = 0.03). The plots represent mean ± SEM scores. (Reproduced from Ashina et al. 2002, by permission of Oxford University Press.)
The question is, how do we explain the reduced blood flow response to exercise in the tender point? Difference in the static load between patients and controls is unlikely because there was no difference between groups, in either absolute force or relative to maximal voluntary force. It could be suggested that patients develop a relative ischaemia in the tender point during static exercise. If so, one would expect that the increase of interstitial lactate concentration would be higher in patients than in controls. However, Ashina and colleagues (91) observed no difference in local increase of interstitial lactate between patients and controls (Fig. 7). This seems to rule out the presence of ischaemia in the tender point of patients with chronic tension-type headache during rest and static exercise.

Mean interstitial concentration of lactate in 16 patients with chronic tension-type headache and in 17 healthy control subjects. There was no difference in change in interstitial concentration of lactate from baseline (Rest) to exercise (Ex 1 and Ex 2) and post-exercise (Pex 1 and Pex 2) periods between patients (▪) and controls (□) (P = 0.38). (Reproduced from Ashina et al. 2002, by permission of Oxford University Press.)
The altered blood flow might be secondary to chronic muscle pain. As mentioned in previous sections, chronic tension-type headache may be caused by prolonged painful input from pericranial myofascial tissues, e.g. tender points, resulting in central sensitization (36, 38). The pathophysiological basis for the painful input from the periphery is still unknown. Once the central sensitization had been established, chronic tension-type headache might be an entirely central process without further or only minimal input from the periphery (9). Because of central sensitization the central interpretation and response to normal sensory input are altered, possibly mainly when input is increased, as during exercise. This may lead to enhanced sympathetically mediated vasoconstriction and thereby a decreased blood flow in response to static exercise (91). This is supported by studies in animals and humans showing that static exercise produces a one-to-one synchronization of activation of the muscle nociceptors (group IV) and muscle nerve sympathetic activity (92). Furthermore, it has been shown that in patients with fibromyalgia a complete sympathetic blockade produced a marked reduction of the number of tender points, suggesting an improvement in microcirculation (93). Moreover, it has been proposed that central sensitization may maintain increased efferent sympathetic outflow that in turn maintains sensitization of sensory afferents (94). These data suggest that the sympathetic outflow may influence or maintain afferent activity in nociceptors or altered blood flow regulation in tender points of patients with chronic tension-type headache.
Summary
A microdialysis study provides in vivo evidence of altered blood flow regulation in tender skeletal muscle during static work in patients with chronic tension-type headache (91). The lack of any difference in local increase of interstitial lactate between patients and controls seems to rule out the presence of ischaemia in the tender point of patients with chronic tension-type headache during static exercise. It is possible that because of increased excitability of neurons in the CNS the central interpretation and response to normal sensory input are altered in patients with chronic tension-type headache. This may lead to enhanced sympathetically mediated vasoconstriction and thereby a decreased blood flow in response to static exercise.
Concluding remarks and future perspectives
Advances made in basic pain research have improved our knowledge of the neurobiology of chronic head pain. Particularly, understanding of molecular mechanisms involved in the process of central sensitization contributes to the development of novel therapeutic approaches for chronic headache. Studies of NO mechanisms suggest that NO may play a key role in the pathophysiology of this disorder and that the anti-nociceptive effect of NOS inhibitor, previously demonstrated only in animal models, may become a novel principle in future treatment of chronic headache. The anti-nociceptive effect of NOS inhibition is probably due to reduction of central sensitization at the level of the spinal dorsal horn or trigeminal nucleus, or both. However, the exact site of action and type of NOS involved in chronic tension-type headache is far from fully understood. Future studies with selective NOS inhibitors are needed to determine which type of NOS is involved and its exact site of action. Whether the anti-nociceptive effect observed in patients was clinically relevant is also debatable, but the importance of the results lies in the proof of the concept and not in the magnitude of the effect.
In the future, chronic dosing should be tested when a safe inhibitor of NOS with a longer half-life becomes available. Other important questions are whether inhibition of NOS would have analgesic effects in other chronic pain conditions, or whether the effect of inhibition of NOS in tension-type headache is due to interaction with specific headache mechanisms. These important issues should be addressed in the future studies in patients with other, non-headache, types of chronic pain. Data from the GTN model of experimental headache in chronic tension-type headache indicate that NO contributes to mechanisms of several types of primary headaches and that NO-related central sensitization may be an important common denominator in the pain mechanisms of primary headaches. However, additional research is needed to prove the role of central sensitization during NO-induced delayed headache. It would be interesting to detect possible sensitization of second-order neurons during the delayed headache by quantitative sensory testing techniques. Studies of the role of neuropeptides in chronic tension-type headache indicate that plasma levels of CGRP, SP, NPY and VIP are normal in patients and largely unrelated to headache state. Investigation of neuropeptides in other compartments, such as CSF or pericranial muscles, with new sensitive methods of analysis is necessary to clarify their role in chronic tension-type headache. Furthermore, CGRP studies in chronic tension-type headache sufferers with pulsating pain quality and in migraine patients with pressing pain quality are required to determine whether increased CGRP levels are associated with pulsating pain quality.
The results of microdialysis study indicate that central sensitization may lead to enhanced sympathetically mediated vasoconstriction which may be responsible for decreased blood flow in tender skeletal muscle during static work in patients with chronic tension-type headache. Thus, it is possible that the central neuroplastic changes may affect the regulation of peripheral mechanisms and thereby lead to increased tenderness and chronic headache. The study also shows that the microdialysis technique offers unique possibilities to study mechanisms responsible for not only head pain but also a wide range of myofascial pain conditions. Using this technique, it would be interesting to measure in vivo concentrations of inflammatory substances or neuropeptides in the tender muscles of patients with chronic tension-type headache.
In summary, the present study contributes to our understanding of the complex mechanisms leading to chronic tension-type headache and provides data that will hopefully lead to new treatment modalities.
Footnotes
Acknowledgements
The studies in this thesis were supported by grants from the University of Copenhagen, the Danish Headache Society, the Danish Medical Association Research Fund, the Danish Hospital Foundation for Medical Research, Region of Copenhagen, the Faroe Islands and Greenland, the Foundation for Research in Neurology, the Novo Nordisk Foundation, the Gerda and Aage Haensch's Foundation, the Mauritzen La Fontane's Foundation, and the Foundation of Jacob Madsen and his wife OIga Madsen.
