Abstract
Nitric oxide (NO) plays an important role in the pathophysiology of primary headaches including chronic tension-type headache (CTTH). Thus, a NO synthase inhibitor reduces headache and muscle hardness while the NO donor glyceryl trinitrate (GTN) causes more headache in patients than in healthy controls. Sensitization of myofascial pain pathways is important in CTTH, and the aim of the present study was to investigate if such mechanisms may also explain GTN-induced immediate headache in patients with CTTH. In a randomized, double-blind, crossover study 16 patients with CTTH and 16 healthy subjects received intravenous infusion of GTN (0.5 μg/kg per min for 20 min) or placebo on two headache-free days separated by at least 1 week. Muscle hardness, myofascial tenderness, mechanical and heat pain thresholds were measured at baseline and at 60 min and 120 min after start of infusion. In patients, GTN infusion resulted in a biphasic response with immediate headache and more pronounced delayed headache. A similar but less pronounced response was seen in controls. There was no difference between GTN and placebo regarding muscle hardness, myofascial tenderness or pressure and heat pain thresholds in either patients or controls (P > 0.05). The unchanged sensitivity of pericranial myofascial pain pathways indicates that peripheral and central sensitization is not involved in the mechanisms of GTN-induced immediate headache.
Keywords
Introduction
Clinical studies have shown that patients with chronic tension-type headache (CTTH) have a persistently increased tenderness to palpation of pericranial myofascial tissues (1–4) and significantly increased hardness of trapezius muscle (4, 5). These and other data (6, 7) demonstrate that ongoing myofascial nociception is important in this condition. In other studies we have shown that central sensitization, i.e. increased excitability of neurones in the central nervous system (CNS), may also be involved in the pathophysiology of chronic tension-type headache (8–10). Recently, we demonstrated that the nitric oxide (NO) donor glyceryl trinitrate (GTN) induces significantly stronger immediate and delayed headache in patients with CTTH than in healthy subjects (11). There are many indications for an arterial origin of GTN-induced immediate headache but no definite evidence (12). It is also unknown whether sensitization of myofascial pain pathways may be involved in mechanisms of the GTN-induced immediate headache. The aim of the present study was to investigate the mechanisms responsible for immediate headache after GTN infusion in patients with CTTH by recording muscle hardness, myofascial tenderness and pressure and heat pain thresholds.
Patients and methods
Subjects
Sixteen patients with a diagnosis of CTTH according to the criteria of the International Headache Society (IHS) (13) and 16 healthy subjects were included (Table 1). The patients were recruited from the out-patient headache clinic at Glostrup University Hospital without respect to presence or absence of myofascial tenderness. All patients underwent a general physical and a neurological examination and completed a diagnostic headache diary during a 4-week run-in period (14). Exclusion criteria were: history of migraine or any other type of primary headaches; any kind of daily medication (including prophylactic headache therapy but not oral contraceptives); excessive use of analgesics (corresponding to > 2 g of aspirin/day); serious somatic or psychiatric diseases including depression (Hamilton Depression Score ≥ 17 (15)). The healthy subjects had never had migraine and had less than 12 headache days per year.
Clinical data on headache patients and healthy controls
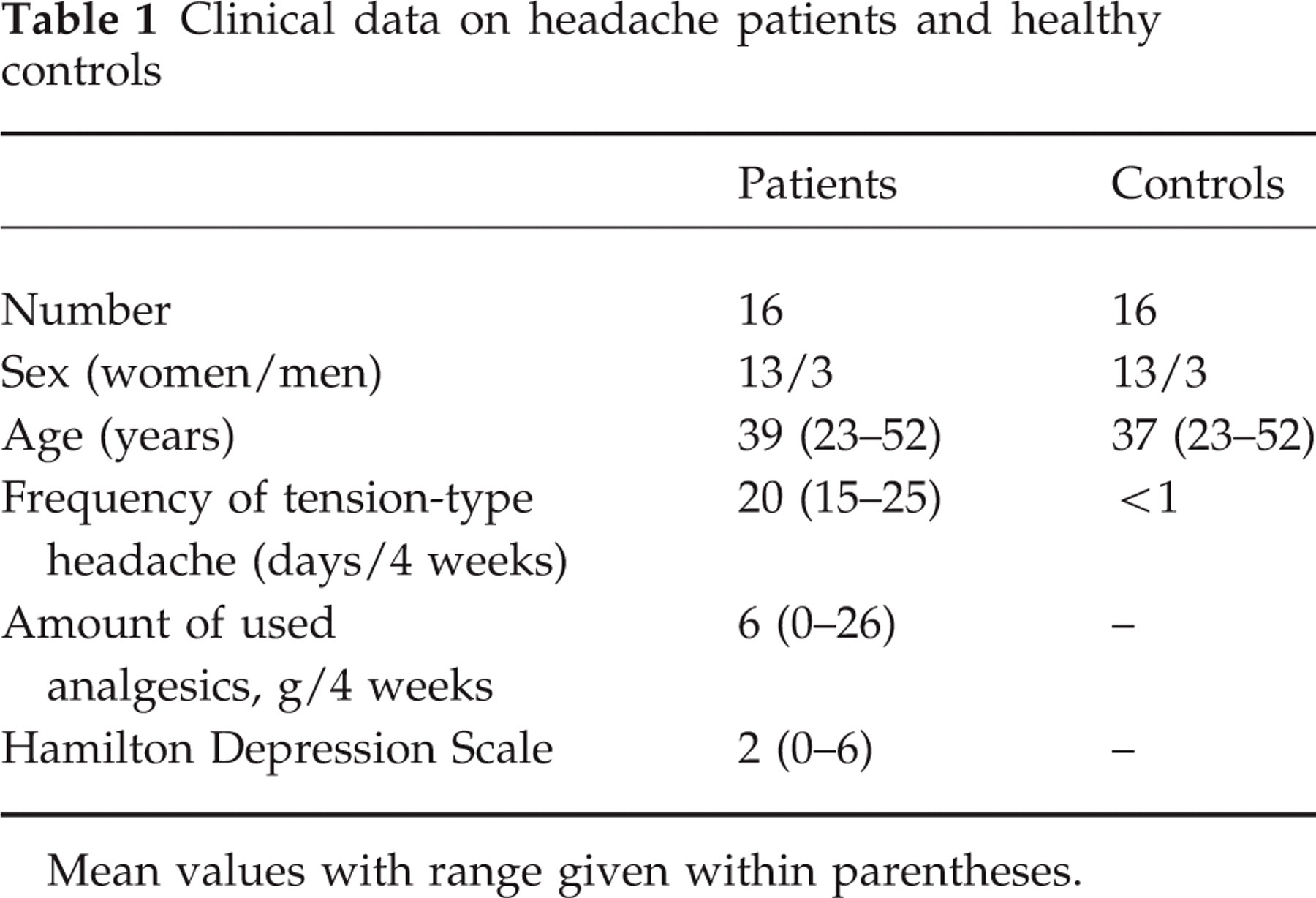
Mean values with range given within parentheses.
All subjects gave written consent to participate in the study, which was approved by the local ethics committee and conducted in accordance with the Declaration of Helsinki.
Apparatus
Muscle hardness
The hardness of the trapezius muscle was measured with a hardness meter, which has previously been described in detail (16). In brief, the hardness meter consists of a laser distance sensor and a pressure terminal with a surface area of 1 cm2. The muscle hardness is estimated by recording the relation between the applied pressure and the displacement of the skin over the muscle. All calculations are performed by a purpose-made software program in order to avoid observer bias. Hardness is expressed in kPa/cm. We have previously demonstrated that the hardness meter can measure muscle hardness reliably if the same observer is used throughout a study (17).
Pressure pain thresholds
An electronic pressure algometer (Somedic AB, Sweden) was used to measure pressure pain thresholds. A circular stimulation probe (0.5 cm2) and a pressure loading rate of 22 kPa/s (1 kPa = 103 N/m2) were used. The algometer has been described in detail elsewhere (18).
Heat pain thresholds
A Marstock thermotester (Somedic AB) was used to measure heat pain thresholds. It consists of a 54 × 28-mm heat stimulator placed in contact with the subject's skin and a push-button for the subject. The temperature increases gradually from the baseline temperature of 32°C by a rate of 1°C/s to a maximum of 52°C.
Procedure
The recordings were performed in a standardized manner by the same observer, a trained technician (H.A.), throughout the study. All variables were recorded at baseline, 60 min and 120 min after start of infusion. The first part of the study examined headache intensity, quality and time profile after infusion of GTN in patients with CTTH and compared GTN-induced headache between patients and controls and has previously been described in detail (11). Briefly, in a double-blind, placebo-controlled, crossover design 16 patients and 16 controls were randomly allocated to receive 0.5 μg/kg per min GTN or placebo (isotonic saline) over 20 min on 2 days separated by at least 1 week. All subjects were examined on days without headache at 09:00 a.m. and they had not suffered from any type of headache for at least 12 h prior to examination. The subjects were not allowed to take any kind of analgesics 24 h prior to examination. Headache intensity was measured on a Verbal Rating Scale (VRS) from 0 to 10 (0 = no headache; 5 = moderate headache; 10 = worst imaginable headache) during and after the start of the infusion.
Muscle hardness
Muscle hardness was measured at a standard anatomical point on the trapezius muscle on the non-dominant side, as previously described (17). Briefly, the point was located on the centre of the descending part of the trapezius muscle midway between the processus spinosus of the seventh cervical vertebra and the acromion. The muscle hardness was calculated as the mean of five consecutive measurements. All recordings were stored in the computer, and they were not analysed before the study was completed and the randomization code was broken.
Total tenderness
Tenderness of pericranial myofascial tissues was recorded according to the Total Tenderness Scoring system (1), which has previously proved to be reliable (19). Eight pairs of muscles and tendon insertions (masseter, temporal, frontal, sternocleidomastoid and trapezius muscles, coronoid and mastoid processes, and neck muscle insertions) were palpated. Tenderness was scored on a four-point (0–3) scale at each location (local tenderness score) and values from left and right sides were summed to a Total Tenderness Score (TTS) (maximum possible score=48).
Pressure pain thresholds
Pressure pain detection thresholds (PPDTs) were measured at the dorsum of the second finger (middle phalanx) and at a fixed point at the anterior part of the temporal muscle as previously described (20). Measurements were performed at the non-dominant side. The PPDT was defined as the pressure at which the sensation changed from pressure alone to a combination of pressure and pain. The subject indicated that the pain threshold was reached by pressing a hand-held button. The algometer display was thereby ‘frozen’ and the pressure was immediately released. Each threshold was calculated as the mean of five consecutive recordings performed with intervals of approximately 30 s.
Heat pain thresholds
The heat pain threshold was defined as the temperature at which the sensation changes from heat alone to a combination of heat and pain. When the pain threshold was reached the subjects activated a push-button. The temperature was then recorded and fell immediately to 32°C. The skin was stimulated in the palm of the non-dominant hand over the thenar eminence. Each threshold was calculated as the mean of five consecutive recordings performed with intervals of approximately 30 s.
Data analysis and statistics
Results are presented as medians with quartiles in parentheses. For each of the variables, the sum of the differences between the pretreatment value and each of the post-treatment values was calculated in order to obtain a summary measure of effect for each treatment (21). Since values were not normally distributed, the summary scores calculated for active treatment and placebo were compared by the Wilcoxon signed ranks test. All analyses were done with SPSS (version 8.0.0). Five percent was accepted as level of significance.
Results
All subjects completed the study.
Headache intensity
In both patients and controls, headache intensity over 12 h observation was significantly stronger on the GTN day than on the placebo day, as previously reported (11). In patients, the area under the headache curve (intensity × duration) on a GTN day was significantly higher than on a placebo day (P = 0.008) (Fig. 1). In patients, GTN infusion resulted in a biphasic response with immediate headache and more pronounced headache several hours later (delayed headache) (Fig. 1). The patients developed significantly stronger headache than controls both during the first hour and during the subsequent 11 h (P = 0.0001) (Fig. 1).

Median headache intensity over time during (20 min) and after infusion of glyceryl trinitrate (GTN) and placebo in 16 patients with chronic tension-type headache and in 16 healthy subjects. Headache was scored on a 10 point Verbal Rating Scale (VRS). In patients, the area under the headache curve (intensity × duration) on a GTN day was significantly higher than on a placebo day (P = 0.008). The headache intensity reached its peak value at 8 h after start of the GTN infusion. Patients developed stronger headache than controls (P = 0.0001). ▪, GTN, patients; •, GTN, controls; □, placebo, patients; Δ, placebo, controls. (Modified from Ashina et al., Brain 2000, by permission of Oxford University Press.)
Muscle hardness
There was no difference between the summary score of muscle hardness recorded during treatment with GTN and placebo in either patients (P = 0.80) or controls (P = 0.23).
Total Tenderness Score
The summary score of tenderness recorded on the GTN day did not differ from the summary score recorded on the placebo day in either patients (P = 0.71) or controls (P = 0.91) (Tables 2 and 3).
Muscle hardness, Total Tenderness Score, pressure pain detection thresholds (PPDT) in the temporal region and in the finger, and heat pain thresholds recorded before, 60 and 120 min after start of the infusion of glyceryl trinitrate (GTN) or placebo in 16 patients with chronic tension-type headache

Median values with quartiles in parentheses are given.
Muscle hardness, Total Tenderness Score and pressure pain detection thresholds (PPDT) in the temporal region and in the finger, and heat pain thresholds recorded before and 60 min and 120 min after start of the infusion of glyceryl trinitrate (GTN) or placebo in 16 healthy controls

Median values with quartiles in parentheses are given.
Pressure pain thresholds
In patients, there was no difference between the summary score of PPDTs recorded during treatment with GTN and placebo in the temporal region (P = 0.96) or in the finger (P = 0.96). In controls, there were also no changes in PPDT in either the temporal region (P = 0.33) or finger (P = 1.00) (Tables 2 and 3).
Heat pain thresholds
The summary score of the heat pain thresholds on the GTN day did not differ from the heat pain thresholds on the placebo day in either patients (P = 0.94) or controls (P = 0.08) (Tables 2 and 3).
Discussion
Previous studies of GTN-induced headache
Intravenous infusion of GTN induces headache during infusion in healthy subjects, migraineurs and in patients with CTTH (11, 22, 23). While this headache rapidly disappears after stopping of the GTN infusion in controls (11, 22, 23), studies in patients with migraine without aura and in patients with CTTH demonstrate that patients developed stronger immediate headache (during infusion and 1 h after infusion) than controls (11, 23). Furthermore, 90% of migraineurs (24) and 91% of CTTH sufferers (11), but only 10–30% of controls, developed a more pronounced delayed headache. Though sharing a similar time profile, the characteristics of the delayed headache in migraine and CTTH were different: 80% of migraine patients developed migraine without aura after infusion of GTN (24) while 87% of patients with CTTH developed a tension-type headache (11).
Myofascial factors and tension-type headache
Previous clinical studies have suggested that myofascial factors contribute to the development of CTTH. Both hardness of trapezius muscle and myofascial tenderness of the pericranial region are persistently increased in patients with CTTH (2, 4). Furthermore, myofascial tenderness was higher during compared with outside of an actual episode of CTTH (4). In the present study, it appears that muscle hardness was higher in healthy controls than in patients (see Tables 2 and 3). However, this difference was not statistically significant and the finding is probably accidental. The inter-individual variability is relatively high when measuring muscle hardness (2, 4), which increases the risk of spurious findings in studies including a relatively small number of subjects. It has to be emphasized that the present study was designed to examine possible changes in the psychophysical variables only within groups, i.e. the variables on GTN day vs. on placebo day. Mechanisms leading to increased muscle hardness and myofascial tenderness are largely unknown, but may include abnormalities in myofascial tissue such as ischaemia or the accumulation of chemical mediators (5) and sensitization of muscle nociceptors (25). Recent studies have suggested that sensitization of myofascial pain pathways at the trigeminal/spinal level may also be involved in the persistently increased hardness and tenderness in CTTH (8, 10).
Mechanisms of immediate headache
In the present study we found that muscle hardness, myofascial tenderness and pressure-pain detection thresholds in the cranial and extracranial regions are unchanged during NO-induced immediate headache. These results seem to rule out sensitization of myofascial peripheral and central pathways as the mechanism of the immediate headache. Unchanged pressure-pain detection thresholds in the temporal region may also indicate no alteration in sensitivity of extracranial vasculature. Experimental studies in humans suggest that NO may directly activate or sensitize nociceptors around blood vessels (26). Intravenous infusion of GTN (0.5 μg/kg per min over 20 min) induced dilatation of the middle cerebral artery which lasted until 1 h after stopping GTN infusion (27). It is therefore more likely that immediate headache after GTN infusion in patients with CTTH may originate from NO-induced activation or sensitization of sensory nerves around intracranial arteries or from NO-induced arterial dilatation, or both, and that alterations in sensitivity of myofascial peripheral and central pathways do not contribute to the immediate headache.
With regard to healthy subjects, the present findings largely confirm a study by Thomsen et al. (28). In this study, pressure pain detection and tolerance thresholds in the finger and temporal region were unaffected after infusion of GTN, except for slightly decreased pressure pain detection and tolerance thresholds in the temporal region at the highest doses (1.0 and 2.0 μg/kg per min).
Mechanisms of delayed headache
The mechanisms leading to the delayed headache are unknown. We found that in most patients with CTTH clinical characteristics of the delayed headache were similar to the characteristics of their usual tension-type headache (11). We suggested that excessive vascular nociception induced by GTN may gradually augment the pre-existing facilitation of myofascial central pain pathways in these patients (11). This convergence could explain why NO does not induce strong delayed headache in healthy subjects, where no pre-existing sensitization is present. It also suggests that, in patients, alterations of some of the measured variables such as myofascial tenderness might occur during the delayed headache, but this has not yet been studied.
In conclusion, our results indicate that neither sensitization of muscle nociceptors nor central sensitization of myofascial pain pathways play a role in GTN-induced immediate headache. We suggest that GTN-induced immediate headache may be explained by increased nociceptive input from the intracranial vasculature. Further prospective studies are required to investigate the mechanisms responsible for NO-induced delayed headache in patients with chronic tension-type headache.
Footnotes
Acknowledgements
We would like to thank Mrs Hanne Andresen for skilful technical assistance. The study was supported financially by the University of Copenhagen.
