Abstract
The aim of the present study was to investigate the impact of static contraction of the shoulder and neck muscles on muscle tenderness and headache in patients with tension-type headache. Twenty patients with frequent episodic tension-type headache and 20 healthy age- and sex-matched controls were examined using a placebo-controlled cross-over design. The subjects performed static contraction of the trapezius muscles (active procedure) or the anterior tibial muscles (placebo procedure) with 10% of maximal force for 30 min. Total tenderness score, local tenderness score and headache intensity were evaluated before and after the static work. Changes in headache intensity were followed for 24 h. Pericranial tenderness increased significantly more in patients than in controls after the active procedure (P = 0.04). The increase in pericranial tenderness tended to be higher after the active procedure than after the placebo procedure in patients (P = 0.08) and in controls (P = 0.07). Sixty per cent of the patients and 20% of the healthy controls developed headache after the active procedure. Fifty per cent of the patients and none of the controls developed headache after the placebo procedure. There was no significant difference in headache development between the active and the placebo procedure in patients or controls. These findings demonstrate that tension-type headache patients are more liable to develop shoulder and neck pain in response to static exercise than healthy controls.
Introduction
Tension-type headache is the most common type of headache with an overall lifetime prevalence of 78% (1). However, our knowledge of the pathophysiology of tension-type headache is limited. Previous studies have mainly focused on the impact of peripheral, i.e. muscular factors, but recent studies suggest that central factors may play an important role (2). Several studies have found significantly increased tenderness (3–5) and hardness (6, 7), of the pericranial muscles in tension-type headache patients as well as a qualitatively altered pain perception from these muscles (8). However it is unknown whether myofascial tenderness is the cause or the consequence of headache. Experimental studies have indicated that individuals exposed to monotonous work involving the shoulder and neck muscles for longer periods frequently develop tender muscles and tension-type headache (9–11). Experimental models provide a unique opportunity to control the causative factors and thereby to study initiating mechanisms of headache, but only few experimental models have focused on the relationship between exercise induced muscle tenderness and headache.
To study the impact of peripheral factors we aimed to develop a controlled method to induce muscle tenderness and headache by static isometric exercise of the trapezius muscle in patients with episodic tension-type headache and in healthy controls.
Methods
Subjects
Twenty patients with episodic tension-type headache, diagnosed according to the criteria of the International Headache Society (IHS) (12), were randomly recruited from the Danish Headache Centre at Glostrup Hospital. Patients aged between 18 and 65 years and with 8–14 headache days per month were included. The exclusion criteria were:
migraine more than 1 day per month;
other neurological, systemic or psychiatric disorders;
overuse of analgesics (>15 days/month);
daily medication except oral contraceptives;
pregnancy;
ongoing headache or intake of analgesics on examination day;
moderate or severe tenderness of the trapezius or the anterior tibial muscles (local tenderness score > 1 (on a scale from 0 to 3)) (13) on the 2 examination days.
None of the patients had recently received any kind of nonpharmacological treatment, e.g. physiotherapy or acupuncture. All patients fulfilled the new diagnostic criteria for frequent episodic tension-type headache (14). Twenty healthy age- and sex-matched subjects (<12 headache days per year) were used as controls. The study was approved by the local Ethics Committee and was conducted in accordance with the Helsinki Declaration. All subjects gave informed consent before participation in the study.
Apparatus
Two force transducers (Bisco Vaegte A/S, Denmark), measuring the force during voluntary contractions, were used to ensure a continuously static and symmetric workload on the muscles. Each transducer was connected to a display, which continuously showed the pressure applied. The display was placed visible to both the subject and the observer. The contact area of the transducer was 10 × 10 cm and it was adjustable in vertical and horizontal directions. The force transducers have previously been described in detail (15).
Local tenderness was measured with a palpometer, which allows control of the applied pressure during palpation. A pressure sensitive plastic film attached to the index finger records the pressure exerted in arbitrary units (13). A standardized pressure (160 units) was applied and the local tenderness was scored by the subjects on a 100 mm Visual Analogue Scale (VAS), where 0 mm was no pain and 100 mm was the worst imaginable pain.
Experimental design
The patients were asked to keep a diagnostic diary of headaches for a 4 week run-in period to ensure the fulfilling of the inclusion criteria. On the initial examination day patients were given a complete physical and neurological examination. The subjects were placed in a dental chair with headrest, adjustable in vertical direction. A standard point was defined on the trapezius muscle on the upper margin of the muscle midway between the processus spinosus of the seventh cervical vertebra and the acromion on the nondominant side. Another standard point was defined on the anterior tibial muscle a third of the distance between apex patellae and the lateral malleol on the nondominant side.
Pericranial tenderness
Pericranial tenderness was evaluated by the Total Tenderness Score system (TTS), which has previously proved to be reliable (13). Eight muscles and tendon insertions (frontal, temporal, masseter, sternocleid and trapezius muscles, posterior neck muscle insertions, mastoid and coronoid processes) were palpated and tenderness was scored in each location on a scale from 0 to 3, and scores from all sites were summed. The maximal possible score was 48. Subjects were asked to evaluate spontaneous pain in the trapezius muscle and in the anterior tibial muscle as well as headache on a 100 mm VAS.
Local tenderness
Local tenderness was measured on the two standard points, at the trapezius muscle and at the anterior tibial muscle with a palpometer as previously described (13).
Procedure
All measurements were performed by the same observer throughout the study (MBC). Subjects were randomized to start either with the active or the placebo procedure and the two procedures were separated by at least one week. The subjects were informed that the purpose of the study was to examine muscle function during exercise. Randomization was done by computer (Medstat, version 1.1). TTS, local tenderness and headache were recorded at baseline and at 0, 30, 60, 90 and 120 min after exercise. For the following 24 h, subjects completed a specific diary with evaluation of headache intensity and spontaneous pain in the shoulder and neck muscles and in the anterior muscle group of the lower leg. Evaluation was done every 4th hour on a VAS. Those subjects who developed headache completed a diary of headache characteristics.
The active procedure consisted of static isometric contraction of the trapezius muscle. Subjects were placed in the dental chair with arms and legs hanging. The height of the dental chair was adjusted so the force transducers rested on the acromion with a light symmetrical pressure on both shoulders. The subjects were then encouraged to elevate the shoulders with maximal force. The maximal voluntary isometric contraction force of the trapezius muscle was calculated as the average of four contractions. The subjects then performed an isometric contraction of the trapezius muscle with 10% of the maximal force for 30 min
The placebo procedure consisted of static isometric contraction of the anterior tibial muscle. Subjects were placed similar to the active procedure. The force transducer and the dental chair were adjusted so the transducer rested on the dorsal side of the feet with a light symmetrical pressure on both feet. No work was done with arms or shoulders. The subjects were encouraged to flex the ankles with maximal force. The maximal voluntary isometric contraction of the anterior tibial muscle was calculated as the average of four contractions. The subjects then performed a voluntary isometric contraction of the anterior tibial muscle with 10% of maximal force for 30 min. Visual inspection of the force transducer display ensured that the contraction force was within ± 200 g of the desired value for both procedures.
Statistical analysis
All data except for local tenderness were normally distributed. Normal distributed data are presented as mean values with standard error of mean. Local tenderness data are presented as medians with quartiles in parentheses. For each of the variables, the sum of the differences between baseline and each of the postexercise values was calculated in order to obtain a summary measure of the effect for each procedure (16). The primary end point was the difference in summary measures of Total Tenderness Score between patients and healthy controls during the active procedure. Normally distributed data were tested with students paired t-test (comparing differences between active and placebo procedures) and unpaired t-tests (comparing differences between patients and controls). Likewise, Wilcoxon Signed Ranks Test and Mann–Whitney test were used for data not normally distributed. The chi-square test was used to compare the observed headache frequency with the expected frequency and McNemar's test was used to compare headache frequencies after the active and the placebo procedure. Five per cent was accepted as level of significance. All data were analysed with SPSS® software, version 11.0 (SPSS Inc., USA).
Results
All included patients and healthy controls completed the study. Clinical characteristics are presented in Table 1. The maximal voluntary force at the active procedure was 24.6 ± 2.2 kg in patients, and 31.0 ± 3.6 kg in controls (P = 0.13). At the placebo procedure the maximal voluntary force was 11.6 ± 0.7 kg in patients and 12.2 ± 0.7 kg in controls (P = 0.53).
Clinical characteristics of headache patients and controls

Mean values are presented with range in parentheses. TTH indicates tension-type headache.
Total tenderness score
Total tenderness score at baseline was significant higher in patients than in controls (P < 0.001). After the active procedure, total tenderness increased significantly more in patients than in controls (P = 0.04), but there was no difference between patients and controls after the placebo procedure (P = 0.23) (Table 2). Total tenderness tended to be higher after the active procedure than after the placebo procedure in patients (P = 0.08) and in controls (P = 0.07) (Fig. 1).
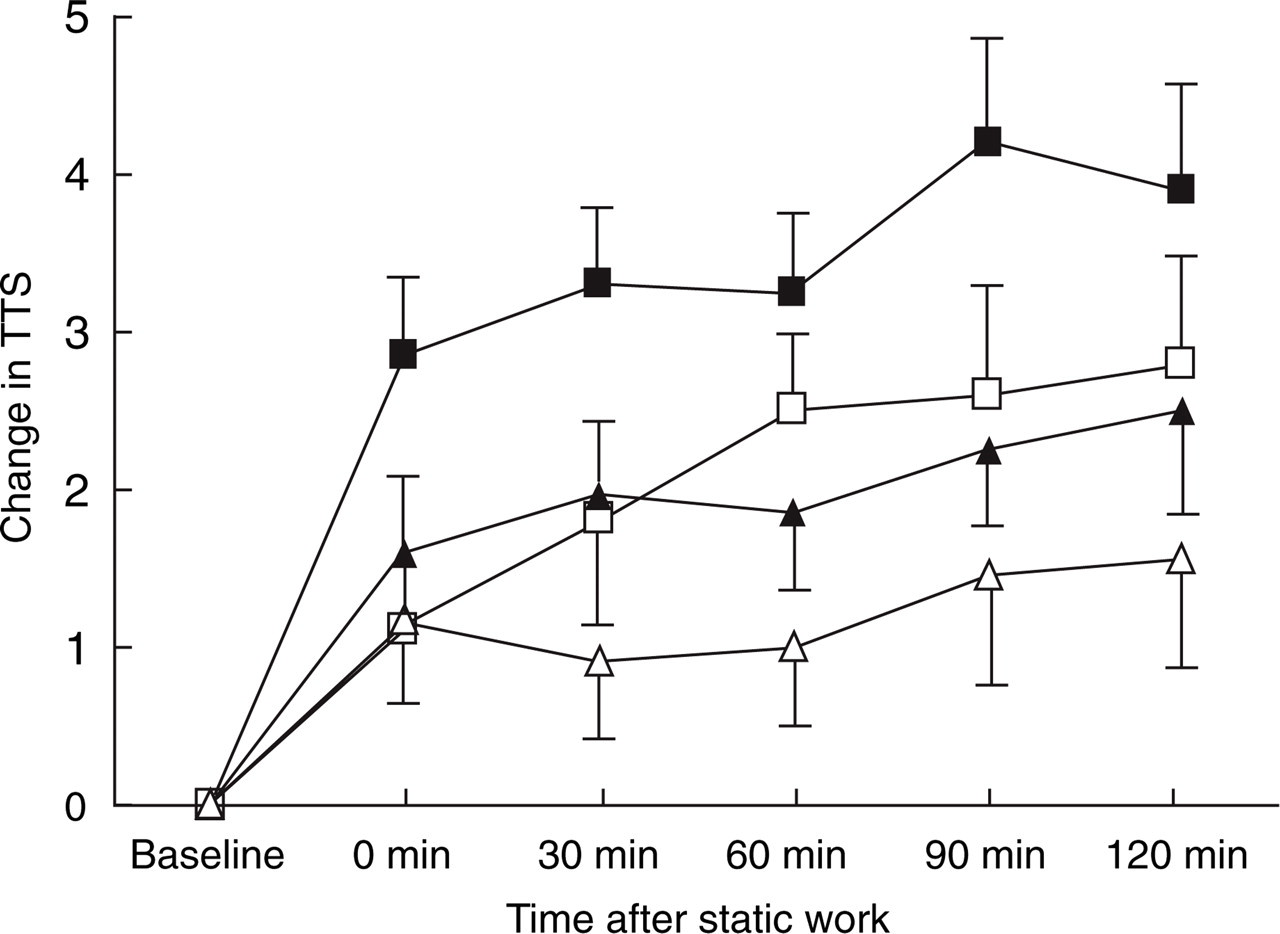
Change in Total Tenderness Score (TTS) in patients (▪,□) and controls (▴,▵) after the active (▪,▴) and after the placebo (□,▵) procedure. The summary score was significantly higher in patients than in controls after the active procedure (P = 0.04). The summary score tended to be higher after the active procedure than after the placebo procedure in both patients (P = 0.08) and controls (P = 0.07). Mean values with standard error of mean are presented.
Total tenderness score (TTS) and local tenderness score (LTS). Baseline values and the sum of differences between baseline and each of the postexercise values, after the active and the placebo procedure

Mean values with standard error of mean;
Median values with quartiles in parentheses.
Local tenderness
At the trapezius muscle local tenderness tended to increase more in patients than in controls after the active procedure, but the difference was not statistically significant (P = 0.11). There was no difference between patients and controls after the placebo procedure (P = 0.55). In patients and in controls local tenderness at the trapezius muscle did not increase more after the active procedure than after the placebo procedure (patients: P = 0.20; controls: P = 0.41). At the tibial muscle there were no differences in local tenderness (Table 2).
Headache
Sixty per cent (12/20) of the tension-type headache patients developed headache within 24 h after the active procedure. This is a significantly higher frequency than expected from their usual headache frequency (P = 0.02). Fifty per cent (10/20) developed headache after the placebo procedure, which was not significantly different from what was expected (P = 0.16). There was no difference in development of headache between the active and the placebo procedure in patients (P = 0.73). Twenty per cent (4/20) of the controls developed headache within 24 h after the active procedure and none (0/20) developed headache after the placebo procedure (P = 0.13) (Fig. 2). All headaches fulfilled the IHS criteria for tension-type headache. The median delay from exercise to development of headache in patients was 1.25 h (0.5–3.5) after the active procedure and 4.0 h (1.4–16.0) after the placebo procedure. There was no difference in intensity of headache in patients between the active and the placebo procedure (Fig. 3). For the few controls who developed headache after the active procedure, the median delay from exercise to development of headache was 2.5 h (0.25–4.0).

Headache development in tension-type headache patients and in controls after the active (▪) and the placebo (□) procedure. ∗∗P = 0.01, NS nonsignificant.

Mean headache intensity in patients after the active (n = 12; ▪) and the placebo (n = 10; □) procedure. There were no significant differences between active and placebo procedures. Mean values with standard error of mean are presented.
Discussion
Tenderness
The major finding of the present study was that pericranial tenderness increased significantly more in tension-type headache patients than in healthy controls in response to static exercise of the trapezius muscle. Previously, only few experimental models have focused on the relationship between exercise induced muscle tenderness and headache. Jensen et al. (17) studied the effect of 30 min of voluntary tooth clenching in 48 migraine patients. Only 8% of the patients developed a migraine attack, while 54% developed muscle-contraction-like headaches. Another study (18) found that sustained tooth clenching induced headache in 69% of tension-type headache patients and in only 17% of healthy controls. The headache was preceded by increased muscle tenderness, thereby supporting the muscle tension hypothesis as an initiating mechanism. However none of these studies were placebo-controlled.
The increased tenderness found in the present study could be caused by peripheral factors, e.g. increased peripheral sensitivity. This is supported by a recent model of myofascial pain where Mork et al. (19) found increased peripheral nociception in patients with frequent episodic tension-type headache. We recently demonstrated decreased muscle blood flow in response to static exercise in patients with chronic tension-type headache (15), but it is unclear whether this altered blood flow could affect peripheral nociception.
The increased tenderness could also be caused by central factors. Thus, it has been demonstrated that chronic tension-type headache patients are more susceptible to mechanical nociceptive inputs (2). Furthermore Bendtsen et al. (8) showed a qualitatively altered pain perception from tender muscles in chronic tension-type headache patients, and it was proposed that the altered pain perception was due to sensitization of the central nervous system. Central pain processing has on the other hand been reported to be normal in episodic tension-type headache patients (2). However, our study group of patients differed clinically from previous studies of episodic tension-type headache, since we examined the most severely affected episodic tension-type headache patients with a high frequency of headache. It is therefore possible that the central nervous system is hyper excitable in these patients and that the increased tenderness could be caused by central sensitization.
In the present study we found a tendency towards a larger increase in tenderness after the active than after the placebo procedure, but the difference did not reach statistical significance. It is possible that a higher workload or a longer period of static contraction of the trapezius muscle with a lower workload could have induced more tenderness after the active procedure than after the placebo procedure. At the placebo procedure no work was done with the upper extremities. Nevertheless both patients and controls developed increased pericranial tenderness after the placebo procedure. This could be explained by stressful conditions in the lab. In support for this, a previous study has demonstrated increased muscle activity in the trapezius muscle in healthy subjects when exposed to mental stress (20).
Headache
In the present study 60% of episodic tension-type headache patients and 20% of healthy controls developed a tension-type like headache within 24 h after the active procedure. This is significantly more than their expected headache frequency. However, there was no difference in headache development between the active and the placebo procedure. This finding suggests that the headache development was not solely caused by muscular factors. This is supported by Neufeld et al. (21) who studied the effect of tooth clenching in tension-type headache patients in a placebo-controlled study. Patients were more likely to develop headache than healthy controls after both the tooth clenching and the placebo procedure, but there was no significant difference in headache development between the tooth clenching and the placebo procedure. It was suggested that the development of headache following noxious stimuli is more strongly related to headache proneness and associated abnormalities in central pain procession than muscle strain induced by tooth clenching.
It could be argued that the subjects in the present study were exposed to mental stress during the procedures, where they were told to keep their eyes on the display and make sure that the contraction force was within narrow limits. Several studies have indicated a causal relationship between mental stress and headache (22–24) and patients with tension-type headache may be more susceptible to mental stress than healthy individuals. However, the present study was not designed to differentiate between muscular and psychological factors. The possible role of mental stress and psychological factors associated with headache should be addressed in future studies.
Methodological considerations
The fact that 50% of tension-type headache patients developed headache after the placebo procedure demonstrates the importance of a placebo-controlled study design. Due to this very high placebo response the present model is not a valid headache model in patients as some of the possible causative factors could not be controlled. On the other hand the model is a simple human model of shoulder and neck pain and further modifications with increased weight load and longer exposure can be recommended. Post exercise muscle tenderness usually increases in intensity up to 24 h after exercise (25). Recordings of tenderness on the following day might therefore demonstrate a further increase in tenderness. The current study group did not include enough males to allow a statistical comparison of tenderness or headache between male and female headache patients and controls. However sex differences in muscle tenderness has previously been reported (26) and it has been hypothesized that there may be sex differences in pain mechanisms. This issue should be addressed in future studies.
Conclusion
In conclusion, pericranial tenderness increased significantly more after static exercise in tension-type headache patients than in controls. This indicates that tension-type headache patients are more liable to develop shoulder and neck pain in response to static exercise than healthy controls. A human model of mechanical stress has been developed and future studies should investigate whether the increased tenderness is caused by peripheral or central mechanisms.
Footnotes
Acknowledgements
The study was supported by the Foundation of Ludvig and Sara Elsass.
