Abstract
Cilostazol, an inhibitor of phosphodiesterase (PDE) type 3, is used clinically in peripheral artery disease. PDE3 inhibitors may be clinically useful in the treatment of delayed cerebral vasospasm after subarachnoid hemorrhage. The authors present the first results on the effect of cilostazol on cerebral hemodynamics in normal participants. In this double-blind, randomized, crossover study, 200 mg cilostazol or placebo was administered orally to 12 healthy participants. Cerebral blood flow was measured using 133Xe inhalation and single photon emission computerized tomography. Mean flow velocity in the middle cerebral arteries (VMCA) was measured with transcranial Doppler, and the superficial temporal and radial arteries diameters were measured with ultrasonography. During the 4-hour observation period, there was no effect on systolic blood pressure (P = 0.28), but diastolic blood pressure decreased slightly compared with placebo (P = 0.04). VMCA decreased 21.5 ± 5.7% after cilostazol and 5.5 ± 12.2% after placebo (P = 0.02, vs. placebo), without any change in global or regional cerebral blood flow. The superficial temporal artery diameter increased 17.6 ± 12.3% (P < 0.001 vs. baseline) and radial artery diameter increased 12.6 ± 8.6% (P < 0.001 vs. baseline). Adverse events, especially headache, were common. The findings suggest that cilostazol is an interesting candidate for future clinical trials of delayed cerebral vasospasm.
Cilostazol (Pletal) is a vasodilator and antiplatelet drug used in the treatment of intermittent claudication (Sorkin and Markham, 1999). Cilostazol may also play a future role in the secondary prevention of ischemic stroke because it significantly decreases the risk of stroke recurrence in stroke patients (Gotoh et al., 2000) and has both neuroprotective and antithrombotic effects (Choi et al., 2002; Kohda et al., 1999).
Cilostazol is a dual inhibitor of phosphodiesterase type 3 (PDE3) and cellular adenosine reuptake (Liu et al., 2001). The cyclic nucleotide PDE family consists of enzymes responsible for the hydrolysis of the 3′ phosphoester bond of the cyclic nucleotide second messengers, cyclic AMP and cyclic GMP. By terminating the messenger signal, PDEs play a pivotal role in the regulation of cyclic nucleotide signaling, and the multiple subtypes and varied tissue distribution make PDEs promising drug targets for a variety of diseases (for review see Beavo, 1995). PDE3 inhibitors may have potential in the treatment of delayed cerebral vasospasm (DCV) (Arakawa et al., 2001; Khajavi et al., 1997). DCV is the major cause of cerebral ischemia after subarachnoid hemorrhage and has a high impact on mortality and morbidity (Zhang et al., 1998). The condition is potentially treatable, but results have been somewhat disappointing to date because the vasodilators tested have limited efficacy or cause hypotension that further compromises cerebral perfusion.
Cilostazol is a marketed PDE3 inhibitor with a favorable safety profile (Pratt, 2001). Cilostazol dilates cerebral arteries in vitro (Birk et al., 2004a; Shiraishi et al., 1998), and the effect is not dependent on a functional vascular endothelium or nitric oxide tone (Birk et al., 2004a). This is important, because dysfunction of the endothelium or of the nitric oxide–cyclic GMP pathway has been proposed to participate in the pathogenesis of DCV (Sobey, 2001). Based on these animal studies, we hypothesized that cilostazol may be a cerebrovascular dilator, but because of blood–brain barrier permeability, species differences and other factors, it is not always possible to extrapolate. We therefore decided to investigate the effect of cilostazol on the large cerebral arteries with corresponding measurements of regional cerebral blood flow (rCBF) and blood flow velocity of the middle cerebral artery (MCA) in normal participants.
MATERIALS AND METHODS
Design and participants
The study was designed as a double-blind, randomized crossover trial in which opaque gelatin capsules containing placebo (lactose, potato starch, magnesium stearate, and talc) or containing two 100-mg cilostazol tablets were administered by mouth on two study days separated by at least 1 week. The participants were randomized to receive active treatment on the first or second study day in a balanced fashion using a computer program (R v. 1.8.1., R Foundation for Statistical Programming).
Sixteen healthy participants were included in the study after a full medical history was collected and a physical examination performed. The participants had no neurologic or cardiovascular disorders, including migraine, and had no daily intake of any medication except oral contraceptives. Four participants did not complete the study: one withdrew after experiencing severe migrainelike headache after receiving cilostazol on day 1, and another due to circumstances unrelated to the study. One participant was excluded because of vomiting a few minutes after the intake of placebo, and another because of a potentially severe adverse event after cilostazol causing junctional escape nodal beats on the ECG with no clinical manifestations. Twelve participants (six men and six women) aged 26 ± 2.0 years and weighing 54 to 100 kg (mean 72.75 ± 14.8 kg) completed the study. All participants gave their informed consent before inclusion. The study was approved by the ethical committee of Copenhagen County and the Danish Medicines Agency, and was conducted according to the Helsinki II declaration.
Experimental protocol
The participants arrived at the clinic at 8.00
Cerebral blood flow velocity
Blood flow velocity in the middle cerebral artery (VMCA) was recorded bilaterally using hand-held 2MHz probes and transcranial Doppler (TCD; Multidop X, DWL, Sipplingen, Germany). A time-averaged mean over four cycles each comprising approximately 4 seconds or approximately 4 cardiac cycles was used as final measure for each time point. Identification of the MCA and marking reproducible fix points was done as previously described (Kruuse et al., 2000; Thomsen and Iversen, 1993). The same experienced investigator performed all insonations and the participants served as their own controls (Thomsen and Iversen, 1993). End-tidal PCO2 (PETCO2) was recorded simultaneously with the TCD measurements using an open mask that caused no respiratory resistance (Datex Normocap 200, Roedovre, Denmark). VMCA measurements at T0, T120, and T240 were performed immediately after CBF measurements to obtain corresponding values. The participants remained in exactly the same supine position during SPECT and TCD measurements. Because rCBFMCA equals the product of VMCA and cross-sectional area of the MCA, changes in cross-sectional area were calculated as ΔA = (rCBFMCA(2) ϗ VMCA(2)−1) ϗ (VMCA(1) ϗ rCBFMCA(1)−1)−1 (Dahl et al., 1989).
Diameter of peripheral arteries
The diameter of the frontal branch of the right temporal artery and the left radial artery (point of maximal pulsation at the distal volar wrist crease) was measured using a high-resolution ultrasound scanner 20 MHz, bandwidth 15 MHz (Dermascan C, Hadsund, Denmark)(Nielsen et al., 1990). Marks were drawn on the skin and the coordinates of these recorded to ensure reproducibility of the measuring points from day to day. Because of technical problems, data from the temporal artery could only be obtained from 11 participants. All measurements were done by the same technician.
Systemic hemodynamics
Heart rate and blood pressure (BP) was recorded using an autoinflatable cuff (Omega 1400, Orlando, FL, U.S.A.). ECG was monitored on an LCD screen and recorded on paper at selected time points (Cardiofax V, Nihon-Cohden, Japan).
Cerebral blood flow measurement
The examination was performed with the subject in the supine position, in quiet surroundings with eyes closed and ears unplugged. Four markers were drawn on the skin to ensure accurate repositioning in each acquisition. PETCO2 was measured during each examination (Datex Normocap 200, Roedovre, Denmark). CBF was measured with 133Xe inhalation (Ceretronix XAS SM 32C, Randers, Denmark) and SPECT, using a brain-dedicated gamma camera (Ceraspect, DSI, Waltham, MA, U.S.A.). The system uses a stationary annular NaI crystal and a fast-rotating collimator. A dynamic protocol of 133Xe inhalation using the Kanno-Lassen algorithm was used (Kanno and Lassen, 1979; Kruuse et al., 2000). Calculation of flow in the perfusion territories of the major cerebral arteries was performed by fitting standard vascular regions of interest. Regional blood flow in the territory of the MCA (rCBFMCA) was calculated as mean of the right and left side, because there was no difference between the sides.
Statistical methods
All data are presented as means and standard deviations, unless otherwise stated. Baseline was calculated as the mean of the measurements of time points T–20 and T–10. Coefficient of variation (CV) was calculated as the standard deviation of difference between measurements divided by group means and reported in percentage (Thomsen and Iversen, 1993).
Effects over time for each variable (in absolute values) on each treatment day were analyzed using univariate analyses of variance with the factors time and participants. To avoid mass significance, the following time points were selected for analysis: baseline, 120 minutes, and 240 minutes. If an overall effect of time was found, significant changes were localized with Dunnett test. Differences between cilostazol and placebo were analyzed using the area under the curve (normalized against baseline) as summary measure with paired two-tailed t-tests. All analyses were performed with SPSS for Windows 11.5 (SPSS Inc., Chicago, IL, U.S.A.), and P < 0.05 was considered significant.
RESULTS
The superficial temporal artery diameter was significantly smaller on cilostazol days (P = 0.03). There were no other significant differences in baseline values between cilostazol and placebo days (Table 1).
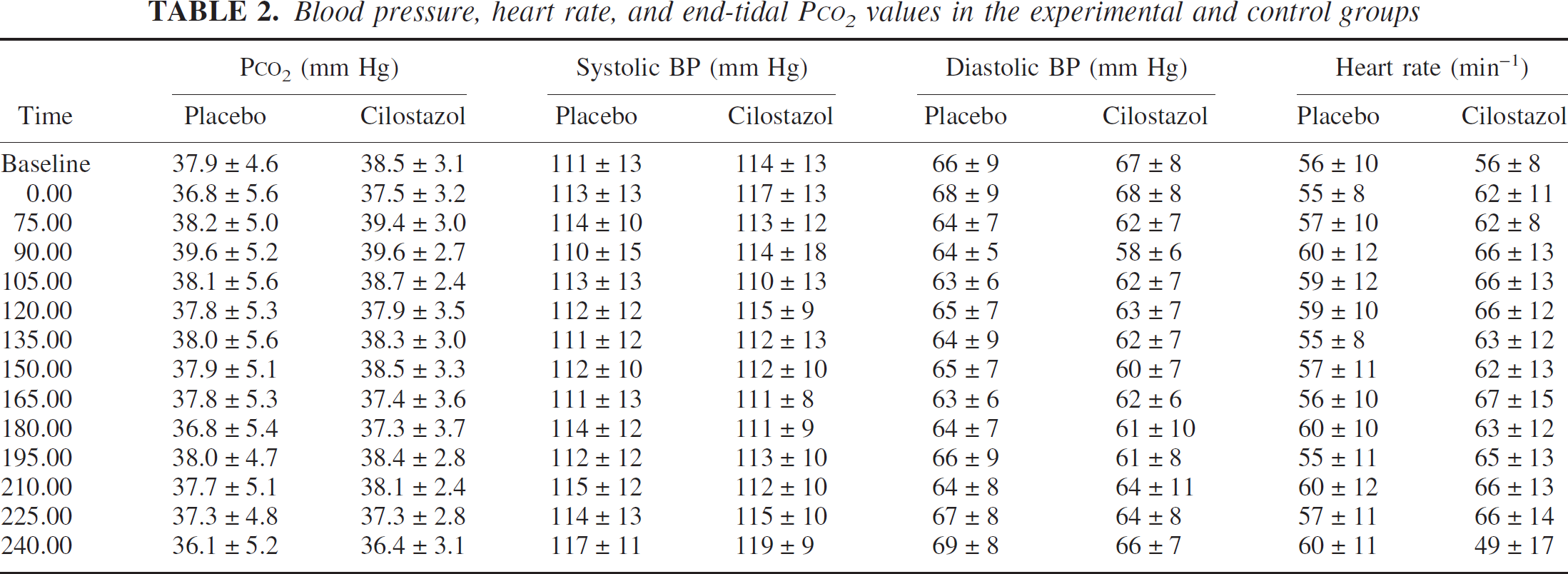
Compared with placebo, cilostazol did not change systolic BP (P = 0.23) or mean BP (P = 0.28), but a minor decrease was seen in diastolic BP (P = 0.03) with a 10.1 ± 5.5% maximum decrease at T150 (Table 2). Heart rate increased significantly after cilostazol administration (P = 0.01); peak increase was 22.4 ± 21.7% at T165.
There were no changes after cilostazol for global CBF (P = 0.77) or for rCBFMCA (P = 0.82) when compared with placebo Fig. 1. Day-to-day CV of baseline was 9.2% for global CBF. PETCO2 measured during CBF acquisition decreased significantly over time after both cilostazol (P = 0.01) and placebo (P = 0.001) administration. The change from baseline was less than 5% in both treatments, and there was no difference between treatments (P = 0.25). rCBF was consequently not corrected for PETCO2.
VMCA decreased over time after cilostazol treatment (P < 0.001), but not after placebo (P = 0.18), and with a significant difference between treatments (P = 0.02). The decrease in velocity after cilostazol apparently did not reach a plateau within the observation period (Fig. 2). At the last time point measured (T240), the decrease in VMCA was 21.5 ± 5.7% after cilostazol treatment and 5.5 ± 12.2% after placebo treatment compared with baseline. The mean difference in response between placebo and cilostazol at T240 was 18.0% (10.6% to 25.4%, 95% CI). The calculated change in cross-sectional area at T240 was 35.6 ± 31.4% after cilostazol treatment and 5.2 ± 13.5% after placebo treatment compared with baseline. Mean difference from placebo was 30.4% (11.4% to 49.3%, 95% CI). The change in cross-sectional area represents a change in MCA diameter of 15.6 ± 14.5% after cilostazol treatment and 2.3 ± 6.6% after placebo treatment. PETCO2 measured during TCD scans changed significantly over time after placebo (P = 0.005) and after cilostazol (P = 0.002) treatments. The mean decrease compared with baseline was 4.9 ± 4.8% after placebo and 5.3 ± 4.3% after cilostazol treatment at T240. There was no difference between treatments (P = 0.94), so we did not correct VMCA for PETCO2.
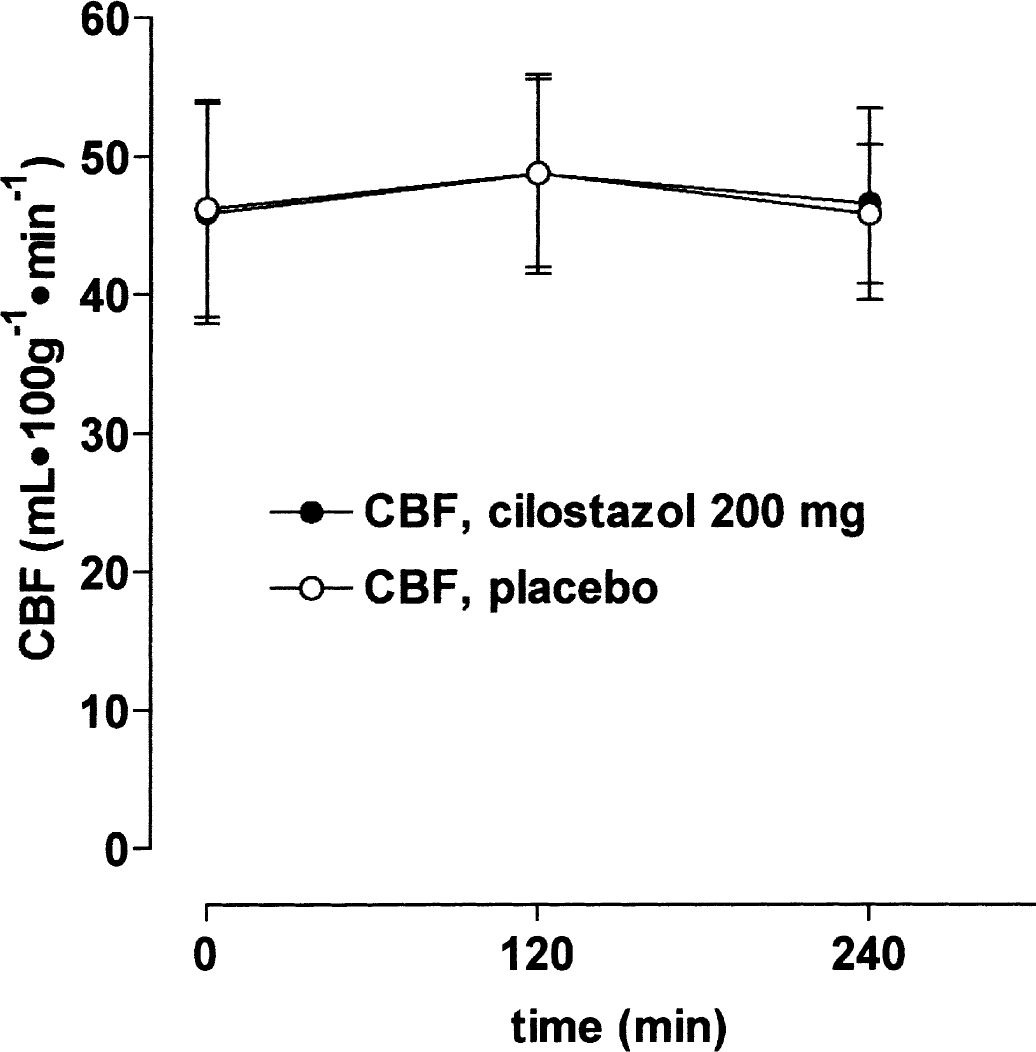
Mean global cerebral blood flow (CBF) (± SD) measured with 133Xe inhalation and single photon emission computerized tomography. There were no changes over time after 200 mg cilostazol ● or placebo ○ treatment or between treatments.
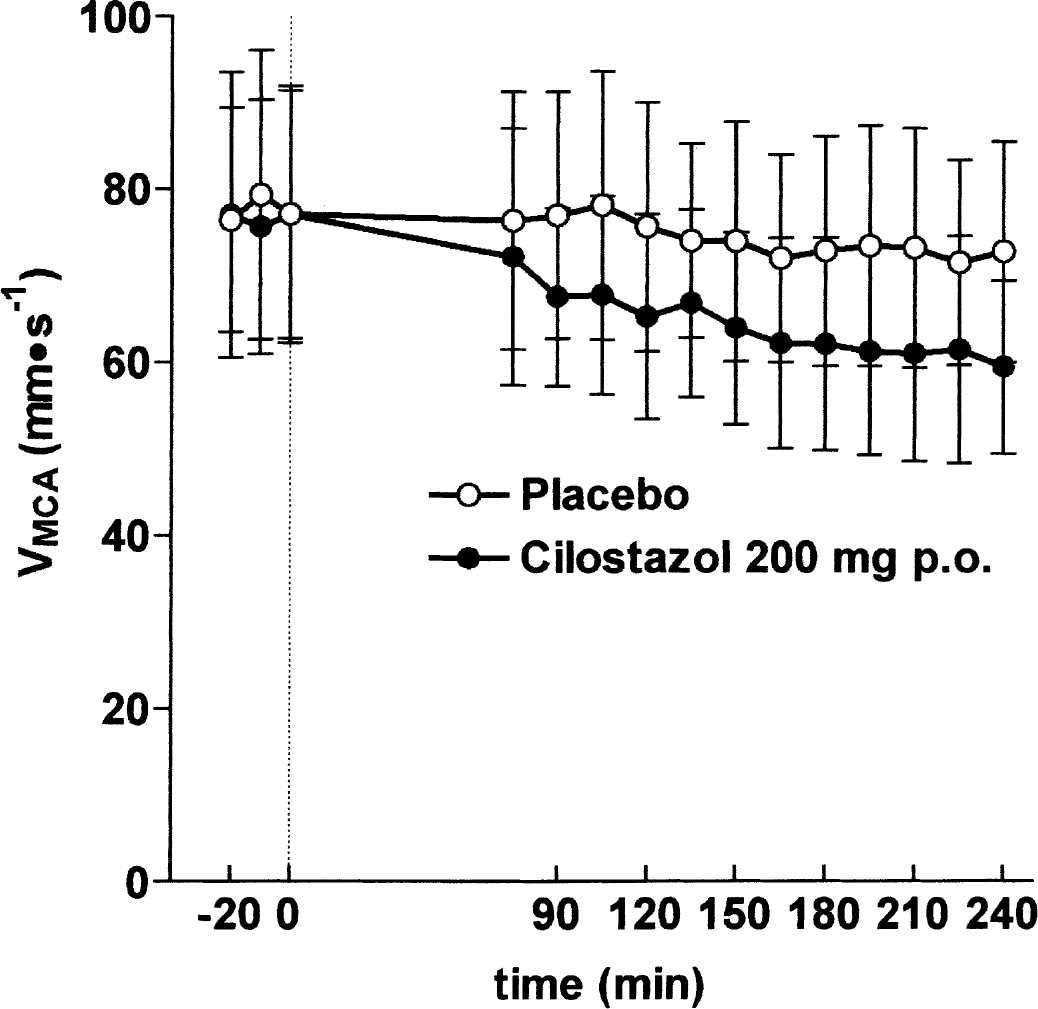
Mean flow velocities in the middle cerebral arteries (± SD) determined with transcranial Doppler ultrasonography after 200 mg cilostazol ● or placebo ○ treatment. Cilostazol significantly reduced flow velocity compared with placebo.
Baseline mean values and standard deviations in the experimental and control groups
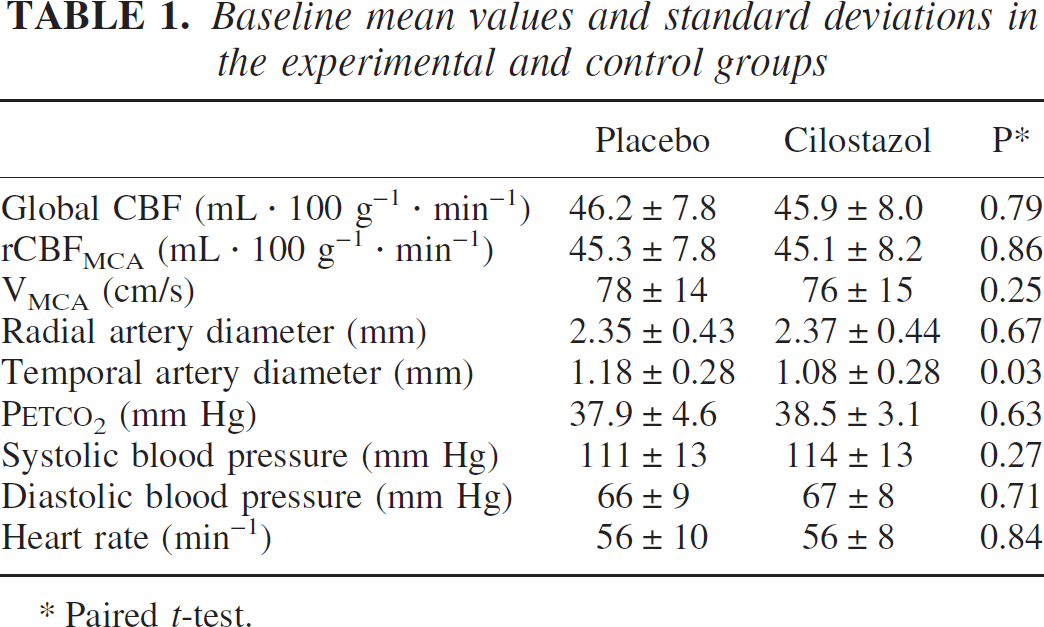
Paired t-test.
Temporal artery diameter increased significantly compared with placebo (P < 0.001). At T240 the change was 17.6 ± 12.3% compared with baseline. At the same time point, mean difference from placebo was 8.3% (–0.2% to 16.7%, 95% CI), which was not significantly different (P = 0.055, one-sample t-test). A considerable overlap was seen between the graphs for placebo and cilostazol (Fig. 3), and the difference was only significant because baseline was different between the two study days, which makes the interpretation of the results difficult. The radial artery diameter similarly increased after cilostazol compared to placebo (P = 0.04); the change compared with baseline was 12.6 ± 8.6% at T240 (Fig. 3). Mean difference from placebo at T240 was 12.1% (1.8% to 22.4%, 95% CI).
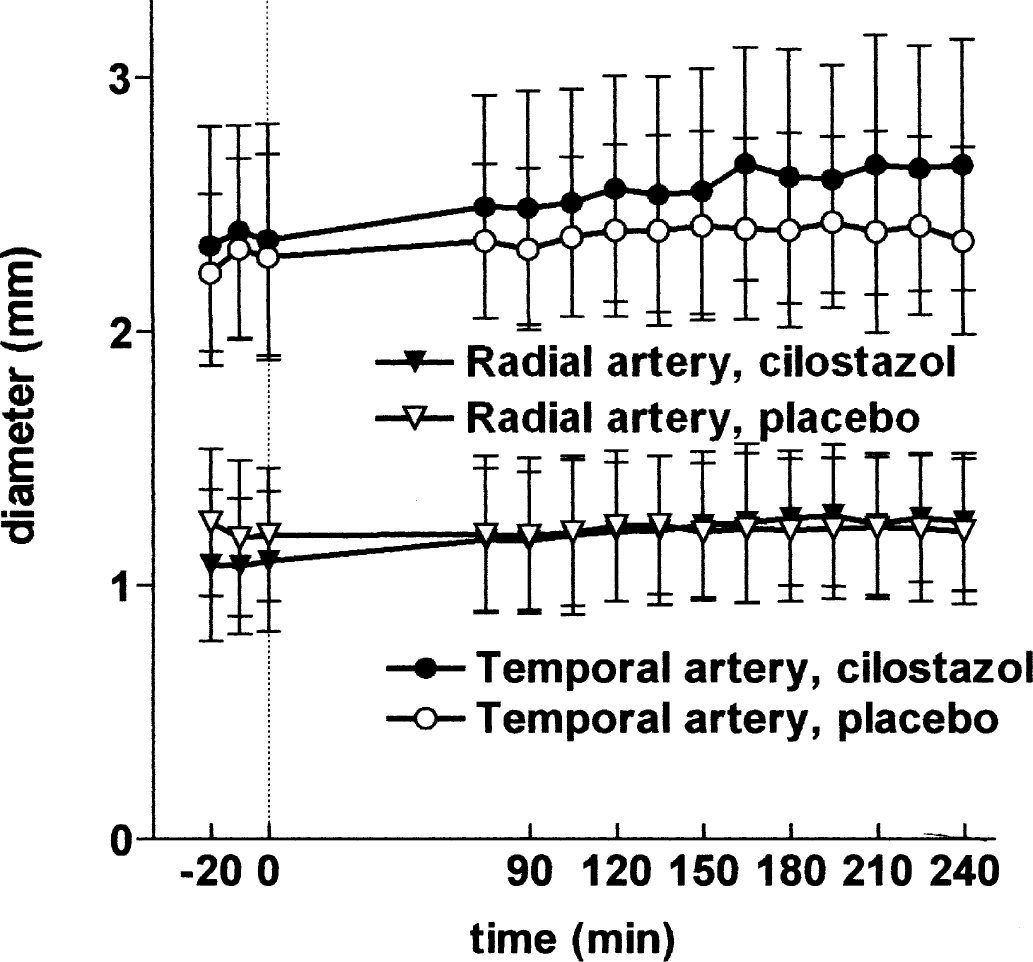
Mean diameter (± SD) of the radial (●, ○) and temporal (θ, ○) artery after 200 mg cilostazol (filled symbols) or placebo (open symbols) treatment, measured with high-resolution ultrasonography. Cilostazol significantly dilated both arteries.
Facial flushing, seen in 8 of 12 subjects (P = 0.03), and headache, seen in 7 of 12 subjects (P = 0.02), was more frequent after cilostazol treatment than after placebo treatment (2 of 12 and 1 of 12 subjects, respectively). Headache data will be published separately.
DISCUSSION
We have developed the combination of methods applied in the present study to provide an extensive and precise description of pharmacologic effects on cephalic hemodynamics. For CBF measurement, positron emission tomography (PET) and magnetic resonance techniques provide better spatial resolution than 133Xe SPECT, but this is of minor importance in pharmacologic studies because no major variation has been described in the regional response of cortical cerebral blood flow to drugs (Edvinsson and Krause, 2001). The day-to-day CV on CBF measurement was 9.2% in the present study, which is similar to that found in previous studies that have used the same method (Vorstrup, 1988). Similar reproducibility on CBF measurements is found using PET (Carroll et al., 2002), whereas both PET and SPECT are clearly superior in terms of day-to-day reproducibility to magnetic resonance (Carroll et al., 2002).
Blood pressure, heart rate, and end-tidal PCO2 values in the experimental and control groups
Transcranial Doppler measurements of velocity (Vmean) as stand-alone are difficult to interpret because velocity is dependent on both the diameter of the MCA and rCBFMCA, but they are useful in combination with quantitative rCBF. TCD measurements are most precise in the MCA with a CV of 26% between subjects, 20% between sides, 16% between days, and 7% 5 minutes apart (Thomsen and Iversen, 1993). To reduce the impact of intersubject and interobserver variability of TCD measurements, the present study was designed as a crossover study and the same trained investigator performed all measurements. In the present study, the day-to-day CV of VMCA at baseline was 6.7%, suggesting relatively high reproducibility. An alternative to these measurements could be magnetic resonance angiography, but this is difficult to time with CBF measurement, and its precision is not yet sufficiently documented and may not be high enough to detect modest pharmacologic effects. High-frequency ultrasound determination of the diameter of the superficial temporal artery and radial artery is highly accurate and reproducible as documented in many previous studies (Iversen et al., 1992; Iversen et al., 1990).
Pharmacology of cilostazol
Cilostazol is a dual inhibitor of PDE3 and cellular adenosine reuptake (Liu et al., 2001). Furthermore, in high concentrations (10 μmol/L), cilostazol inhibits recombinant human PDE5, one of the major cyclic GMP hydrolyzing enzymes (Sudo et al., 2000), and correspondingly increases cyclic CMP in a cyclic nucleotide assay (Birk et al., 2004a). However, PDE5 inhibition does not appear to play any significant role in the functional relaxant response to cilostazol (Birk et al., 2004a), and the highly potent and selective PDE5 inhibitor sildenafil does not dilate the MCA or increase CBF in a similar experimental setup as the present (Kruuse et al., 2003; Kruuse et al., 2002). Inhibition of adenosine reuptake may likewise participate in the relaxant response to cilostazol, because adenosine is a dilator of human cerebral arteries in vitro (Hardebo and Edvinsson, 1979). However, the infusion of adenosine in doses up to 120 μg · kg−1 · min−1, which dilated the superficial temporal artery and caused marked systemic effects, did not dilate the MCA significantly (Birk, 2004b). We therefore suggest that among the known pharmacodynamic effects of cilostazol, PDE3 inhibition is the most likely candidate to mediate any cerebrovascular effect.
Systemic hemodynamic effects
Cilostazol dilated all arteries examined to a similar degree, which suggests a general vasodilating effect on peripheral arteries. Similar changes have been seen in the dorsal pedal artery (Okuda et al., 1992). The arterial dilatation did not cause significant hemodynamic changes apart from a slight decrease in diastolic BP. Heart rate increased significantly, which could be a compensatory mechanism. However, cilostazol, like other PDE3 inhibitors, is believed to have a direct positive chronotropic effect on the heart (Schror, 2002).
Effect of PDE3 inhibition on large cerebral arteries
PDE3 is expressed in human aortic vascular smooth muscle cells (Palmer and Maurice, 2000), but PDE3 expression has not been demonstrated directly in cerebral arteries in any species. However, functional studies suggest a role. Cilostazol dilated rabbit MCA in vitro by a cyclic AMP-mediated mechanism (Shiraishi et al., 1998), and the PDE3 inhibitor siguazodan dilated canine basilar arteries, although with low efficacy (Willette et al., 1997). Using TCD, the PDE3 inhibitor milrinone showed a 20% increase in VMCA in patients during general anesthesia after a coronary bypass operation (Sulek et al., 2002). This observation would suggest either MCA constriction or an increase in CBF (Dahl et al., 1989). The study had no control group, and mean baseline values were less than 40 mm · s−1, which is lower than reference values (Thomsen and Iversen, 1993). The most likely explanation is that the increase in VMCA reflected a spontaneous increase in CBF after recovering from the surgical procedure. Using angiography, milrinone has been shown to prevent or even reverse the development of vasospasm in a canine subarachnoid hemorrhage model (Khajavi et al., 1997). This was also shown in a preliminary open study of seven patients treated with selective intraarterial infusion of milrinone into the lumen of the affected artery (Arakawa et al., 2001). The present results strongly support that PDE3 plays a role in the regulation of the diameter of the large cerebral arteries. We found an approximately 30% increase in MCA cross-sectional area, which is at least equivalent to the responses to the most potent arterial dilators that have previously been studied in man: glyceryl trinitrate, histamine, calcitonin gene–related peptide (Iversen et al., 1989; Lassen et al., 1998; Lassen et al., 1995). The decrease in VMCA is not likely explained by the slight 10% decrease in diastolic BP, because of CBF autoregulation (Aaslid et al., 1989).
Effect on CBF
In the present study, there was no effect of cilostazol on rCBF within the 4-hour observation period. Because maximum effect was apparently not obtained during this period for some parameters, it cannot be excluded that rCBF increase at a later time point. However, there is no trend in the results (Fig. 1) suggesting this is the case. The mean difference in response between cilostazol and placebo at T240 was 4% (–9.6% to 17.4%, 95% CI). This suggests that PDE3 plays a differential role in cerebral arteries and arterioles.
Previous studies (Kobayashi et al., 1985; Oishi et al., 2000; Yu et al., 2000) on the effect of PDE3 inhibition have reached conflicting results. An open 133Xe SPECT study included 14 patients 2 months to 2.5 years after cerebral infarction. Mean increase in global CBF was 11.9 ± 20.3%, P < 0.05, after 2 weeks of treatment with cilostazol 200 mg/d. There was an open control group, but these patients had mixed neurologic diagnoses. In another unblinded study, 20 patients in the chronic stage of cerebral infarction were switched from 200-mg/d ticlopidine to 200-mg/d cilostazol. Using 99mTc-ECD SPECT, it was suggested that rCBF increased 7% to 12% during cilostazol as compared with ticlopidine treatment in some, but not all, regions of interest (Oishi et al., 2000). However, this method cannot be used to study global CBF changes, only changes in the regional distribution of CBF. The effects of cilostazol in both studies were measured 2 weeks or 3 months, respectively, after obtaining control values. The increase in CBF could therefore reflect a period effect (e.g., delayed recovery after the stroke). Another possible explanation for the CBF increase is that the effect of cilostazol on CBF increased with repeated dosing. The plasma concentrations after the present dose equal plasma concentrations during the standard dosage regimen of 100 mg twice daily (Bramer et al., 1999). Olprinone, another PDE3 inhibitor, increased global CBF 4% to 16% in 15 patients aged 67 ± 8 years as measured by 99mTc-ECD SPECT (Yu et al., 2000). Apart from using a tracer unsuitable for obtaining absolute rCBF values, this study examined a compound about which little is known regarding PDE3 selectivity, and PETCO2 was not measured. In summary, the previous studies indicate a minor effect of PDE3 inhibitors on CBF, but study design and methods were not optimal.
Vasodilators are obvious candidates for the treatment of DCV, but may also be harmful, e.g., by counteracting the hypertension that is believed to be beneficial in DCV (Dorsch, 2002). If it dilates cerebral arterioles and thereby increases CBF, this will result in steal phenomena, i.e., the shunting of perfusion to normal areas of the brain. Because cilostazol dilates the MCA without adversely affecting CBF and blood pressure, we suggest that this compound is an interesting candidate for future clinical DCV trials. Adverse events, especially headache, were common and caused four participants to discontinue the study. Used for the treatment of DCV, this may be a lesser problem, considering the severity of the disorder. Furthermore, used in intermittent claudication, headache only caused discontinuation of treatment in 3.7% of the patients on the 100-mg twice-daily dosage compared with 0.3% on placebo. However, it remains to be resolved if the tolerability of cilostazol is different in subarachnoid hemorrhage patients and whether it can be safely used during DCV considering the antiplatelet actions of the drug. Whether the dilatation caused by cilostazol is sufficient to treat DCV, whether cilostazol is also effective in an older age group than studied presently, and whether DCV itself influences the efficacy of cilostazol (Sobey and Faraci, 1998) will have to be tested in future clinical trials.
Footnotes
Acknowledgements
The authors thank Lene Elkjaer and Kirsten Brunsgaard for their excellent technical assistance.
