Abstract
Because heat shock proteins (HSPs) have an important protective function against ischemia/anoxia in mammalian brain, the authors investigated the expression of Hsp72 and Hsc73 in the anoxia-surviving turtle brain. Unlike the mammalian brain, high levels of Hsp72 were found in the normoxic turtle brain. Hsp72 levels were significantly increased by 4 hours of anoxia, remained constant until 8 hours, and then decreased to baseline at 12 hours. By contrast, Hsc73 was progressively increased throughout 12 hours of anoxia. This differential expression suggests different protective roles: Hsp72 in the initial downregulatory transition phase, and Hsc73 in maintaining neural network integrity during the long-term hypometabolic phase.
Of the various heat shock protein (HSP) families, those associated with Hsp70 are particularly important in providing protection from stress-related damage (Snoeckx et al., 2001). The nomenclature of the Hsp70 family is somewhat complex (Snoeckx et al., 2001). The constitutive member of the HSP70 family is referred to as Hsc70, Hsp73, or Hsc73, depending on the particular study. Hsc73 is the most abundant HSP found in normal cells and is regarded as a constitutive protein because it is present in non-stressed tissues and is only slightly inducible by stress (Snoeckx et al., 2001). By contrast, Hsp72 (also called Hsp70), the stress-inducible HSP70 family member, is hardly seen in unstressed conditions (Snoeckx et al., 2001) but is highly induced by stresses including ischemia and hypoxia (Murphy et al., 1999, Snoeckx et al., 2001). However, the protection provided by HSPs in anoxia/ischemia in the brain is clearly limited because the mammalian brain does not survive many minutes of oxygen deprivation.
In contrast to the high vulnerability of the mammalian brain to hypoxia, the freshwater turtle (Trachemys scripta) brain can withstand anoxia for days at room temperature (Bickler and Donohoe, 2002; Lutz et al., 2003a). Initially (hours), important energy savings come from an extensive but coordinated downregulation of ATP-consuming processes. The following phase (days) involves maintaining the cellular and circuit integrity of the deeply depressed brain (Hochachka and Lutz, 2001). Whereas in the mammalian brain it is difficult to distinguish between protective and pathologic responses to such a severe stress because the two are interwoven, in the turtle brain we have the advantage of being able to separate out the key functional responses (Bickler and Donohoe, 2002). Many of the mechanisms behind this remarkable survival capacity have been the subjects of intense research (Lutz et al., 2003a). However, we have little knowledge on the role of HSPs in protecting the turtle brain in anoxia. The object of this study was to examine the effect of early and progressive anoxia on the expression of Hsp72 and Hsc73 in the turtle brain.
MATERIALS AND METHODS
Tissue preparation
Freshwater turtles weighing 300 to 500 g were obtained from a commercial supplier (Lemberger, Oshkosh, WI, U.S.A.). Animals were individually placed in sealed 2-L plastic chambers at 25°C and subjected to one of the following conditions: normoxia, 4 hours of anoxia (99.99% N2), 8 hours of anoxia, or 12 hours of anoxia. Animals were killed and the brains removed. For protein extraction, frozen samples were ground to a powder in liquid nitrogen, resuspended in TRIzol reagent (Life Technologies, Rockville, MD, U.S.A.), homogenized in a handheld glass homogenizer, and extracted exactly according to the manufacturer's instructions.
Immunodetection protocol
Proteins were separated electrophoretically on an SDS-polyacrylamide (12%) gel at 100 V for 2 hours. Subsequently, proteins were transferred for 1 hour at 100 V onto nitrocellulose membranes (Hybond ECL, Amersham Biosciences, Piscataway, NJ, U.S.A.), which were blocked overnight at 4°C in 5% nonfat dried milk in Tris-buffered saline (25-mmol/L Tris-Cl, pH7.5, at 20°C; 150-mmol/L NaCl) and then incubated for 1 hour with a rabbit polyclonal antibody against Hsp72 diluted 1;1,000, with a rat monoclonal antibody against Hsc73 diluted 1:1,000, or with a monoclonal antibody against actin diluted 1:1,000 (Chemicon, Temecula, CA, U.S.A.) with 5% milk in Tris-buffered saline/Tween (SPA-815; StressGen, Victoria, B.C.). After washing in Tris-buffered saline/Tween, the membranes were incubated for 1 hour with a goat antirabbit secondary antibody (1;1,000 dilution, Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.), a goat antirat antibody (1:1,000 dilution, Santa Cruz Biotechnology), or a goat antimouse antibody (1:1,000, Sigma, St. Louis, MO, U.S.A.), respectively, each of which was horseradish peroxidase conjugated. The protein antibody complex was visualized by chemiluminescence (ECL, Amersham) and quantified using a digital camera with image-analysis software (NIH Image 1.60).
Statistical analysis
Results are expressed as mean ± SD. Statistical significance was evaluated using analysis of variance. A value of P < 0.05 was used to denote statistical significance.
RESULTS
To determine whether levels of Hsp72 and Hsc73 protein were modified during anoxia, protein was extracted from homogenized brain tissue obtained from turtles exposed to 0, 4, 8, and 12 hours of anoxia. Antibodies to Hsp72 and Hsc73 used for this study were previously reported to react specifically with turtle isoforms detecting in each case a single discrete spot by two-dimensional gel electrophoresis (Scott et al., 2003). Hsp72 was readily detected in normoxic brain, indicating a high level of baseline expression (Fig. 1) that contrasts with the almost-undetectable levels reported in mammalian brain. Hsc73 was also readily detected in normoxic turtle brain (Fig. 1) at levels that may be comparable with those of mammalian brains (Lipton, 1999).
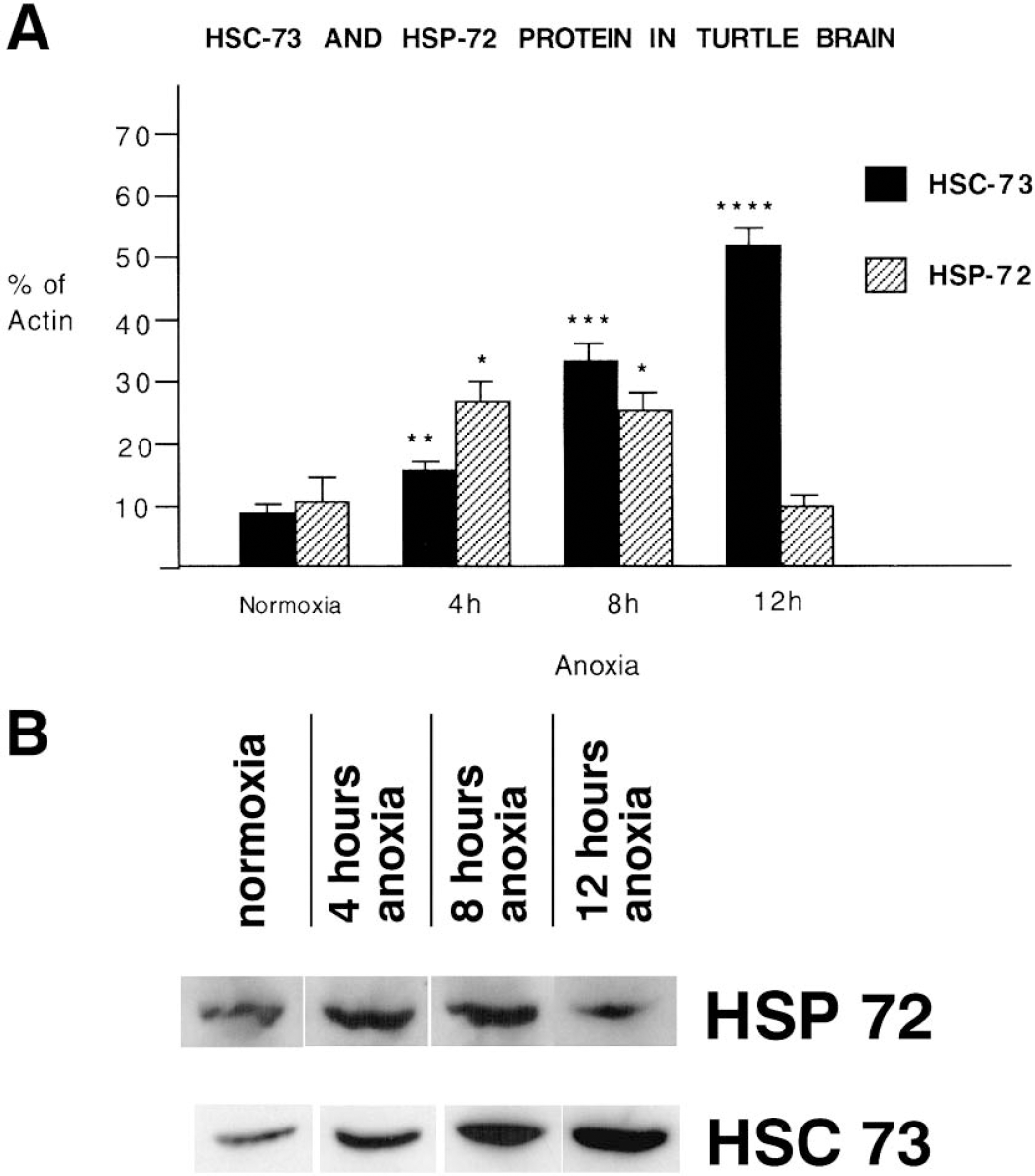
Western blot analysis of levels of Hsp72 and Hsc73 in anoxic turtle brain.
During anoxia, Hsc73 was progressively and substantially increased compared to normoxic levels, rising to 180% ± 12% (P < 0.05%, n = 4) at 4 hours of anoxia to 560% ± 19% at 12 hours anoxia (P < 0.01, n = 4) (Fig. 1). There were no significant changes in levels of expression of actin over the same time periods of anoxia. Hsp72 was also found to be highly inducible, with a substantial increase of 264% ± 13% (P < 0.05) at 4 hours anoxia (Fig. 1). However, at 8 hours anoxia there was no further change with levels of HSP expression of 258% ± 11% (P < 0.05, n = 4) compared with normoxic controls, and by 12 hours Hsp72 had returned to levels (97% ± 7%, n = 4) that did not differ significantly from normoxic control values (Fig. 1).
DISCUSSION
The high levels of Hsp72 in the normoxic turtle brain contrast with the mammalian brain, where in normoxic unstressed brains only low levels of Hsp72 are detected. For example, in the unstressed rabbit brain, values of Hsp72 are only 1.5% to 6.0% those for Hsc73 (Snoeckx et al., 2001). The inducement of Hsp72 by a short mild exposure to ischemia/hypoxia is thought to be an important feature of preconditioning in the mammalian brain, providing temporary protection for a subsequent otherwise injurious ischemic/anoxic insult (Marber et al., 1993). In this regard, the high constitutive levels of Hsp72 in normoxic turtle brain indicate that the turtle has an element of preconditioning already expressed.
Our results show that both Hsp72 and Hsc73 are highly induced in anoxia, which differs from the findings of Scott et al. (2003) who reported a twofold increase in Hsp72 but no increase in Hsp73 after a 24-hour forced dive. However, the current study examined changes in progressive anoxia up to 12 hours, whereas Scott et al. (2003) measured changes after 24 hours of forced submergence. Forced submergence might also be an additional stress for the HSP system.
Hsp72 is strongly expressed early in anoxia but declines to normoxic levels in late (12 hours) anoxia, suggesting that Hsp72 may have a role in stabilizing and rescuing proteins during the initial period of orchestrated metabolic downregulation (Lutz et al., 2003a).
By contrast, the progressive increased expression of Hsc73 during long-term anoxia may indicate that this protein may have an important role in maintaining the structural integrity of the neuronal network during long-term anoxia. In the rat brain, Hsc70 (Hsc73) is important in the maintenance of normal synaptic structure (Bronk et al., 2001), in synaptic plasticity, and in GABA neurotransmission (Jin et al., 2003).
Banking of HSP in anoxia may also be a protective measure against a threatened massive release of damaging ROS that would be expected when the anoxic brain is reperfused with oxygenated blood (Lutz et al., 2003b). There is evidence that increased HSP protects against ROS damage (Plumier et al., 1995).
