Abstract
While medical science has struggled to find ways to counteract anoxic brain damage with limited success, evolution has repeatedly solved this problem. The best-studied examples of anoxia-tolerant vertebrates are the crucian carp and some North American Freshwater turtles. These can survive anoxia for days to months, depending of temperature. Both animals successfully fight any major fall in brain ATP levels, but the strategies they use to accomplish this are quite divergent. The anoxic turtle suppresses brain activity to such a degree that it becomes virtually comatose. The underlying mechanisms involve closing down ion conductances and releasing GABA and adenosine. By contrast, the crucian carp remains active in anoxia, although it suppresses selected brain functions, and avoids lactate self-poisoning by producing an exotic anaerobic end-product. These animals provide unique models for studying anoxic survival mechanisms both on a molecular and physiological level.
Due to its high intrinsic rate of oxygen consumption, the brain is one of the first organs to fail in anoxia, and for most vertebrates, a continuous access to oxygen is a prerequisite for survival. However, there are exceptions: some vertebrates can survive for months without any oxygen. Indeed, evolution has solved the problem of anoxic brain survival several times. Unfortunately, medical science has so far had very limited success in doing the same.
Particularly in aquatic environments, a continuous access to oxygen cannot be taken for granted. Taking up oxygen from water has its special problems. The oxygen concentration in air is 30 times higher than that in fully aerated water, and oxygen diffuses some 10,000 times faster in air than in water. Consequently, some aquatic habitats can become partially or even totally depleted of oxygen. Waters that temporarily become hypoxic range from ice covered lakes and ponds (Ultsch, 1989) to tropical freshwater systems (Chapman et al., 1995; Val et al., 1998), and even coral reefs (Nilsson et al., 2004; Nilsson and Östlund-Nilsson, 2004) Vertebrates that live in habitats where hypoxia, or even anoxia, are regularly encountered have evolved physiological mechanisms to cope with such challenges (Lutz and Storey, 1997). Often these mechanisms involve supplemental air breathing, but in some cases, the organ systems, including the brain, of these animals have been adapted to survive on little or no oxygen.
The most extreme examples of anoxia tolerant vertebrates are probably the crucian carp (Carassius carassius) and some species of North American freshwater turtle, particularly Trachemys scripta and Chrysemys picta. The crucian carp inhabits small lakes and ponds in Northern Europe, which during the winter may become anoxic for several months due to a thick ice cover which blocks oxygen diffusion as well as light needed for photosynthesis (Holopainen and Hyvärinen, 1985). The turtles have evolved their anoxia tolerance in response to overwintering in anoxic mud on the bottom of lakes and rivers, with no access to the atmosphere (Ultsch, 1989). The turtles are primarily air breathers, but can supplement alveolar respiration with cutaneous, esophageal or rectal respiration. However, these additional routes of oxygen uptake are not essential since they indeed do survive even in anoxic water (Ultsch and Jackson, 1982). In both turtles and crucian carp, the capacity for anoxic survival is extended over a wide temperature range. At close to 0°C, these vertebrates can survive anoxia for several months, while at room temperature, they tolerate anoxia for at least a day or two (Fig. 1).
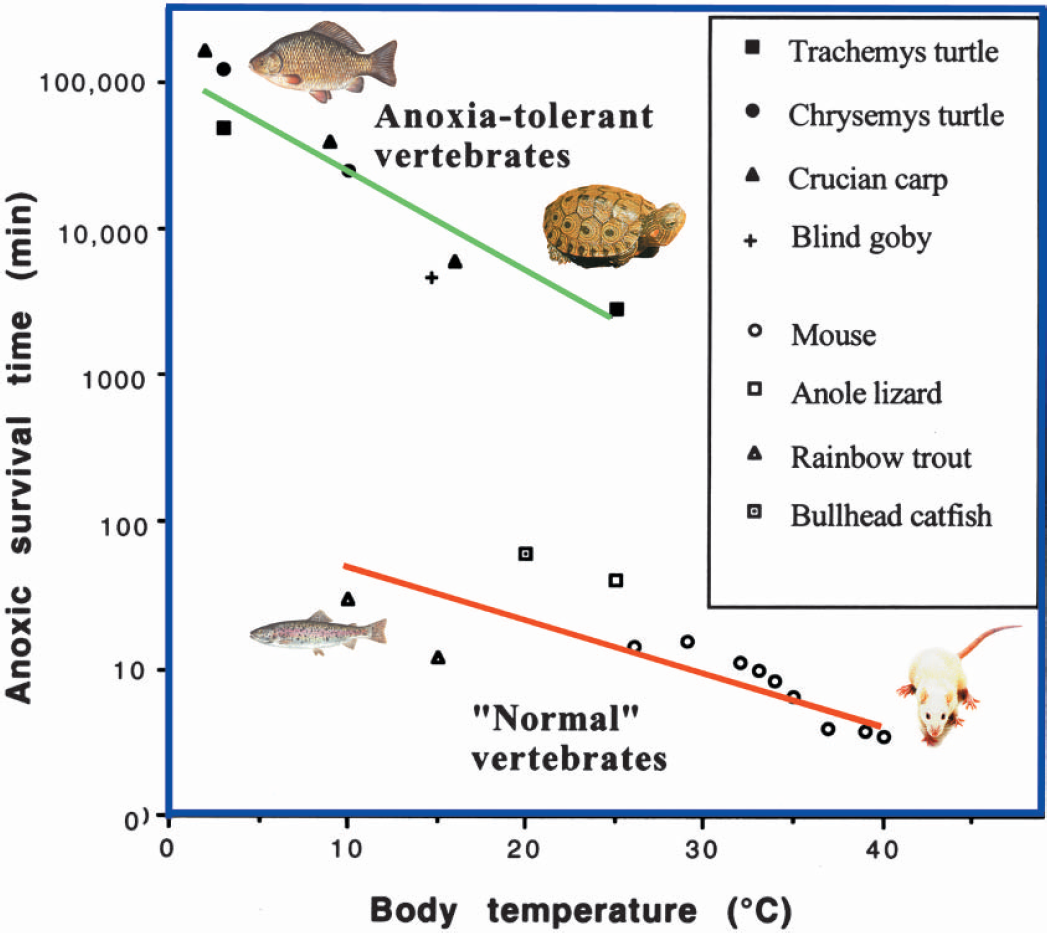
Anoxic survival time in “normal” vertebrates and in anoxia tolerant vertebrates. Note that cold blooded vertebrates in general are as sensitive to anoxia as mammals, if temperature is taken into account, and that the anoxia-tolerant vertebrates survive anoxia about 1000 times longer than other vertebrates. In general, metabolic rate falls sharply with body temperature. A main reason why anoxia tolerant vertebrates survive anoxia longer at cold temperatures is probably that their glycogen stores last longer. For anoxia-intolerant vertebrates, a main benefit of a reduced temperature is that it slows down the loss of ATP and ion homeostasis, and the subsequent onset of degenerative processes. Redrawn from Lutz et al. (2003).
There are also some other, but less well studied, examples of anoxia-tolerant vertebrates, like the Californian blind goby (Typhlogobius californiensis) that can survive 80 hours of anoxia at 15°C (Congleton, 1974). However, it should be emphasized that hypoxia tolerance is not a general phenomena in aquatic vertebrates (Fig. 1). Fish brains, for example, appear to have about the same weight specific rate of metabolism as mammalian brains, if temperature is taken into account (Nilsson, 1996). Moreover, studies on rainbow trout (Oncorhynchus mykiss) show that it has a sensitivity to anoxia that equals that of mammals, and just like a mammal, the trout brain rapidly loses ion homeostasis and releases glutamate when exposed to anoxia (Nilsson et al., 1993a; Hylland et al., 1995).
Over the last two decades, anoxia tolerant vertebrates have become model animals in the study of anoxic survival. Unlike mammals, survival in anoxia is the control situation in turtles and crucian carp. This allows us to selectively block various mechanisms to evaluate their role in anoxic survival. This situation is the complete opposite to that in the mammalian brain, where death is the norm in anoxia, and any experimental attempts to extend anoxic survival by boosting or blocking a particular mechanism is likely to be hampered by failures of other functions. In mammals, anoxia is synonymous with catastrophe, and a major problem with studying the anoxic mammalian brain is that the physiological changes are so rapid and complex, making it extremely difficult to sort out damaging events from useful defense mechanisms (Bickler et al., 2002; Lutz et al., 2003).
In this review, we will summarize the present knowledge on anoxic survival mechanism in crucian carp and freshwater turtles, as well as in some other emerging examples of anoxia-tolerant vertebrates. The focus will be on the brain, but we will also discuss physiological and ecological factors that are intimately connected to the anoxic survival of these animals. Evolution has produced anoxia tolerant animals several times, and it is now quite clear that there is biodiversity also in anoxic survival (Lutz and Nilsson, 1997; Hochachka and Lutz, 2001). Thus, we will describe similarities as well as differences in the strategies and mechanisms employed by these different animals to cope with anoxia.
Metabolic depression, electric activity and a rare metabolic end product
The crucian carp and the freshwater turtles deviate from mammals in one crucial aspect: they maintain brain ATP levels when exposed to anoxia (Fig. 2). Clearly, many of the deadly processes initiated by anoxia are directly or indirectly triggered by the inability of the anoxic mammalian brain to maintain ATP levels. When oxidative phosphorylation stops in anoxia, the brain is left with glycolysis as the only major ATP producing process. Unfortunately, glycolysis has an ATP yield that is less than 1/10 of that of the complete oxidation of glucose to CO2 and H2O (Hochachka and Somero, 2002). As far as we can see, there are only two ways that brain ATP levels can be defended in anoxia. Either the use of ATP is drastically suppressed (a strategy often termed metabolic depression), or the glycolytic production of ATP is strongly up-regulated (Pasteur effect). The crucian carp and the turtles clearly differ in the degree to which these two options are utilized.
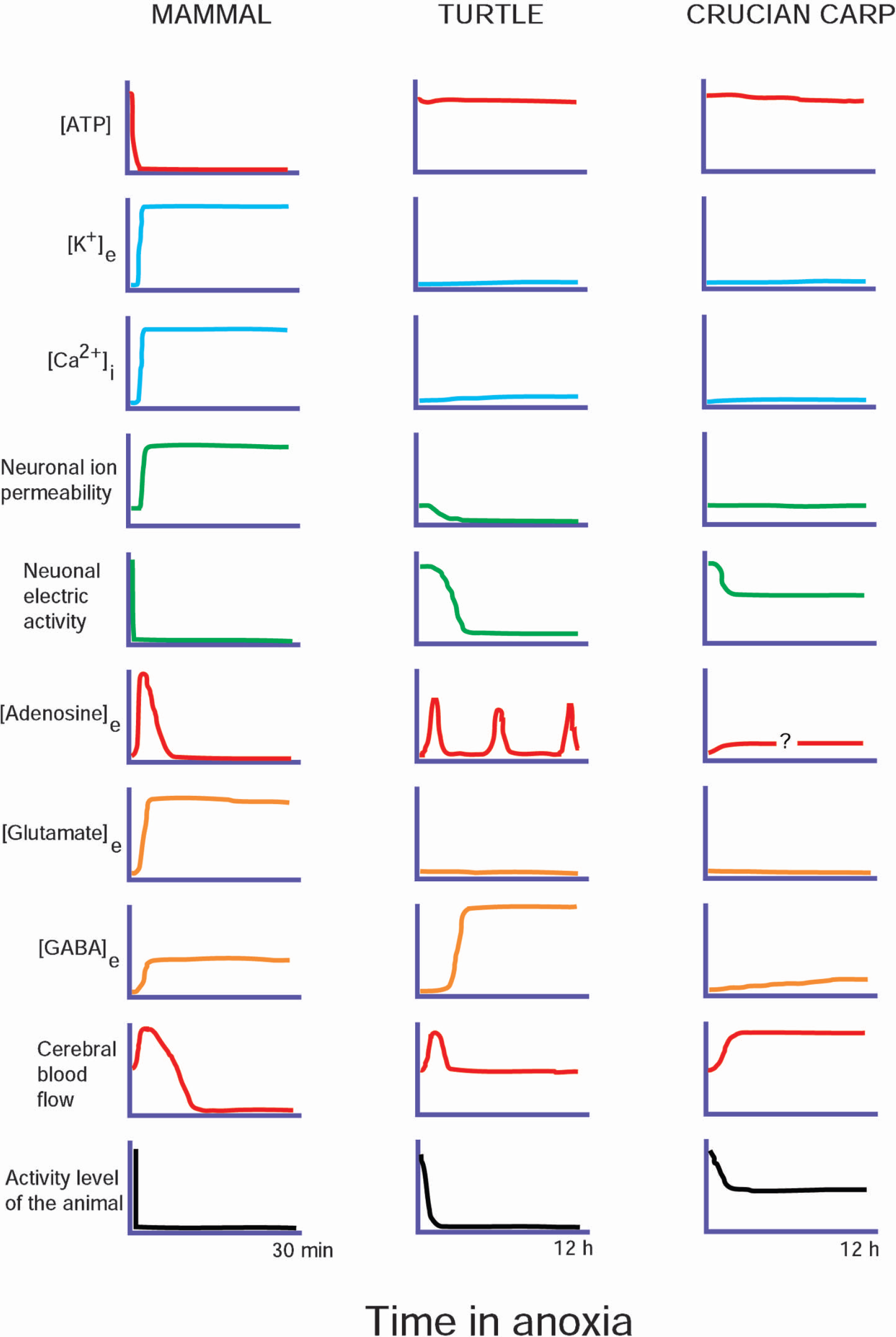
Changes in major factors determining life and death in the anoxic brain of mammals and in the anoxia-tolerant turtle and crucian carp. While many of the changes in the mammalian brain are maladaptive, reflecting a deadly catastrophe, those seen in the turtle and crucian carp are adaptive processes aimed to promote anoxic survival. The differences between the turtle and the crucian carp reflects their discrepant strategies for anoxic survival: maximal neuronal suppression in the turtle, after some initial adjustments, and continuous activity in the crucian carp. See text for more detailed discussion on the various factors.
The level of physical activity displayed by these animals during anoxia already discloses an obvious difference between the anoxic survival strategies of the crucian carp and the turtle. While anoxic turtles are virtually comatose, the crucian carp still swims around in anoxia, although at a reduced level (Nilsson et al., 1993b). This difference in activity level was recently found to be paralleled by striking differences in the circulatory adjustments to anoxia. In the turtle, anoxia leads to peripheral vasoconstriction and a blunted autonomic control. Simultaneously, heart rate and cardiac output fall by 80% (Hicks and Farrell, 2000a, b; Stecyk et al., 2004). By contrast, heart rate, cardiac output and autonomic control are maintained for several days in the anoxic crucian carp (JAW Stecyk, K-O Stensløkken, AP. Farrell, GE Nilsson, unpublished). Both the turtle and crucian carp show a doubling in brain blood flow within the first minutes of anoxia. However, while this increase in brain blood flow is sustained in the anoxic crucian carp (Nilsson et al., 1994), probably in order to allow maintained high neural activity level, cerebral circulation falls back to pre-anoxic levels within the first hours of anoxia in the comatose turtle (Hylland et al., 1994; Stecyk et al., 2004). Nevertheless, in both cases the increase in brain blood flow appears to be mediated by adenosine, as it can be fully blocked by aminophylline.
The difference in activity between crucian carp and turtles is also reflected on the metabolic level. Whole body metabolism (measured as heat production) is more drastically reduced in the turtle than in the crucian carp. A 90 to 95% reduction in heat production has been measured in turtles (Jackson, 1968), while in the goldfish (Carassius auratus), a close relative to the crucian carp, anoxia reduces heat production to 1/3 of the normoxic level (Van Waversveld et al., 1989). With regard to the brain, there are no direct measurements of metabolic rate in vivo in these animals. However, estimates based on lactate production in anoxia suggests a 70 to 80% fall in brain ATP turnover in the anoxic turtle brain (Lutz et al., 1984), so that brain energy needs can be fully met even if anaerobic glycolysis is suppressed (Storey, 1996). In the crucian carp, on the other hand, measurements of lactate and heat production (microcalorimetry) in telencephalic brain slices suggest a mere 30% reduction in ATP turnover and an up-regulation of glycolysis (Johansson et al., 1995).
More than 50% of brain ATP use appears to be devoted to ion pumping needed to maintain electric gradients over cell membranes, which in turn are a prerequisite for electric activity and transport of neurotransmitters and metabolites (Erecinska and Silver, 1994). Thus, suppressing electric activity should be an effective way of reducing the use of ATP. Indeed, this is what anoxic turtles do. After a few hours of anoxia, EEG in turtle brain is suppressed to an extent that it is hardly measurable, although periodic minor activity bursts can be recorded (Fernandes et al., 1997). Also electrically evoked activity is strongly suppressed in the anoxic turtle brain (Feng et al., 1988a, b).
For the crucian carp, remaining active in anoxia will of course mean that it has to endure anoxia with the brain turned on (Lutz and Nilsson, 1997; Nilsson, 2001). Although EEG activity has not been recorded in anoxic crucian carp (not an easy task due to electric disturbance related to water movements), it is unlikely that its EEG is suppressed to the degree seen in the anoxic turtle. However, the anoxic crucian carp appears to suspend some sensory functions of less importance under the long anoxic winter, such as vision and hearing. Light evoked potentials are down regulated in the anoxic crucian carp retina and optic tectum (Johansson et al., 1997), and sound evoked potentials in the auditory nerve of goldfish (Suzue et al., 1987) virtually disappears in anoxia. In addition, if given the chance, the anoxic goldfish (and most likely the anoxic crucian carp) will reduce its metabolic requirements by moving to cooler water (Rausch et al., 2000). This “voluntary” hypothermia is seen in many animals when exposed to hypoxia, including mammals (Wood et al., 1985), and is an excellent example of how the solutions provided by natural selection can hint at powerful tools, in this case hypothermia, to counteract hypoxic damage in human tissues (Polderman, 2004).
So how can the anoxic crucian carp sustain such a high degree of activity compared to the turtles? The answer may lie in an adaptation that is unique to the crucian carp and two of its closest relatives (goldfish and bitterling). It is the exotic ability to turn lactate into ethanol during anoxia (Shoubridge and Hochachka, 1980; Nilsson, 1988). The reaction takes place in muscle in three steps. Lactate is turned into pyruvate by lactate dehydrogenase, pyruvate is turned into acetaldehyde by an untypical pyruvate dehydrogenase, and finally acetaldehyde it turned into ethanol by alcohol dehydrogenase. All other tissues, including the brain, can only produce lactate in anoxia. The lactate is transported in the blood to the muscle from which the ethanol is released back into the blood for transport to the gills where it leaves the fish. The blood level of ethanol does not rise high enough to significantly suppress nervous activity (reaching a steady state level below 10 mM; Van Waarde et al., 1993). This metabolic arrangement allows the crucian carp to endure long-term anoxia without suffering acidosis and a buildup of lactate. The occurrence of ethanol production probably relieved the crucian carp from the necessity of reducing lactate production through extreme metabolic depres ion. The turtles are not in the possession of the ethanol producing pathway and after several months in anoxia, lactate levels of up to 200 mM are often seen in turtle blood and tissues (Jackson, 2002). The turtle does its best to buffer the lactate load with calcium carbonate from its bones and shell (Jackson, 2002). The ethanol producing strategy has of course an energetic drawback, as this very energy-rich compound is released to the water and forever lost. The crucian carp accounts for this by having a liver with the largest glycogen store of any vertebrate, and the only factor that appears to limit its anoxia endurance is the total depletion of this store (Nilsson, 1990).
When examining how neural metabolic depression is achieved, it becomes apparent that the differences displayed by crucian carp and turtles in physical and metabolic activity are also reflected in the mechanisms employed to suppress brain energy demands in anoxia.
The hypoxia signal
In order to initiate the appropriate defense mechanisms to accommodate hypoxia, it is first necessary to detect it. There are two basic sets of detectors of hypoxia: (1) metabolic indicators that are triggered when the cell goes into a state of hypoxia-induced energy deficiency, and (2) molecular oxygen sensors that can be directly linked to gene regulation (Lutz and Prentice, 2002).
Metabolic indicators of hypoxia.
In the mammal, low oxygen quickly results in a fall in ATP and a consequent increase in the release of adenosine, and it is well established that both changes initiate a variety of mechanisms to reduce metabolic demand (Lipton, 1999). The hypothesis that adenosine plays a critical role in anoxia tolerance in the turtle is supported by the findings that (1) there is an almost immediate, substantial rise in extracellular adenosine in the anoxic turtle brain, probably linked to the simultaneous fall in ATP (Nilsson and Lutz, 1992), and (2) that adenosine receptors mediate an increase in cerebral blood flow early in anoxia (Hylland et al., 1994), a reduction in membrane K+ leakage (Pek and Lutz., 1997), a reduction in glutamate efflux (Milton et al., 2002), and a down regulation in NMDA receptor activity and whole cell conductance (Buck and Bickler, 1998; Ghai and Buck, 1999). Finally, blocking adenosine A1 receptors pharmacologically causes rapid depolarization in the anoxic turtle brain slice (Peréz-Pinzón et al., 1993). One reason why adenosine can be so effective in the turtle brain is that, unlike the mammal, no deterioration in adenosine binding sites are seen over 24 hours anoxia (Lutz and Manuel, 1999). In addition, it is of course likely that the adenosine release in the turtle brain is acting on a system that is in fact capable of making ATP production meet energy need, while in the mammal, all the mechanisms activated by adenosine are not enough to achieve a balanced energy budget in face of such a drastic event as anoxia.
There is also evidence that adenosine has an important role in protecting the brain in anoxia tolerant fish. In the crucian carp the anoxia induced increase in brain blood flow is probably adenosine moderated since it is blocked by superfusing the brain with the adenosine receptor blocker aminophylline (Nilsson et al., 1994). In hepatocytes from the related goldfish, adenosine was shown to have a powerful depressant effect on both protein synthesis and Na+/K+ ATPase activity (Krumschnabel et al., 2000). Furthermore adenosine suppresses K+ stimulated Ca2+-dependent glutamate release in goldfish cerebellar slices (Rosati et al., 1995), and blocking adenosine receptors in anoxic crucian carp causes a 3-fold increase in the rate of ethanol release to the water which is indicative of a significant inhibition of the metabolic depression (Nilsson, 1991). However, unlike the turtle, microdialysis experiments have so far failed to detect an increase in extracellular adenosine in the anoxic crucian carp brain (P Hylland and GE Nilsson, unpublished).
Molecular oxygen sensors.
Putative sensors of oxygen in cells include membrane proteins such as ionotropic ion channels, particularly voltage–dependent potassium channels (Kv channels), membrane or cytosolic heme proteins, mitochondrial proteins and/or oxygen sensitive transcription factors such as HIF-1 and NF-kappaB (Bickler and Donohoe, 2002). In the mammal, one of the most important hypoxia driven factors, the hypoxia-inducible HIF-1alpha, plays a major role in coordinating many adaptive responses to hypoxia (Haddad, 2002).
Many of these components have been identified, and presumably operate, in the turtle brain: HIF-1alpha mRNA has been detected in turtle brain and interestingly shows no changes in level during anoxia and subsequent reoxygenation. (Prentice et al., 2003). This is consistent with the findings in mammalian systems that HIF-1 regulation does not occur at the transcriptional level but primarily at the post-translational level. Gene transcription of Kv channels is reversibly regulated by oxygen supply in the turtle brain (Prentice et al., 2003). Interestingly, in the anoxic turtle brain NF-kappaB shows maximal DNA binding at six hours of anoxia (Lutz and Prentice, 2002). It is possible that the late translocation of NF-kappaB to consensus DNA binding sites is part of a preemptive defense mechanism against reoxygenation ROS damage and incipient apoptosis (Lutz and Prentice, 2002).
Molecular effectors
In turtles, there is a differential expression between normoxic and anoxic turtle brains of transcripts encoding the immediate early gene products c-fos and c-jun, heat shock proteins (Hsp) and the apoptosis regulators bcl-2 and bax (Prentice et al., 2003). Nad5 and Cox1, which are candidate molecular components of the oxygen sensing/anoxia adaptation pathway, are both induced by hypoxia in the brain of turtles (Cai and Storey, 1996).
The ATP dependent Hsps are of particular interest. These are highly evolutionarily conserved chaperones, which recognize and bind nascent and unfolded proteins, thus preventing aggregation, and facilitating correct protein folding (Giffard et al., 2004). The Hsp70 family in particular suppresses multiple types of cell death pathways, including necrotic death, classical apoptosis, and programmed cell death that is independent of caspases and not blocked by bcl-2 (Giffard et al., 2004).
Hypoxia/ischemia produces an increase in the inducible Hsp72 in the mammalian brain (Lipton, 1999) and in the turtle. In the turtle brain Hsp 72 is induced in early anoxia, peaking at about 8 hours anoxia but then falls, reaching normoxic levels at 12 hours in anoxia (Prentice et al., 2004). However, unlike mammals, where heat shock cognate (Hsc)73 is only slightly inducible, in the turtle brain Hsc73 is progressively elevated over 12 hours of anoxia (Prentice et al., 2004). This differential expression of Hsp proteins suggests that Hsp72 and Hsc73 have different roles during brain anoxia. The (comparatively) short term rise and fall in Hsp 72 may be relate to it having a protective role during the initial transition to the hypometabolic state, a period of substantial metabolic changes. The continued and increased presence of Hsc 73 during long term anoxia indicates that this protein may be involved in “housekeeping” roles that are necessary to ensure the functional integrity of the neuronal network during the long term hypometabolic phase.
Ion channels
In contrast to mammals, turtles and crucian carp appear to maintain a well regulated ion-homeostasis during anoxia with no or only minor rises in extracellular [K+] and intracellular [Ca2+] (Sick et al., 1982; Johansson and Nilsson, 1995; Nilsson, 2001). In turtles, there is a slow and relatively small rise in extracellular [Ca2+] during anoxia that may reflect the increase in blood and extracellular [Ca2+] related to the breakdown of bone and shell needed to buffer the lactate load (Bickler, 1998). A moderate and controlled elevation in intracellular [Ca2+] (less than 300 nM) has been suggested to initiate neuroprotective mechanisms in turtles and may in some mammalian models play a similar role (Bickler 1998; Bickler and Hansen, 1998; Bickler and Donohoe, 2002; Bickler, 2004). Indeed, while too much [Ca2+] is clearly deadly, too little may have apoptotic effects as neurons appear to have a [Ca2+] set-point for survival (Johnson et al., 1992; Zipfel et al., 2000).
Since ion pumping accounts for more than 50% of the energy use of the normoxic neuron, a reduction in ion permeability, often termed “channel arrest”, can have important savings (Lutz et al., 1985; Hochachka, 1985). Indeed, turtles appear to utilize this strategy extensively, as anoxia causes a substantial down regulation in K+, Na+ and Ca2+ fluxes in turtle brain neuronal membranes. In the turtle cerebral cortex, intracellular to extracellular K+ flux is reduced by about 50% over the first hour of anoxia and plateaus at about 30% of the normoxic flux over the next few hours (Pek and Lutz, 1998). Opened KATP channels and activated adenosine A1 receptors, mediate this down-regulation of K+ flux (Pek and Lutz, 1998). Correspondingly, the mRNA levels of Kv channels, which are important determinants of brain electrical activity and may act as oxygen sensors, are reduced to less than 1/5 of normoxic levels at 4 hours of anoxia and restored to normal within 4 hours of subsequent reoxygenation (Prentice et al., 2003). This indicates that in the turtle brain, gene transcription of Kv channels is reversibly regulated by oxygen supply.
There is a 40% decrease in the density of voltage gated Na+ channels in the anoxic turtle cerebellum (Peréz-Pinzón et al., 1992)—a probable cause of the elevation in the action potential threshold that occurs in the anoxic turtle brain (Sick et al., 1993). Interestingly, Xia and Haddad (1993) reported a decrease in Na+ channel mRNA and saxitoxin binding density in the brains of rats that had been exposed to hypoxia (10% O2) for one month. They speculated that, as in the turtle, down-regulation of Na+ channels during long-term hypoxia in mature mammalian brains is an adaptive cellular response, aimed at minimizing the mismatch between energy supply and demand.
The NMDA glutamate receptor is a high-flux cation channel that is highly permeable to Ca2+ and is required for fast excitatory neurotransmission. Over-stimulation of this receptor from the excessive glutamate release characterizing the anoxic/ischemic mammalian brain results in a massive inflow of Ca2+. This uncontrolled rise in Ca2+ activates a wide variety of pathological processes with ultimately fatal consequences (Siesjö, 1994). In the turtle brain, the NMDA glutamate receptor activity is progressively reduced during anoxia (Bickler et al., 2000), a process mediated by the activation of phosphatase 1 or 2A (Bickler and Donohoe, 2002) and adenosine receptors (Buck and Bickler, 1998). Application of adenosine to turtle brain slices results in a reduction in N-methyl-D-aspartate receptor (NMDA) open probability and whole cell conductance (Buck and Bickler 1995; 1998; Ghai and Buck, 1999).
While “channel arrest” appears to be an important protective response in the anoxic turtle brain, this strategy does not appear to be used in the crucian carp. There is no evidence for reduced neural K+ or Ca2+ permeability or fluxes during anoxia in the crucian carp brain (Johansson and Nilsson, 1995; Nilsson, 2001). This difference is likely to be related to the contrasting physiological states that these species adopt in anoxia. The continued physical activity of the crucian carp in anoxia is possibly not compatible with such a drastic measure for metabolic depression as closing down ion conductances.
Neurotransmitters
Unlike the mammal, the turtle brain avoids increases in the excitatory neurotransmitters glutamate and dopamine during anoxia (Nilsson and Lutz, 1991; Milton and Lutz, 1998). Low extracellular glutamate levels are maintained in the anoxic turtle brain through a reduction in glutamate release combined with the continued operation of glutamate uptake transporters (Milton et al., 2002). In early anoxia, reduction in glutamate release is mediated by the activation of adenosine receptors and the opening of K+ ATP channels (Milton et al., 2002). Since glutamate uptake is energetically expensive the continued transporter activity suggests that glutamate release has an important function in anoxia survival.
In contrast to glutamate, extracellular GABA starts to rise after about 2 hours of anoxia, reaching levels 80 times higher than normal (Nilsson and Lutz, 1991). The rise in GABA is accompanied by an increase in GABAA receptor number, which continues to increase for at least 24 hours (Lutz and Leone-Kabler, 1995). The up-regulation in GABAA receptors may function to increase the effectiveness of the inhibitory action of GABA. It could be argued that the GABA release functions as an endogenous anesthesia for the anoxic turtle brain, reducing metabolism and protecting the anoxic brain in much the same way as anesthetics do in mammals (Lutz et al., 2003) – providing an evolutionary precedent for such treatments.
Like the turtle, extracellular GABA levels rise in the brain of anoxic crucian carp while extracellular glutamate remains low (Hylland and Nilsson, 1999). However, compared to the massive increase in extracellular [GABA] in the anoxic turtle brain, the rise in extracellular [GABA] in the crucian carp brain (telencephalon) is quite modest, the level being doubled after about 5 hours of anoxia (Hylland and Nilsson, 1999). There is also a considerable individual variation in the extracellular [GABA] rise during anoxia, which may suggest that GABA is involved in fine tuning neural suppression.
Interestingly, the potential for GABA release in the crucian carp telencephalon appears to be considerably higher than that for glutamate. Hylland and Nilsson (1999) found that running a high-[K+] Ringer through the microdialysis probe, to depolarize the surrounding tissue, caused the extracellular GABA level to rise 14 times, while that of glutamate was barely doubled (Hylland and Nilsson, 1999). Moreover, when the crucian carp telencephalon was superfused with the glycolytic inhibitor iodoacetate, forcing neural ATP levels to plummet, the resultant increase in extracellular [GABA] was both faster and more massive (a 10-fold increase after 30 minutes) than that of glutamate (a 3-fold increase after 2 hours) (Hylland and Nilsson, 1999). These results could be taken as evidence for a second line of defense: energy deficiency in crucian carp brain may initiate a major GABA release which could cause a neuronal depression large enough to allow ATP levels to be restored.
The metabolic interrelation between GABA and glutamate is interesting from an anoxia-defense perspective. GABA is synthesized from glutamate in a single oxygen-independent step, catalyzed by glutamate decarboxylase (GAD). By contrast, both the synthesis of glutamate and the breakdown of GABA are linked to oxygen dependent processes and will therefore stop in anoxia. The net effect is a steady increase in [GABA] and a corresponding fall in [glutamate] during anoxia (Siesjö, 1978; Nilsson and Lutz, 1993). GABA is the major inhibitory neurotransmitter and glutamate the major excitatory neurotransmitter in all vertebrates as well as many invertebrates, including the platyhelminthes, the most primitive group of animals known to possess neurotransmitters (Gerschenfeld, 1973; Usherwood, 1978; Koopowitz and Keenan, 1982; McGeer and McGeer, 1989; Restifo and White, 1990). This suggests that their respective roles were fixed early in evolution and that there is a considerable selection pressure to maintain these roles. It has been hypothesized that the underlying selection pressure is hypoxia: a system where a fall in oxygen will automatically make inhibitory neurotransmitter levels rise and excitatory fall is likely to be desirable for maintaining hypoxic metabolic depression, and may in some animals be a mechanism for inducing metabolic depression during hypoxia (Nilsson and Lutz, 1993). Indeed, blocking the hypoxia induced rise in brain tissue [GABA] pharmacologically (using the GAD inhibitor isoniazid) in crucian carp makes the fish release three times as much ethanol to the water (while normoxic oxygen consumption is unaffected), suggesting a profound inhibition of metabolic depression (Nilsson, 1992).
Still, a release of glutamate during energy deficiency may initiate protective mechanisms. In a close relative to the crucian carp, the goldfish, one class of glutamate receptors, the group II metabotropic glutamate receptors, appears to be involved in attenuating the effects of anoxia in brain (Poli et al., 2003). Such a mechanism could be particularly important in the goldfish, which appears to be somewhat less well adapted to anoxia than the crucian carp (possibly a side effect of long domestication), and may experience elevated extracellular [glutamate] as a consequence of a poorly defended ATP level (Van Ginneken et al., 1996).
Anoxic preconditioning from a comparative perspective
During the last decade, much effort in ischemia research has been a focused on a phenomenon called preconditioning (Dirnagl et al., 2003). Both the brain and heart of mammals show ischemic/hypoxic preconditioning, i.e. the seemingly paradoxical situation that sublethal ischemia/hypoxia exposures subsequently protects the tissue from damage caused by a more severe ischemic insults. As pointed out by Dirnagl et al. (2003), hypoxic preconditioning was shown to reduce anoxic damage in the mammalian brain already by Dahl and Balfour (1964). However, in comparative physiology, a similar, or maybe identical, phenomenon was known already in the 1950s under the term hypoxia acclimation (or acclimatization when it occurs in the natural habitat).
It appears likely that the mechanisms underlying hypoxia acclimatization have evolved because of their significant survival value for the animal. In nature, aquatic animals are often exposed to progressively more severe hypoxia due to seasonal changes in oxygen availability. The darkness of the winter in the Northern hemisphere will inevitably result in reduced rates of photosynthetic oxygen production by plants, particularly if the aquatic habitat becomes ice covered. As a result, oxygen consumption could readily exceed oxygen production, and water oxygen levels will start to fall. In 1955, Shepard showed that trout (Salvelinus fontinalis) acclimated to hypoxia increased their ability to take up oxygen from the water. In 1957, Prosser and co-workers showed that in the hypoxia acclimated goldfish the rate of oxygen consumption of the whole animal was decreased as well as that of its brain and muscle tissue. These early results have an interesting parallel in recent studies on mammalian hearts, showing that ischemic preconditioning lowers the rate of cardiac oxygen consumption (Rudzinski et al., 2002; Tanoue et al., 2002). However, it is still not clear if preconditioning reduces brain oxygen demands (Dirnagl et al., 2003).
There are even lunar effects that appear to result in adaptive hypoxic acclimatization, as illustrated by the epaulette shark (Hemiscyllium ocellatum) on the Great Barrier Reef. At temperatures close to 30 °C, this small shark tolerates several hours of severe hypoxia (pO2 = 7.5 mmHg) and at least an hour of anoxia, without any drop in brain [ATP] or delayed neuronal death (Wise et al. 1998; Renshaw and Dyson, 1999; Renshaw et al., 2002, Routley et al., 2002). The epaulette shark inhabits shallow reef platforms that can become cut off from the surrounding ocean at low tides. When this occurs at night, the respiration of the coral reef organisms can lead to drastically reduced oxygen levels.
The epaulette shark's encounter with hypoxia on the reef does not happen abruptly. The hypoxic episodes occur during a period of spring tides. Initially during such a period, the tides become lower and lower over subsequent nights. Thus, the epaulette shark will experience longer and longer periods of hypoxia, allowing it to gradually acclimatize to hypoxia over a period of a few days. This situation becomes a natural parallel to hypoxic preconditioning.
Laboratory experiments on the epaulette shark show that repeated exposures to hypoxia suppress the shark's rate of oxygen consumption (Routley et al., 2002). Adenosine has been implicated in ischemic preconditioning in mammals, and interestingly, adenosine appears to play an important role in the hypoxia tolerance of the epaulette shark and in its acclimatization to anoxia. Renshaw et al (2002) found that, after about 36 minutes in anoxia, epaulette sharks goes into deep neural depression (loss of the righting reflex and responses to external stimuli) while brain ATP levels are still maintained. However, in a second anoxic episode, 24 hours later, the neural depression occurred faster (after 16 minutes) unless the sharks had been treated with the adenosine receptor blocker aminophylline. In the aminophylline treated sharks, the comatose state was not reached until after 54 minutes, at which point brain ATP levels had started to fall (Renshaw et al., 2002).
Hypoxic preconditioning in the epaulette shark may also involve nitric oxide. Exposing epaulette sharks to hypoxia causes a dramatic increase in the activity of nitric oxide synthase in blood vessels and neurons in the brain (Renshaw and Dyson, 1999). The functional significance of this nitric oxide synthase upregulation is unknown. However, it is tempting to suggest that it is involved in a preconditioning response, particularly since there is evidence for an involvement of this enzyme in hypoxic preconditioning in mammalian models (Gidday et al., 1999; Kapinya et al., 2002; Willmot and Bath, 2003).
However, the champions of anoxia tolerance, turtles and crucian carp, will survive anoxia also without preconditioning. Here, all the mechanisms needed for anoxic survival are apparently already in place, as they readily tolerate anoxia without any warning. Indeed, there is evidence that can be interpreted as if the turtle is “constitutively preconditioned”. It is in possession of many of the key mechanisms identified in mammalian preconditioning These include HSP synthesis, particularly Hsp 72, and Bcl-2 induction, activation of KATP channels, activation of NF-kB and, perhaps most important, the activation of adenosine receptors (Lipton, 1999; Prentice et al., 2003, 2004; Yellon and Downey, 2003). These animals therefore may serve as valuable models to identify the critically important elements of mammalian preconditioning and their mode of protection.
Conclusions
Anoxic survival strategies in crucian carp and turtles show both similarities and differences (Fig. 3). A key strategy adopted by both animals is to defend their brain ATP levels during anoxia by matching ATP use with glycolytic ATP production. Hereby, a loss of ion homeostasis over the neural membrane can be avoided. However, the means by which the energy budget, and thereby ATP levels, is defended show striking differences. The turtle displays a profound neuronal depression initially achieved through adenosine release and subsequently maintained through a down regulation of ion conductances (“channel arrest”) and a massive release of GABA. This renders the anoxic turtle virtually comatose. The crucian carp, which remains active in anoxia, although at a reduced level, does not appear to down regulate ion conductances or rely on a massive GABA release. Instead, it up-regulates glycolysis and suppresses some neural functions. Possibly a regulated moderate increase in GABAergic activity is responsible for tuning down energy use in the crucian carp brain. The reason why the crucian carp can afford a relatively high level of activity during anoxia is probably related to its ability to produce ethanol as the major anaerobic end product, thereby avoiding an enormous lactate load. Thus, a life threatening fall in ATP levels can be avoided without such a drastic measure as channel arrest.
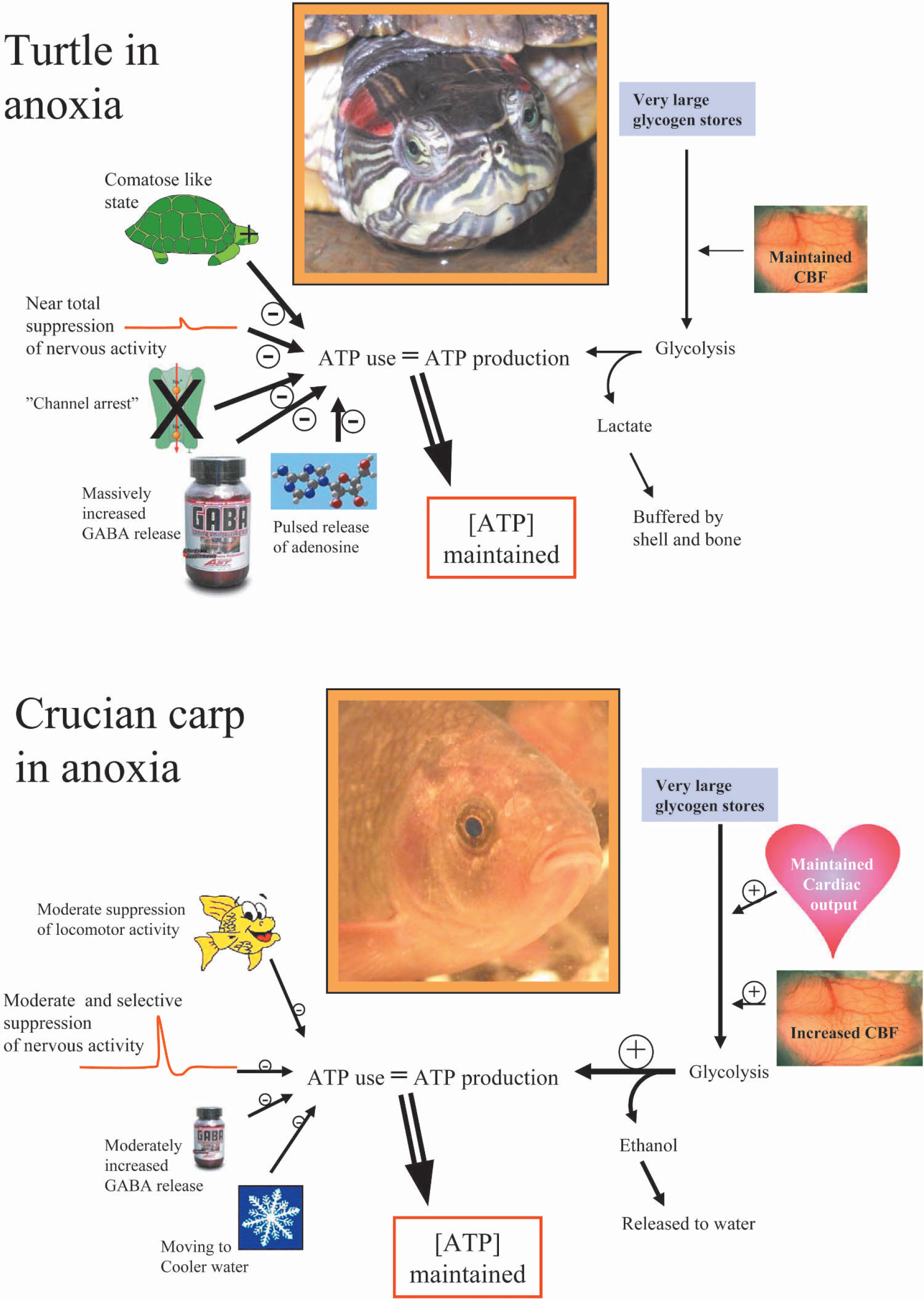
The key to anoxic survival in both turtles and crucian carp is their ability to maintain brain tissue ATP levels during anoxia. This can only be done by matching ATP use with glycolytic ATP production. The turtle strategy is to reduce ATP use as much as possible by a near total suppression of nervous activity mediated by reduced ion permeability of membranes and release of inhibitory factors like GABA and adenosine. Hereby glycolytic lactate production can be reduced to levels that can be tolerated through buffering. The crucian carp remains active in anoxia and is able to match ATP use with ATP production through upregulated glycolysis supported by maintained cardiac output and increased cerebral blood flow (CBF). Here, the main role of its relatively modest metabolic depression is probably to save on the glycogen stores, since it avoids lactate poisoning and acidosis by turning lactate into ethanol.
Much remains to be learned, particularly with regard to molecular mechanisms involved in the anoxic survival strategies adopted by anoxia-tolerant animals. Here, the turtles and crucian carp provide unique opportunities for revealing adaptive molecular mechanisms, since these animals actually survive anoxia long enough to allow substantial, adaptive and coordinated changes in gene expression to take place.
Compared to the vast research resources focused on studying the anoxic/ischemic catastrophe of the mammalian brain, the collective research effort put into clarifying mechanisms of anoxic survival of anoxia-tolerant brains is very modest. In this field, we are therefore still asking very basic questions and the knowledge of anoxic survival mechanisms are rarely as detailed as that of the mechanisms of death in the anoxic mammalian brain. Hopefully, this review can stimulate more neurobiologists to take on the rewarding task of finding out how evolution has solved the problems of anoxic brain survival.
